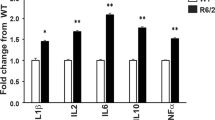
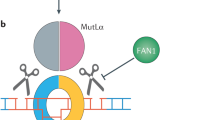
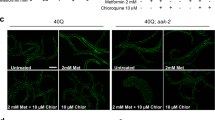
Abstract
Huntington's disease (HD) is an autosomal dominant neurological disease. It is a fatal neurological disorder affecting 5–10 out of 10 000 people. While there are intensive research efforts focusing on uncovering molecular mechanisms of the pathogenesis of HD, a number of studies have begun to look for effective therapies for HD. There is a large body of encouraging news on novel therapeutic developments. The present paper reviews drugs used for symptomatic treatment of HD and experimental therapies targeting HD molecular pathology.
Similar content being viewed by others
Article PDF
References
The Huntington's Disease Collaborative Research Group. A novel gene containing a trinucleotide repeat that is expanded and unstable on Huntington's disease chromosomes. Cell 1993; 72: 971–83.
Qin ZH, Gu ZL . Huntingtin processing in pathogenesis of Huntington disease. Acta Pharmacol Sin 2004; 25: 1243–9.
Hughes RE, Olson JM . Therapeutic opportunities in polyglutamine disease. Nature Med 2001; 7: 419–23.
Scherzinger E, Lurz R, Turmaine M, Mangiarini L, Hollenbach B, Hasenbank R, et al. Huntingtin-encoded polyglutamine expansions form amyloid-like protein aggregates in vitro and in vivo. Cell 1997; 90: 549–58.
Huang CC, Faber PW, Persichetti F, Mittal V, Vonsattel JP, MacDonald ME, et al. Amyloid, formation by mutant huntingtin: threshold, progressivity and recruitment of normal polyglutamine proteins. Somatic Cell Mol Gen 1998; 24: 217–33.
Scherzinger E, Sittler A, Schweiger K, Heiser V, Lurz R, Hasenbank R, et al. Self-assembly of polyglutamine-containing huntingtin fragments into amyloid-like fibrils: implications for Huntington's disease pathology. Proc Natl Acad Sci USA 1999; 96: 4604–9.
Davies SW, Turmaine M, Cozens BA, DiFiglia M, Sharp AH, Ross CA, et al. Formation of neuronal intranuclear inclusions underlies the neurological dysfunction in mice transgenic for the HD mutation. Cell 1997; 90: 537–48.
Heiser V, Scherzinger E, Boeddrich A, Nordhoff E, Lurz R, Schugardt N, et al. Inhibition of huntingtin fibrillogenesis by specific antibodies and small molecules: implications for Huntington's disease therapy. Proc Natl Acad Sci USA 2000; 97: 6739–44.
Pollitt SK, Pallos J, Shao J, Desai UA, Ma AA, Thompson LM, et al. A rapid cellular FRET assay of polyglutamine aggregation identifies a novel inhibitor. Neuron 2003; 40: 685–94.
Heiser V, Engemann S, Brocker W, Dunkel I, Boeddrich A, Waelter S, et al. Identification of benzothiazoles as potential polyglutamine aggregation inhibitors of Huntington's disease by using an automated filter retardation assay. Proc Natl Acad Sci USA 2002; 99: 16400–6.
Schiefer J, Landwehrmeyer GB, Luesse HG, Sprunken A, Puls C, Milkereit A, et al. Riluzole prolongs survival time and alters nuclear inclusion formation in a transgenic mouse model of Huntington's disease. Mov Disord 2002; 17: 4: 748–57.
Tanaka M, Machida Y, Niu S, Ikeda T, Jana NR, Doi H, et al. Trehalose alleviates polyglutamine-mediated pathology in a mouse model of Huntington disease. Nat Med 2004; 10: 148–54.
Wang J, MacDonald ME, Gusella JF . Reversal of a full-length mutant huntingtin neuronal cell phenotype by chemical inhibitors of polyglutamine-mediated aggregation. BMC Neurosci 2005; in press.
Sanchez I, Mahlke C, Yuan J . Pivotal role of oligomerization in expanded polyglutamine neurodegenerative disorders. Nature 2003; 421: 373–9.
Aiken CT, Tobin AJ, Schweitzer ES . A cell-based screen for drugs to treat Huntington's disease. Neurobiol Dis 2004; 16: 546–55.
Sittler A, Lurz R, Lueder G, Priller J, Lehrach H, Hayer-Hartl MK, et al. Geldanamycin activates a heat shock response and inhibits huntingtin aggregation in a cell culture model of Huntington's disease. Human Mol Genet 2001; 10: 1307–15.
Kahlem P, Green H, Djian P . Transglutaminase action imitates Huntington's disease: selective polymerization of Huntingtin containing expanded polyglutamine. Mol Cell 1998; 1: 595–601.
Karpuj MV, Garren H, Slunt H, Price DL, Gusella J, Becher MW, et al. Transglutaminase aggregates huntingtin into nonamyloidogenic polymers, and its enzymatic activity increases in Huntington's disease brain nuclei. Proc Natl Acad Sci USA 1999; 96: 7388–93.
Lorand L, Parameswaran KN, Stenberg P, Tong YS, Velasco PT, Jonsson NA, et al. Specificity of guinea pig liver transglutaminase for amine substrates. Biochemistry 1979; 18: 1756–65.
Dedeoglu A, Kubilus JK, Jeitner TM, Matson SA, Bogdanov M, Kowall NW, et al. Therapeutic effects of cystamine in a murine model of Huntington's disease. J Neurosci 2002; 22: 8942–50.
Karpuj MV, Becher MW, Springer JE, Chabas D, Youssef S, Pedotti R, et al. Prolonged survival and decreased abnormal movements in transgenic model of Huntington disease, with administration of the transglutaminase inhibitor cystamine. Nat Med 2002; 8: 143–9.
Goldberg YP, Nicholson DW, Rasper DM, Kalchman MA, Koide HB, Graham RK, et al. Cleavage of huntingtin by apopain, a proapoptotic cysteine protease, is modulated by the polyglutamine tract. Nat Gen 1996; 13: 442–9.
Wellington CL, Singaraja R, Ellerby L, Savill J, Roy S, Leavitt B, et al. Inhibiting caspase cleavage of huntingtin reduces toxicity and aggregate formation in neuronal and nonneuronal cells. J Biol Chem 2000; 275: 19831–8.
Kim YJ, Yi Y, Sapp E, Wang Y, Cuiffo B, Kegel KB, et al. Caspase 3-cleaved N-terminal fragments of wild-type and mutant huntingtin are present in normal and Huntington's disease brains, associate with membranes, and undergo calpaindependent proteolysis. Proc Natl Acad Sci USA 2001; 98: 12784–9.
Gafni J, Hermel E, Young JE, Wellington CL, Hayden MR, Ellerby LM . Inhibition of calpain cleavage of huntingtin reduces toxicity: accumulation of calpain/caspase fragments in the nucleus. J Biol Chem 2004; 279: 20211–20.
Lunkes A, Lindenberg KS, Ben-Haiem L, Weber C, Devys D, Landwehrmeyer GB, et al. Proteases acting on mutant huntingtin generate cleaved products that differentially build up cytoplasmic and nuclear inclusions. Mol Cell 2002; 10: 259–69.
DiFiglia M, Sapp E, Chase KO, Davies SW, Bates GP, Vonsattel JP, et al. Aggregation of huntingtin in neuronal intranuclear inclusions and dystrophic neurites in brain. Science 1997; 277: 1990–3.
Hodgson JG, Agopyan N, Gutekunst CA, Leavitt BR, LePiane F, Singaraja R, et al. A YAC mouse model for Huntington's disease with full-length mutant huntingtin, cytoplasmic toxicity, and selective striatal neurodegeneration. Neuron 1999; 23: 181–92.
Kim M, Lee HS, LaForet G, McIntyre C, Martin EJ, Chang P, et al. Mutant huntingtin expression in clonal striatal cells: dissociation of inclusion formation and neuronal survival by caspase inhibition. J Neurosci 1999; 19: 964–73.
Chen M, Ona VO, Li M, Ferrante RJ, Fink KB, Zhu S, et al. Minocycline inhibits caspase-1 and caspase-3 expression and delays mortality in a transgenic mouse model of Huntington disease. Nat Med 2000; 6: 797–801.
Wang X, Zhu S, Drozda M, Zhang W, Stavrovskaya IG, Cattaneo E, et al. Minocycline inhibits caspase-independent and -dependent mitochondrial cell death pathways in models of Huntington's disease. Proc Natl Acad Sci USA 2003; 100: 10483–7.
Kiechle T, Dedeoglu A, Kubilus J, Kowall NW, Beal MF, Friedlander RM, et al. Cytochrome C and caspase-9 expression in Huntington's disease. Neuromolecular Med 2002; 1: 183–95.
Ona VO, Li M, Vonsattel JP, Andrews LJ, Khan SQ, Chung WM, et al. Inhibition of caspase-1 slows disease progression in a mouse model of Huntington's disease. Nature 1999; 399: 263–7.
Sanchez I, Xu CJ, Juo P, Kakizaka A, Blenis J, Yuan J . Caspase 8 is required for cell death induced by expanded polyglutamine repeats. Neuron 1999; 22: 623–33.
Zuccato C, Ciammola A, Rigamonti D, Leavitt BR, Goffredo D, Conti L, et al. Loss of huntingtin-mediated BDNF gene transcription in Huntington's disease. Science 2001; 293: 493–8.
Li SH, Cheng AL, Zhou H, Lam S, Rao M, Li H, et al. Interaction of Huntington disease protein with transcriptional activator Sp1. Mol Cell Biol 2002; 22: 1277–87.
Li SH, Li XJ . Huntingtin-protein interactions and the pathogenesis of Huntington's disease. Trends Gen 2004; 20: 146–54.
Luthi-Carter R, Strand A, Peters NL, Solano SM, Hollingsworth ZR, Menon AS, et al. Decreased expression of striatal signaling genes in a mouse model of Huntington's disease. Human Mol Gen 2000; 9: 1259–71.
Chan EY, Luthi-Carter R, Strand A, Solano SM, Hanson SA, DeJohn MM, et al. Increased huntingtin protein length reduces the number of polyglutamine-induced gene expression changes in mouse models of Huntington's disease. Human Mol Gen 2002; 11: 1939–51.
Yu ZX, Li SH, Nguyen HP, Li XJ . Huntingtin inclusions do not deplete polyglutamine-containing transcription factors in HD mice. Human Mol Gen 2002; 11: 905–14.
Dunah AW, Jeong H, Griffin A, Kim YM, Standaert DG, Hersch SM, et al. Sp1 and TAFII130 transcriptional activity disrupted in early Huntington's disease. Science 2002; 296: 2238–43.
Steffan JS, Bodai L, Pallos J, Poelman M, McCampbell A, Apostol BL, et al. Histone deacetylase inhibitors arrest polyglutaminedependent neurodegeneration in Drosophila. Nature 2001; 413: 739–43.
Hockly E, Richon VM, Woodman B, Smith DL, Zhou X, Rosa E, et al. Suberoylanilide hydroxamic acid, a histone deacetylase inhibitor, ameliorates motor deficits in a mouse model of Huntington's disease. Proc Natl Acad Sci USA 2003; 100: 2041–6.
Minamiyama M, Katsuno M, Adachi H, Waza M, Sang C, Kobayashi Y, et al. Sodium butyrate ameliorates phenotypic expression in a transgenic mouse model of spinal and bulbar muscular atrophy. Hum Mol Genet 2004; 13: 1183–92.
Alvarez RD, Barnes MN, Gomez-Navarro J, Wang M, Strong TV, Arafat W, et al. A cancer gene therapy approach utilizing an anti-erbB-2 single-chain antibody-encoding adenovirus (AD21): a phase I trial. Clin Cancer Res 2000; 6: 3081–7.
Marasco WA, Haseltine WA, Chen SY . Design, intracellular expression, and activity of a human anti-human immunodeficiency virus type 1 gp120 single-chain antibody. Proc Natl Acad Sci USA 1993; 90: 7889–93.
Lecerf JM, Shirley TL, Zhu Q, Kazantsev A, Amersdorfer P, Housman DE, et al. Human single-chain Fv intrabodies counteract in situ huntingtin aggregation in cellular models of Hunting-ton's disease. Proc Natl Acad Sci USA 2001; 98: 4764–9.
Colby DW, Garg P, Holden T, Chao G, Webster JM, Messer A, et al. Development of a human light chain variable domain (V(L)) intracellular antibody specific for the amino terminus of huntingtin via yeast surface display. J Mol Biol 2004; 342: 901–12.
Khoshnan A, Ko J, Patterson PH . Effects of intracellular expression of anti-huntingtin antibodies of various specificities on mutant huntingtin aggregation and toxicity. Proc Natl Acad Sci USA 2002; 99: 1002–7.
Xia H, Mao Q, Paulson HL, Davidson BL . siRNA-mediated gene silencing in vitro and in vivo. Nat Biotechnol 2002; 20: 1006–10.
Caplen NJ, Taylor JP, Statham VS, Tanaka F, Fire A, Morgan RA . Rescue of polyglutamine-mediated cytotoxicity by doublestranded RNA-mediated RNA interference. Hum Mol Gen 2002; 11: 175–84.
Xia H, Mao Q, Eliason SL, Harper SQ, Martins IH, Orr HT, et al. RNAi suppresses polyglutamine-induced neurodegeneration in a model of spinocerebellar ataxia. Nat Med 2004; 10: 816–20.
Barbacid M . The Trk family of neurotrophin receptors. J Neurobiol 1994; 25: 1386–403.
Koroshetz WJ, Jenkins BG, Rosen BR, Beal MF . Energy metabolism defects in Huntington's disease and effects of coenzyme Q10. Ann Neurol 1997; 41: 160–5.
Winn SR, Lindner MD, Lee A, Haggett G, Francis JM, Emerich DF . Polymer-encapsulated genetically modified cells continue to secrete human nerve growth factor for over one year in rat ventricles: behavioral and anatomical consequences. Exp Neurol 1996; 40: 126–38.
Qin ZH, Wang Y, Chase TN . Stimulation of NMDA receptors induces apoptosis in rat brain. Brain Res 1996; 725: 166–76.
Bemelmans AP, Horellou P, Pradier L, Brunet I, Colin P, Mallet J . Brain-derived neurotrophic factor-mediated protection of striatal neurons in an excitotoxic rat model of Huntington's disease, as demonstrated by adenoviral gene transfer. Hum Gene Ther 1999; 10: 2987–97.
Pérez-Navarro F, Canudas, AM, Åkerud, P, Alberch J, Arenas E . Brain-derived neurotrophic factor, neurotrophin-3, and neuro-trophin-4/5 prevent the death of striatal projection neurons in a rodent model of Huntington's disease. J Neurochem 2000; 75: 2190–9.
Emerich DF, Winn SR, Hantraye PM, Peschanski M, Chen EY, Chu Y, et al. Protective effect of encapsulated cells producing neurotrophic factor CNTF in a monkey model of Huntington's disease. Nature 1997; 386: 395–9.
McBride JL, During MJ, Wuu J, Chen EY, Leurgans SE, Kordowera JH . Structural and functional neuroprotection in a rat model of Huntington's disease by viral gene transfer of GDNF. Exp Neurol 2003; 181: 213–23.
Kells AP, Fong DM, Dragunow M, During MJ, Young D, Connor B, AAV-mediated gene delivery of BDNF or GDNF is neuroprotective in a model of Huntington disease. Mol Therapy 2004; 9: 682–8.
Mittoux V, Joseph JM, Conde F, Palfi S, Dautry C, Poyot T, et al. Restoration of cognitive and motor functions by ciliary neurotrophic factor in a primate model of Huntington's disease. Hum Gene Ther 2000; 20; 11: 1177–87.
Isacson O, Brundin P, Gage FH, Bjorklund A . Neural grafting in a rat model of Huntington's disease: progressive neurochemical changes after neostriatal ibotenate lesions and striatal tissue grafting. Neuroscience 1985; 16: 799–817.
Watts C, Brasted PJ, Dunnett SB . The morphology, integration, and functional efficacy of striatal grafts differ between cell suspensions and tissue pieces. Cell Transplant 2000; 9: 395–407.
Palfi S, Conde F, Riche D, Brouillet E, Dautry C, Mittoux V, et al. Fetal striatal allografts reverse cognitive deficits in a primate model of Huntington disease. Nat Med 1998; 4: 963–6.
Armstrong RJ, Watts C, Svendsen CN, Dunnett SB, Rosser AE . Survival, neuronal differentiation, and fiber outgrowth of propagated human neural precursor grafts in an animal model of Huntington's disease. Cell Transplant 2000; 9: 55–64.
Xu ZC, Wilson CJ, Emson PC . Restoration of thalamostriatal projections in rat neostriatal grafts: an electron microscopic analysis. J Comp Neurol 1991; 303: 22–34.
Chena GJ, Jengb CH, Lina SZ, Tsaic SH, Wangd Y, Chianga YH . Fetal striatal transplants restore electrophysiological sensitivity to dopamine in the lesioned striatum of rats with experimental Huntington's disease. J Biomed Sci 2002; 9: 303–10.
Dunnett SB, Carter RJ, Watts C, Torres EM, Mahal A, Mangiarini L, et al. Striatal transplantation in a transgenic mouse model of Huntington's disease. Exp Neurol 1998; 154: 31–40.
Madrazo I, Franco-Bour RE, Castrejon H, Cuevas C, Ostrosky-Solis F . Fetal striatal homotransplantation for Huntington's disease: first two case reports. Neurol Res 1995; 17: 312–5.
Philpott LM, Kopyov OV, Lee AJ, Jacques S, Duma CM, Caine S, et al. Neuropsychological functioning following fetal striatal transplantation in Huntington's chorea: three case presentations. Cell Transplant 1997; 6: 203–12.
Kopyov OV, Jacques S, Lieberman A, Duma CM, Eagle KS . Safety of intrastriatal neurotransplantation for Huntington's disease patients. Exp Neurol 1998; 49: 97–108.
Freemana TB, Cicchettid F, Hauserb RA, Deaconf TW, Li XJ, Herschi SM, et al. Transplanted fetal striatum in Huntington's disease: phenotypic development and lack of pathology. Proc Natl Acad Sci USA 2000; 97: 13877–82.
Gaura VR, Bachoud-Levi AC, Ribeiro MJ, Nguyen JP, Frouin V, Baudic S, et al. Striatal neural grafting improves cortical metabolism in Huntington's disease patients. Brain 2004; 127: 65–72.
Bachoud-Lévi AC, Nguyen JP, Brugières P, Lefaucheur JP, Cesaro, P. P . Rémy, P, et al. Motor and cognitive improvements in patients with Huntington's disease after neural transplantation. Lancet 2000; 356: 1975–9.
Bachoud-Lévi AC, Bourdet C, Brugiéres P, Nguyen JP, Grandmougin T, Haddad B, et al. Safety and tolerability assessment of intrastriatal neural allografts in five patients with Huntington's disease. Exp Neurol 2000; 161: 194–202.
Fink JS, Schumacher JM, Ellias SL, Palmer EP, Saint-Hilaire M, Shannon K, et al. Porcine xenografts in Parkinson's disease and Huntington's disease patients: preliminary results. Cell Transplant 2000; 9: 273–8.
Rosser AE, Barker RA, Harrower T, Watts C, Farrington M, Ho AK, et al. Unilateral transplantation of human primary fetal tissue in four patients with Huntington's disease: NEST-UK safety report. J Neurol Neurosurg Psychiatry 2002; 73: 678–85.
Hauser RA, Furtado S, Cimino CR, Delgado H, Eichler S, Schwartz S, et al. Bilateral human fetal striatal transplantation in Huntington's disease. Neurology 2002; 58: 687–95.
Beal MF . Coenzyme Q10 administration and its potential for treatment of neurodegenerative diseases. BioFactors 1999; 9: 261–6.
Schillinga G, Coonfielda ML, Rossb CA, Borchelta DR . Coenzyme Q10 and remacemide hydrochloride ameliorate motor deficits in a Huntington's disease transgenic mouse model. Neurosci Lett 2001; 315: 149–53.
Ferrante RJ, Andreassen OA, Dedeoglu A, Ferrante AL, Jenkins BG, Hersch SM, et al. Therapeutic effects of coenzyme Q10 and remacemide in transgenic mouse models of Huntington's disease. J Neurosci 2002; 22: 1592–9.
Huntington Study Group. A randomized, placebo-controlled trial of coenzyme Q10 and remacemide in Huntington's disease. Neurology 2001; 57: 397–404.
Andreassen OA, Dedeoglu A, Ferrante RJ, Jenkins BG, Ferrante KL, Homas M, et al. Creatine increases survival and delays motor symptoms in a transgenic animal model of Huntington's disease. Neurobiol Disease 2001; 8: 479–91.
Dedeoglu A, Kubilus JK, Yang L, Ferrante KL, Hersch SM, Beal MF, et al. Creatine therapy provides neuroprotection after onset of clinical symptoms in Huntington's disease transgenic mice. J Neurochem 2003; 85: 1359–67.
Verbessem P, Lemiere J, Eijnde BO, Swinnen S, Vanhees L, van Leemputte M, et al. Creatine supplementation in Huntington's disease: a placebo-controlled pilot trial. Neurology 2003; 61: 925–30.
van Dellen A, Blakemore C, Deacon R, York D, Hannan AJ . Delaying the onset of Huntington's in mice. Nature 2000; 404: 721–2.
Glass M, van Dellen A, Blakemore CC, Hannand CAJ, Faullb RLM . Delayed onset of Huntington's disease in mice in an enriched environment correlates with delayed loss of cannabinoid CB1 receptors. Neuroscience 2004; 123: 207–12.
Hockly E, Cordery PM, Woodman B, Mahal A, van Dellen A, Blakemore C, et al. Environmental enrichment slows disease progression in R6/2 Huntington's disease mice. Ann Neurol 2002; 51: 235–42.
Spires TL, Grote HE, Varshney NK, Cordery PM, van Dellen A, Blakemore C, et al. Environmental enrichment rescues protein deficits in a mouse model of Huntington's disease, indicating a possible disease mechanism. J Neurosci 2004; 24: 2270–6.
Duan W, Guo Z, Jiang H, Ware M, Li X-J, Mattson MP . Dietary restriction normalizes glucose metabolism and BDNF levels, slows disease progression, and increases survival in huntingtin mutant mice. Proc Natl Acad Sci USA 2003; 100: 2911–6.
Leroi I, Michalon M . Treatment of the psychiatric manifestations of Huntington's disease: a review of the literature. Can J Psychiatry 1998; 43: 933–40.
Craufurd D, Thompson JC, Snowden JS . Behavioral changes in Huntington disease. Neuropsychiatry Neuropsychol Behav Neurol 2001; 14: 219–26.
Girotti F, Carella F, Scigliano G, Grassi MP, Soliveri P, Giovannini P, et al. Effect of neuroleptic treatment on involuntary movements and motor performances in Huntington's disease. J Neurol Neurosurg Psychiatry 1984; 47: 848–52.
Barr AN, Fischer JH, Koller WC, Spunt AL, Singhal A . Serum ha loperidol concentration and choreiform movements in Huntington's disease. Neurology 1988; 38: 84–8.
Erdemoglua AK, Boratavb C . Risperidone in chorea and psychosis of Huntington's disease. Eur J Neurol 2001; 9: 177–85.
Rosas HD, Koroshetz WJ, Jenkins BG, Chen YI, Hayden DL, Beal MF, et al. Riluzole therapy in Huntington's disease (HD). Mov Disorders 1999; 14: 326–30.
Seppi K, Mueller J, Bodner T, Brandauer E, Benke T, Weirich-Schwaiger H, et al. Riluzole in Huntington's disease (HD): an open label study with one year follow up. J Neurol 2001; 248: 866–9.
Bonelli RM, Niederwieser G, Diez J, Tringer PK . Riluzole and olanzapine in Huntington's disease. Eur J Neurol 2001; 9: 177–85.
Verhagen Metman, L, Morris MJ, Farmer C, Gillespie M, Mosby K, Wuu J, et al. Huntington's disease: a randomized, controlled trial using the NMDA-antagonist amantadine. Neurology 2002; 59: 694–9.
Lucetti C, Del Dotto, P, Gambaccini G, Dell' Agnello, G, Bernardini S, Rossi G, et al. IV amantadine improves chorea in Huntington's disease: an acute randomized, controlled study. Neurology 2003; 60: 1995–7.
Stewart JT, Mounts ML, Clark, RL Jr . Aggressive behavior in Huntington's disease: treatment with propranolol. J Clin Psychiatry 1987; 48: 106–8.
Leonard DP, Kidson MA, Brown JG, Shannon PJ, Taryan S . A double blind trial of lithium carbonate and haloperidol in Huntington's chorea. Aust N Z J Psychiatry 1975; 9: 115–8.
Parsa, MA, Szigethy E, Voci JM, Meltzer HY . Risperidone in treatment of choreoathetosis of Huntington's disease. J Clin Psychopharmacol 1997; 17: 134–5.
Dipple HC . The use of olanzapine for movement disorder in Huntington's disease: a first case report. J Neurol Neurosurg Psychiatry 1999; 67: 123–4.
Paleacu D, Anca M, Giladi N . Olanzapine in Huntington's disease. Acta Neurol Scand 2002; 105: 441–4.
Zhang L, Zhang RY, Li L, Wang H, Xia J . Effect of Chinese compound 962 on behavior deficit in Huntington's disease model rats. Chin J Behav Med Sci 2002; 3: 243–6. Chinese.
Zhang RY, Zhang L, Wang H, Yei CF . Effects of Shen-wu capsule on motor function andcontent of dopamine in striatum in Huntington model rats. Chin J Rehabil 2002; 9: 513–5. Chinese.
Zhang RY, Li L, Zhang L, Wang H, Yei CF . Effects of Shenwu capsule on the behavior and neurotrophic factors in the rats with mimic Huntington's disease. J Beijing University TCM 2003; 26: 34–8.
Albin RL . Fetal striatal transplantation in Huntington's disease: time for a pause. [editorial commentaries: huntington's disease]. J Neurol Neurosurg Psychiatry 2002; 73: 612.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China (No 30370506).
Rights and permissions
About this article
Cite this article
Qin, Zh., Wang, J. & Gu, Zl. Development of novel therapies for Huntington's disease: hope and challenge. Acta Pharmacol Sin 26, 129–142 (2005). https://doi.org/10.1111/j.1745-7254.2005.00520.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2005.00520.x