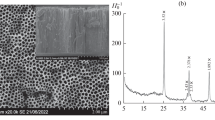
Abstract
We have fabricated and tested a photoelectrochemical (PEC) cell where the aqueous electrolyte has been replaced by a proton conducting hydrated Nafion® polymer membrane. The membrane was sandwiched between a TiO2-based photoanode and a Pt/C-based cathode. The performance was tested with two types of photoanode electrodes, a thermally prepared TiO2 film on Ti foil (T-TiO2) and a nanostructured TiO2 films in the form of highly ordered nanotubes (TNT) of different lengths. Firstly, photovoltammetry experiments were conducted under asymmetric conditions, where the anode was immersed in deionized water, while the cathode was kept in ambient air. The results showed a high incident photon-to-current efficiency (IPCE) of 19% under unassisted conditions (short-circuit, 0 V vs. cathode) with short TNT (ca. 1 μm) under 4 mW cm−2 illumination with UV-A rich light. Secondly, the deionized water was replaced by 0.5 M Na2SO4 and now the performance was higher with longer nanotubes, assigned to increased ionic conductivity inside the tubes. An unassisted (0 V) IPCE of 33% was achieved with nanotubes of ca. 8 μm. The presented solid-state PEC cell minimizes the electrode distance and volume of the device, and provides a way towards compact practical applications in solar water splitting.
Article PDF
Similar content being viewed by others
References
A. Fujishima and K. Honda, Electrochemical Photolysis of Water at a Semiconductor Electrode, Nature, 1972, 238(5358), 37–38.
R. van de Krol and M. Grätzel, Photoelectrochemical Hydrogen Production, in Electronic Materials: Science & Technology, ed. H. L. Tuller, 2012, Springer, New York, Dordrecht, Heidelberg, London.
B. A. Pinaud, et al., Technical and economic feasibility of centralized facilities for solar hydrogen production via photocatalysis and photoelectrochemistry, Energy Environ. Sci., 2013, 6(7), 1983–2002.
S. Ichikawa and R. Doi, 1st World Conference Environmental Catalysis For a Better World and LifeHydrogen production from water and conversion of carbon dioxide to useful chemicals by room temperature photoelectrocatalysis, Catal. Today, 1996, 27(1), 271–277.
S. Ichikawa, Chemical conversion of carbon dioxide by catalytic hydrogenation and room temperature photoelectrocatalysis, Energy Convers. Manage., 1995, 36(6-9), 613–616.
J. Georgieva, et al., An all-solid photoelectrochemical cell for the photooxidation of organic vapours under ultraviolet and visible light illumination, Electrochem. Commun., 2009, 11(8), 1643–1646.
B. Seger and P. V. Kamat, Fuel Cell Geared in Reverse: Photocatalytic Hydrogen Production Using a TiO2/Nafion/Pt Membrane Assembly with No Applied Bias, J. Phys. Chem. C, 2009, 113(43), 18946–18952.
Y. Li, et al., A novel photoelectrochemical cell with selforganized TiO2 nanotubes as photoanodes for hydrogen generation, Int. J. Hydrogen Energy, 2011, 36(22), 14374–14380.
R. Marschall, et al., Composite proton-conducting polymer membranes for clean hydrogen production with solar light in a simple photoelectrochemical compartment cell, Int. J. Hydrogen Energy, 2012, 37(5), 4012–4017.
K.-T. Jeng, et al., Membrane electrode assembly-based photoelectrochemical cell for hydrogen generation, Int. J. Hydrogen Energy, 2010, 35(20), 10890–10897.
K. O. Iwu, et al., Effects of temperature, triazole and hot-pressing on the performance of TiO2 photoanode in a solid-state photoelectrochemical cell, Electrochim. Acta, 2014, 12, 66–74.
J. M. Spurgeon and N. S. Lewis, Proton exchange membrane electrolysis sustained by water vapor, Energy Environ. Sci., 2011, 4(8), 2993–2998.
G. C. Brunauer, et al., UV-Light-Driven Oxygen Pumping in a High-Temperature Solid Oxide Photoelectrochemical Cell, Adv. Funct. Mater., 2016, 26(1), 120–128.
S. P. Albu, et al., 250 pm long anodic TiO2 nanotubes with hexagonal self-ordering, Phys. Status Solidi RRL, 2007, 1(2), R65–R67.
V. Zwilling, M. Aucouturier and E. Darque-Ceretti, Anodic oxidation of titanium and TA6 V alloy in chromic media. An electrochemical approach, Electrochim. Acta, 1999, 45(6), 921–929.
C. A. Grimes and G. K. Mor, TiO2Nanotube Arrays: Synthesis, Properties and Applications, Springer Science + Business Media, New York, 2009.
K. Zhu, et al., Removing Structural Disorder from Oriented TiO2 Nanotube Arrays: Reducing the Dimensionality of Transport and Recombination in Dye-Sensitized Solar Cells, Nano Lett., 2007, 7(12), 3739–3746.
D. Kowalski, D. Kim and P. Schmuki, TiO2 nanotubes, nanochannels and mesosponge: Self-organized formation and applications, Nano Today, 2013, 8(3), 235–264.
F. Fabregat-Santiago, et al., High Carrier Density and Capacitance in TiO2 Nanotube Arrays Induced by Electrochemical Doping, J. Am. Chem. Soc., 2008, 130(34), 11312–11316.
G. K. Mor, et al., A review on highly ordered, vertically oriented TiO2 nanotube arrays: Fabrication, material properties, and solar energy applications, Sol. Energy Mater. Sol. Cells, 2006, 90(14), 2011–2075.
J. M. Macak, et al., Smooth Anodic TiO2 Nanotubes, Angew. Chem., Int. Ed., 2005, 44(45), 7463–7465.
K. O. Iwu, et al., Solid-state photoelectrochemical H2 generation with gaseous reactants, Electrochim. Acta, 2013, 12, 320–325.
A. K. Seferlis and S. G. Neophytides, On the kinetics of photoelectrocatalytic water splitting on nanocrystalline TiO2 films, Appl. Catal., B, 2013, 132-133, 543–552.
R. Beranek, (Photo)electrochemical Methods for the Determination of the Band Edge Positions of TiO2-Based Nanomaterials, Adv. Phys. Chem., 2011, 12, 20.
Y. Ma, et al., Titanium Dioxide-Based Nanomaterials for Photocatalytic Fuel Generations, Chem. Rev., 2014, 114(19), 9987–10043.
W. W. Gärtner, Depletion-Layer Photoeffects in Semiconductors, Phys. Rev., 1959, 116(1), 84–87.
L. V. Taveira, et al., Impedance Behavior of TiO2 Nanotubes Formed by Anodization in NaF Electrolytes, J. Electrochem. Soc., 2008, 155(6), C293–C302.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 3.0 License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/3.0/.
About this article
Cite this article
Xu, K., Chatzitakis, A. & Norby, T. Solid-state photoelectrochemical cell with TiO2 nanotubes for water splitting. Photochem Photobiol Sci 16, 10–16 (2017). https://doi.org/10.1039/c6pp00217j
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c6pp00217j