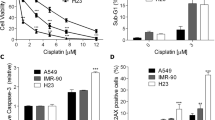
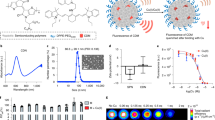
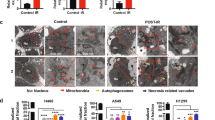
Abstract
Increased expression of manganese superoxide dismutase (Mn-SOD), one of the mitochondrial enzymes involved in the redox system, has been shown to diminish the cytotoxic effects of several anti-cancer modalities, including tumour necrosis factor-α, ionizing radiation, certain chemotherapeutic agents and hyperthermia. We asked if Mn-SOD is a potential target to augment the sensitivity of cancer cells to various anti-cancer treatments and for this we established stable Mn-SOD antisense RNA expressing cell clones from two human colon cancer cell lines, HCT116 (p53 wild-type) and DLD1 (p53 mutant-type). Suppression of Mn-SOD in HCT116 was accompanied by an increased sensitivity to radiation, hyperthermia and doxorubicin, as compared with findings in controls. The mitochondrial permeability transition, as measured by a decrease of the mitochondrial transmembrane potential was more intensely induced by radiation in HCT116 antisense clones than in the control, an event followed by a greater extent of DNA fragmentation. Apoptosis was also induced by hyperthermia more intensely in HCT116 antisense clones than in the control. On the other hand, DLD1 antisense clones did not exhibit any enhancement of sensitivity to any of these treatments. These data support the possibility that inhibition of Mn-SOD activity renders colon cancer cells with wild-type p53 susceptible to apoptosis induced by radiation, hyperthermia and selected anti-cancer drugs. Therefore, we suggest that Mn-SOD could be a target molecule to overcome the resistance to anti-cancer treatments in some colon cancer cells carrying wild-type p53. © 2000 Cancer Research Campaign
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Beauchamp C and Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44: 276–287
Buttke TM and Sandstrom PA (1994) Oxidative stress as a mediator of apoptosis. Immunol Today 15: 7–10
Church SL, Grant JW, Ridnour LA, Oberley LW, Swanson PE, Meltzer PS and Trent JM (1993) Increased manganese superoxide dismutase expression suppresses the malignant phenotype of human melanoma cells. Proc Natl Acad Sci USA 90: 3113–3117
Cobbs CS, Levi DS, Aldape K and Israel MA (1996) Manganese superoxide dismutase expression in human central nervous system tumours. Cancer Res 56: 3192–3195
Fisher DE (1994) Apoptosis in cancer therapy: crossing the threshold. Cell 73: 539–542
Fridovich I (1997) Superoxide anion radical (O2·–), superoxide dismutases, and related matters. J Biol Chem 272: 18515–18517
Fujimura M, Morita-Fujimura Y, Kawase M, Copin JC, Calagui B, Epstein CJ and Chan PH (1999) Manganese superoxide dismutase mediates the early release of mitochondrial cytochrome c and subsequent DNA fragmentation after permanent focal cerebral ischemia in mice. J Neurosci 19: 3412–3422
Graeber TG, Osmanian C, Jacks T, Housman DE, Koch CJ, Lowe SW and Giaccia AJ (1996) Hypoxia-mediated selection of cells with diminished apoptotic potential in solid tumours. Nature 379: 88–91
Green DR and Reed JC (1998) Mitochondria and apoptosis. Science 281: 1309–1312
Higuchi M, Honda T, Proske RJ and Yeh ETH (1998) Regulation of reactive oxygen species-induced apoptosis and necrosis by caspase 3-like proteases. Oncogene 17: 2753–2760
Hirose K, Longo DL, Oppenheim JJ and Matsushima K (1993) Overexpression of mitochondrial manganese superoxide dismutase promotes the survival of tumour cells exposed to interleukin-1, tumour necrosis factor, selected anticancer drugs, and ionising radiation. FASEB J 7: 361–368
Hotz HA, Gong J, Traganos F and Darzynkiewicz Z (1994) Flow cytometric detection of apoptosis: comparison of the assay of in situ DNA degradation and chromatin changes. Cytometry 15: 237–244
Janssen YMW, Houten BV, Borm PJA and Mossman BT (1993) Cell and tissue responses to oxidative damage. Lab Invest 69: 261–274
Janssen AML, Bosman CB, Sier CFM, Griffioen G, Kubben FJGM, Lamers CBHW, van Krieken JHJM, van de Velde CJH and Verspaget HW (1998) Superoxide dismutases in relation to the overall survival of colorectal cancer patients. Br J Cancer 78: 1051–1057
Johnson TM, Yu Z-X, Ferrans VJ, Lowenstein RA and Finkel T (1996) Reactive oxygen species are downstream mediators of p53-dependent apoptosis. Proc Natl Acad Sci USA 93: 11848–11852
Kastan MB, Onyekwere O, Sidransky D, Vogelstein B and Craig RW (1991) Participation of p53 protein in the cellular response to DNA damage. Cancer Res 51: 6304–6311
Keyer K and Imlay JA (1996) Superoxide accelerates DNA damage by elevating free-iron levels. Proc Natl Acad Sci USA 93: 13635–13640
Kinnula VL, Pietarinen-Runtti P, Raivio K, Kahlos K, Pelin K, Mattson K and Linnainmaa K (1996) Manganese superoxide dismutase in human pleural mesothelioma cell lines. Free Radical Biol Med 21: 527–532
Landriscina M, Remiddi F, Ria F, Palazzotti B, De Leo ME, Iacoangeil M, Rosselli R, Scerrati M and Galeotti T (1996) The level of MnSOD is directly correlated with grade of brain tumours of neuroepithelial origin. Br J Cancer 74: 1877–1885
Li JJ and Oberley LW (1997) Overexpression of manganese-containing superoxide dismutase confers resistance to the cytotoxicity of tumor necrosis factor α and/or hyperthermia. Cancer Res 57: 1991–1998
Li JJ, Oberley LW, St. Clair DK, Ridnour LA and Oberley TD (1995) Phenotypic changes induced in human breast cancer cells by overexpression of manganese-containing superoxide dismutase. Oncogene 10: 1989–2000
Lowe SW, Ruley HE, Jacks T and Housman DE (1993) p53-dependent apoptosis modulates the cytotoxicity of anticancer agents. Cell 74: 957–967
Marzo I, Brenner C, Zamzami N, Susin SA, Beutner G, Brdiczka D, Remy R, Xie Z-H, Reed JC and Kroemer G (1998) The permeability transition pore complex: a target for apoptosis regulation by caspases and bcl-2-related proteins. J Exp Med 187: 1261–1271
Miyashita T and Reed JC (1995) Tumor suppresser p53 is a direct transcriptional activator of the human bax gene. Cell 80: 293–299
Muschel RJ, Soto DE, McKenna WG and Bernhard EJ (1998) Radiosensitization and apoptosis. Oncogene 17: 3359–3363
Oberley LW and Buettner GR (1979) Role of superoxide in cancer: a review. Cancer Res 39: 1141–1149
Oberley TD and Oberley LW (1997) Antioxidant enzyme levels in cancer. Histol Histopathol 12: 525–535
Oberley LW and Spitz DR (1985) Nitroblue tetrazolium. In: CRC Handbook of Methods for Oxygen Radical Research, Greenwald RA (ed), pp 217–225. CRC Press Inc: Boca Raton, FL
Polyak K, Xia Y, Zweier JL, Kinzler KW and Vogelstein B (1997) A model for p53-induced apoptosis. Nature 389: 300–305
Shimizu S, Eguchi Y, Kamiike W, Waguri S, Uchiyama Y, Matsuda H and Tsujimoto Y (1996) Bcl-2 blocks loss of mitochondrial membrane potential while ICE inhibitors act at a different step during inhibition of death induced by respiratory chain inhibitors. Oncogene 13: 21–29
Sinha BK and Mimnaugh ED (1990) Free radicals and anticancer drug resistance: oxygen free radicals in the mechanisms of drug cytotoxicity and resistance by certain tumours. Free Radical Biol Med 8: 567–581
Susin SA, Zamzami N and Kroemer G (1998) Mitochondria as regulators of apoptosis: doubt no more. Biochim Biophys Acta 1366: 151–165
Susin SA, Lorenzo HK, Zamzami N, Marzo I, Snow BE, Brothers GM, Mangion J, Jacotot E, Costantini P, Loeffler M, Larochette N, Goodlett DR, Aebersold R, Siderovski DP, Penninger JM and Kroemer G 1999 a) Molecular characterization of mitochondrial apoptosis-inducing factor. Nature 397: 441–446
Susin SA, Lorenzo HK, Zamzami N, Marzo I, Brenner C, Larochette N, Provost M-C, Alzari PM and Kroemer G 1999 b) Mitochondrial release of caspase-2 and -9 during the apoptotic process. J Exp Med 189: 381–393
Tishler RB, Calderwood SK, Coleman CN and Price BD (1993) Increases in sequence specific DNA binding by p53 following treatment with chemotherapeutic and DNA damaging agents. Cancer Res 53: 2212–2216
Toh Y, Kuninaka S, Oshiro T, Ikeda Y, Nakashima H, Baba H, Kohnoe S, Okamura T, Mori M and Sugimachi K (2000) Overexpression of manganese superoxide dismutase mRNA may correlate with aggressiveness in gastric and colorectal adenocarcinomas. Int J Oncol 17: 107–112
Yoshikawa T, Kokura S, Tainaka K, Itani K, Oyamada H, Kaneko T, Naito Y and Kondo M (1993) The role of active oxygen species and lipid peroxidation in the antitumor effect of hyperthermia. Cancer Res 53: 2326–2329
Wallace DC (1999) Mitochondrial diseases in man and mouse. Science 283: 1482–1488
Wong GHW, Elwell JH, Oberley LW and Goeddel DV (1989) Manganous superoxide dismutase is essential for cellular resistance to cytotoxicity of tumor necrosis factor. Cell 58: 923–931
Zamzami N, Marchetti P, Castedo M, Zanin C, Vayssiere J-L, Petit PX and Kroemer G (1995) Reduction in mitochondrial potential constitutes an early irreversible step of programmed lymphocyte death in vivo. J Exp Med 181: 1661–1672
Zhong W, Oberley LW, Oberley TD, Yan T, Domann FE and St Clair DK (1996) Inhibition of cell growth and sensitization to oxidative damage by overexpression of manganese superoxide dismutase in rat glioma cells. Cell Growth Differ 7: 1175–1186
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Kuninaka, S., Ichinose, Y., Koja, K. et al. Suppression of manganese superoxide dismutase augments sensitivity to radiation, hyperthermia and doxorubicin in colon cancer cell lines by inducing apoptosis. Br J Cancer 83, 928–934 (2000). https://doi.org/10.1054/bjoc.2000.1367
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2000.1367
Keywords
This article is cited by
-
Impairment of electron transport chain and induction of apoptosis by chrysin nanoparticles targeting succinate-ubiquinone oxidoreductase in pancreatic and lung cancer cells
Genes & Nutrition (2023)
-
Ion transport mechanisms for smoke inhalation–injured airway epithelial barrier
Cell Biology and Toxicology (2020)