Abstract
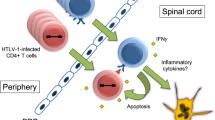
Human T-cell lymphotropic virus type I (HTLV-I) infection is associated with a variety of human diseases. In particular, there are two major diseases caused by HTLV-I infection. One is an aggressive neoplastic disease called adult T-cell leukemia (ATL), and another is a chronic progressive inflammatory neurological disease called HTLV-I-associated myelopathy/tropical spastic paraparesis (HAM/TSP). It is still unknown why one virus causes these different diseases. With regard to HAM/TSP, virus-host immunological interactions are an considered to be important cause of this disease. Coexisting high HTLV-I proviral load and HTLV-I-specific T cells (CD4+ T cells and CD8+ T cells) is an important feature of HAM/TSP. Histopathological studies indicate the existence of an inflammatory reaction and HTLV-I-infected cells in the affected lesions of HAM/TSP. Therefore, the immune response to HTLV-I probably contributes to the inflammatory process of the central nervous system lesions in HAM/TSP patients.
Similar content being viewed by others
References
Abe M, Umehara F, Kubota R, Moritoyo T, Izumo S, Osame M (1999). Activation of macrophage/microglias with expression of MRP8 and MRP14 is associated with the lesional activities in the spinal cord lesions in HTLV-I associated myelopathy. J Neurol 246: 358–364.
Aye MM, Matsuoka E, Moritoyo T, et al (2000). Histopathological analysis of four autopsy cases of HTLV-I-associated myelopathy/tropical spastic paraparesis: inflammatory changes occur simultaneously in the entire central nervous system. Acta Neuropathol 100: 245–252.
Azimi N, Nagai M, Jacobson S, Waldmann TA (2001). IL-15 plays a major role in the persistence of Tax-specific CD8 cells in HAM/TSP patients. Proc Natl Acad Sci USA 98: 14559–14564.
Biddison WE, Kubota R, Kawanishi T, et al (1997). Human T cell leukemia virus type I (HTLV-I)—specific CD8+ CTL clones from patients with HTLV-I—associated neurologic disease secrete proinflammatory cytokines, chemokines, and matrix metalloproteinase. J Immunol 159: 2018–2025.
Bieganowska K, Hollsberg P, Buckle GJ, et al (1999). Direct analysis of viral-specific CD8+ T cells with soluble HLA-A2/Tax11-19 tetramer complexes in patients with human T cell lymphotropic virus-associated myelopathy. J Immunol 162: 1765–1771.
Cavrois M, Gessain A, Gout O, Wain-Hobson S, Wattel E (2000). Common human T cell leukemia virus type 1 (HTLV-1) integration sites in cerebrospinal fluid and blood lymphocytes of patients with HTLV-1—associated myelopathy/tropical spastic paraparesis indicate that HTLV-1 crosses the blood-brain barrier via clonal HTLV-1-infected cells. J Infect Dis 82: 1044–1050.
Furukawa Y, Yamashita M, Usuku K, Izumo S, Nakagawa M, Osame M (2000). Phylogenetic subgroups of HTLV-I in tax gene and their association with different risk to HTLV-I—associated myelopathy/tropical spastic parapraresis. J Infect Dis 182: 1343–1349.
Furuya T, Nakamura T, Shirabe S, et al (1997). Heightened transmigrating activity of CD4-positive T cells through reconstituted basement membrane in patients with human T-lymphotropic virus type I-associated myelopathy. Proc Assoc Am Physicians 109: 228–236.
Gessain A, Barin F, Vernant JC, et al (1985). Antibodies to human T-lymphotropic virus type-I in patients with tropical spastic paraparesis. Lancet 2: 407–410.
Goon PK, Hanon E, Igakura T, et al (2002). High frequencies of Th1-type CD4+ T cells specific to HTLV-1 Env and Tax proteins in patients with HTLV-1-associated myelopathy/tropical spastic paraparesis. Blood 99: 3335–3341.
Grant C, Barmak K, Alefantis T, et al (2002). Human T cell leukemia virus type I and neurologic disease: events in bone marrow, peripheral blood, and central nervous system during normal immune surveillance and neuroinflammation. J Cell Physiol 190: 133–159.
Greten TF, Slansky JE, Kubota R, et al (1998). Direct visualization of antigen-specific T cells: HTLV-1 Tax11-19-specific CD8+ T cells are activated in peripheral blood and accumulate in cerebrospinal fluid from HAM/TSP patients. Proc Natl Acad Sci USA 95: 7568–7573.
Hanon E, Goon P, Taylor GP, et al (2001). High production of interferon gamma but not interleukin-2 by human T-lymphotropic virus type I-infected peripheral blood. Blood 98: 721–726.
Hanon E, Stinchcombe JC, Saito M, et al (2000). Fratricide among CD8+ T lymphocytes naturally infected with human T cell lymphotropic virus type I. Immunity 13: 657–664.
Hara H, Morita M, Iwaki T, et al (1994). Detection of human T lymphotrophic virus type I (HTLV-I) proviral DNA and analysis of T cell receptor V beta CDR3 sequences in spinal cord lesions of HTLV-I-associated myelopathy/tropical spastic paraparesis. J Exp Med 180: 831–839.
Hausmann S, Biddison WE, Smith KJ, et al (1999). Peptide recognition by two HLA-A2/Tax11-19-specific T cell clones in relationship to their MHC/peptide/TCR crystal structures. J Immunol 162: 5389–5397.
Izumo S, Ijichi T, Higuchi I, Tashiro A, Takahashi K, Osame M (1992). Neuropathology of HTLV-I-associated myelopathy: a report of two autopsy cases. Acta Paediatr Jpn 34: 358–364.
Jacobson S, Shida H, McFarlin DE, Fauci AS, Koenig S (1990). Circulating CD8+ cytotoxic lymphocytes specific for HTLV-I in patients with HTLV-I associated neurological disease. Nature 348: 245–248.
Jeffery KJ, Siddiqui AA, Bunce M, et al (2000). The influence of HLA class I alleles and heterozygosity on the outcome of human T cell lymphotropic virus type I infection. J Immunol 165: 7278–7284.
Jeffery KJM, Usuku K, Hall SE, et al (1999). HLA alleles determine human T-lymphotropic virus-I (HTLV-I) proviral load and the risk of HTLV-I-associated myelopathy. Proc Natl Acad Sci USA 96: 3848–3853.
Kaplan J, Osame M, Kubota H (1990). The risk of developing HTLV-I associated myelopathy/tropical spastic paraparesis (HAM/TSP) among persons infected with HTLV-I. J AIDS 3: 1096–1101.
Kira J, Fujihara K, Itoyama Y, Goto I, Hasuo K (1991). Leukoencephalopathy in HTLV-I-associated myelopathy/tropical spastic paraparesis: MRI analysis and a two year follow-up study after corticosteroid therapy. J Neurol Sci 106: 41–49.
Koenig S, Woods RM, Brewah YA, et al (1993). Characterization of MHC class I restricted cytotoxic T cell responses to tax in HTLV-1 infected patients with neurologic disease. J Immunol 151: 3874–3883.
Kubota R, Kawanishi T, Matsubara H, Manns A, Jacobson S (1998). Demonstration of human T lymphotropic virus type I (HTLV-I) tax-specific CD8+ lymphocytes directly in peripheral blood of HTLV-I—associated myelopathy/tropical spastic paraparesis patients by intracellular cytokine detection. J Immunol 161: 482–488.
Kubota R, Osame M, Jacobson S (2000). Retrovirus: human T-cell lymphotropic virus type I-associated diseases and immune dysfunction. In: Effects of microbes on the immune system. Cunningham MW, Fujinami RS (eds). Philadelphia: Lippincott Williams & Wilkins, pp 349–371.
Kubota R, Umehara F, Izumo S, et al (1994). HTLV-I proviral DNA amount correlates with infiltrating CD4+ lymphocytes in the spinal cord from patients with HTLV-I-associated myelopathy. J Neuroimmunol 53: 23–29.
Lehky TJ, Flerlage N, Katz D, et al (1996). Human T-cell lymphotropic virus type II-associated myelopathy: clinical and immunologic profiles. Ann Neurol 40: 714–723.
Lehky TJ, Fox CH, Koenig S, et al (1995). Detection of human T-lymphotropic virus type I (HTLV-I) tax mRNA in the central nervous system of HTLV-I—associated myelopathy/tropical spastic paraparesis patients by in situ hybridization. Ann Neurol 37: 167–175.
Levin MC, Krichavsky M, Berk J, et al (1998). Neuronal molecular mimicry in immune-mediated neurologic disease. Ann Neurol 44: 87–98.
Levin MC, Lee SM, Kalume F, et al (2002). Autoimmunity due to molecular mimicry as a cause of neurological disease. Nat Med 8: 509–513.
Makino M, Shimokubo S, Wakamatsu SI, Izumo S, Baba M (1999). The role of human T-lymphotropic virus type 1 (HTLV-1)-infected dendritic cells in the development of HTLV-1—associated myelopathy/tropical spastic paraparesis. J Virol 73: 4575–4581.
Matsuoka E, Takenouchi H, Hashimoto K, et al (1998). Perivascular T-cells are infected with HTLV-I in the spinal cord lesions with HAM/TSP: double staining of immunohistochemistry and PCR in situ hybridization. Acta Neuorpathol 96: 340–346.
Matsuoka E, Usuku K, Jonosono M, Takenouchi N, Izumo S, Osame M (2000). CD44 splice variant involvement in the chronic inflammatory disease of the spinal cord: HAM/TSP. J Neuroimmunol 102: 1–7.
Mor F, Cohen IR (1992). T cells in the lesion of experimental autoimmune encephalomyelitis. Enrichment for reactivities to myelin basic protein and to heat shock proteins. J Clin Invest 90: 2447–2455.
Moritoyo T, Reinhart TA, Moritoyo H, et al (1996). Human T-lymphotropic virus type I-associated myelopathy and tax gene expression in CD4C T lymphocytes. Ann Neurol 40: 84–90.
Nagai M, Brennan MB, Sakai JA, Mora CA, Jacobson S (2001a). CD8C T cells are an in vivo reservoir for human T-cell lymphotropic virus type I. Blood 98: 1858–1861.
Nagai M, Kubota R, Greten TF, Schneck JP, Leist TP, Jacobson S (2001b). Increased activated human T cell lymphotropic virus type I (HTLV-I) Tax11—19-specific memory and effector CD8+ cells in patients with HTLV-I-associated myelopathy/tropical spastic paraparesis: correlation with HTLV-I provirus load. J Infect Dis 183: 197–205.
Nagai M, Usuku K, Matsumoto W, et al (1998). Analysis of HTLV-I proviral load in 202 HAM/TSP patients and 243 asymptomatic HTLV-I carriers: high proviral load strongly predisposes to HAM/TSP. J NeuroVirol 4: 586–593.
Nagai M, Yamano Y, Brennan MB, Mora CA, Jacobson S (2001c). Increased HTLV-I proviral load and preferential expansion of HTLV-I Tax-specific CD8+ T cells in cerebrospinal fluid from patients with HAM/TSP. Ann Neurol 50: 807–812.
Nagai M, Yashiki S, Fujiyoshi T, et al (1996). Characterization of a unique T-cell clone established from a patient with HAM/TSP which recognized HTLV-I-infected T-cell antigens as well as spinal cord tissue antigens. J Neuroimmunol 65: 97–105.
Nakamura T, Furuya T, Nishiura Y, Ichinose K, Shirabe S, Eguchi K (2000). Importance of immune deviation toward Th1 in the early immunopathogenesis of human T-lymphotropic virus type I-associated myelopathy. Med Hypotheses 54: 777–782.
Osame M, Usuku K, Izumo S, et al (1986). HTLV-I associated myelopathy, a new clinical entity [letter]. Lancet 1: 1031–1032.
Parker CE, Nightingale S, Taylor GP, Weber J, Bangham CR (1994). Circulating anti-Tax cytotoxic T lymphocytes from human T-cell leukemia virus type I-infected people, with and without tropical spastic paraparesis, recognize multiple epitopes simultaneously. J Virol 68: 2860–2868.
Richardson JH, Edwards AJ, Cruickshank JK, Rudge P, Dalgleish AG (1990). In vivo cellular tropism of human T-cell leukemia virus type 1. J Virol 64: 5682–5687.
Sakai JA, Nagai M, Brennan MB, Mora CA, Jacobson S (2001). In vitro spontaneous lymphoproliferation in patients with human T-cell lymphotropic virus type I-associated neurologic disease: predominant expansion of CD8+ T cells. Blood 98: 1506–1511.
Silva EA, Otsuki K, Leite AC, et al (2002). HTLV-II infection associated with a chronic neurodegenerative disease: clinical and molecular analysis. J Med Virol 66: 253–257.
Szymocha R, Akaoka H, Dutuit M, et al (2000). Human T-cell lymphotropic virus type 1-infected T lymphocytes impair catabolism and uptake of glutamate by astrocytes via Tax-1 and tumor necrosis factor alpha. J Virol 74: 6433–6441.
Umehara F, Abe M, Koeeda Y, Izumo S, Osame M (2000). Axonal damage revealed by accumulation of β-amyloid precursor protein in HTLV-I—associated myelopathy. J Neurol Sci 176: 95–101.
Umehara F, Izumo S, Nakagawa M, et al (1993). Immunocytochemical analysis of the cellular infiltrate in the spinal cord lesions in HTLV-I—associated myelopathy. J Neuropathol Exp Neurol 52: 424–430.
Umehara F, Izumo S, Ronquillo AT, Matsumuro K, Osame M (1994a). Cytokine expression in the spinal cord lesions in HTLV-I-associated myelopathy. J Neuropathol Exp Neurol 53: 72–77.
Umehara F, Izumo S, Takeya M, Sato E, Osame M (1996). Expression of adhesion molecules and monocyte chemoattractant protein-1(MCP-1) in the spinal cord lesions in HTLV-I-associated myelopathy. Acta Neuropathol 91: 343–350.
Umehara F, Nakamura A, Izumo S, et al (1994b). Apoptosis of T-lymphocytes in the spinal cord lesions in HTLV-I-associated myelopathy: a possible mechanism to control viral infection in the central nervous system. J Neuropathol Exp Neurol 53: 617–624.
Umehara F, Okada Y, Fujimoto N, Abe M, Izumo S, Osame M (1998). Expression of matrix metalloproteinases and tissue inhibitors of metalloproteinases in HTLV-I-associated myelopathy. J Neuropathol Exp Neurol 57: 839–849.
Usuku K, Sonoda S, Osame M, et al (1988). HLA haplotype-linked high immune responsiveness against HTLV-I in HTLV-I—associated myelopathy: comparison with adult T-cell leukemia/lymphoma. Ann Neurol 23: S143-S150.
Vine AM, Witkover AD, Lloyd AL, et al (2002). Polygenic control of human T lymphotropic virus type I (HTLV-I) provirus load and the risk of HTLV-I-associated myelopathy/tropical spastic paraparesis. J Infect Dis 186: 932–939.
Yamano Y, Nagai M, Brennan M, et al (2001). Correlation of human T-cell lymphotropic virus type 1 (HTLV-1) mRNA with proviral DNA load, virus-specific CD8+ T cells, and disease severity in HTLV-I-associated myelpathy (HAM/TSP). Blood 99: 88–94.
Yashiki S, Fujiyoshi T, Arima N, et al (2001). HLA-A*26, HLA-B*4002, HLA-B*4006, and HLA-B*4801 alleles predispose to adult T cell leukemia: the limited recognition of HTLV type I tax peptide anchor motifs and epitopes to generate anti-HTLV type 1 tax CD8(+) cytotoxic T lymphocytes. AIDS Res Hum Retroviruses 17: 1047–1061.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nagai, M., Osame, M. Human T-cell lymphotropic virus type I and neurological diseases. Journal of NeuroVirology 9, 228–235 (2003). https://doi.org/10.1080/13550280390194028
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1080/13550280390194028