Abstract
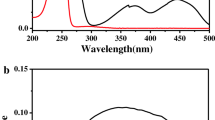
The photooxygenation of model compounds containing the two enantiomers of naproxen (NPX) covalently linked to histidine (His), tryptophan (Trp) and tyrosine (Tyr) has been investigated by steady state irradiation, fluorescence spectroscopy and laser flash photolysis. The NPX–His systems presented the highest oxygen-mediated photoreactivity. Their fluorescence spectra matched that of isolated NPX and showed a clear quenching by oxygen, leading to a diminished production of the NPX triplet excited state (3NPX*–His). Analysis of the NPX–His and NPX–Trp photolysates by UPLC-MS–MS revealed in both cases the formation of two photoproducts, arising from the reaction of singlet oxygen (1O2) with the amino acid moiety. The most remarkable feature of NPX–Trp systems was a fast and stereoselective intramolecular fluorescence quenching, which prevented the efficient formation of3NPX*–Trp, thus explaining their lower reactivity towards photooxygenation. Finally, the NPX–Tyr systems were nearly unreactive and exhibited photophysical properties essentially coincident with those of the parent NPX. Overall, these results point to a type II photooxygenation mechanism, triggered by generation of1O2 from the3NPX* chromophore.
Similar content being viewed by others
Notes and references
L. I. Grossweiner and K. C. Smith, Photochemistry, in The Science of Photobiology, ed. K. C. Smith, Plenum Press, New York, 2nd edn, 1989, pp. 47–78.
L. Pretali and A. Albini, in CRC Handbook of Organic Photochemistry and Photobiology, ed. A. Griesbeck, M. Oelgemö ller and F. Ghetti, CRC Press, Boca Raton, FL, 3rd edn, 2012, pp. 369–391.
C. S. Foote, Definition of type I and type II photosensitized oxidation, Photochem. Photobiol. 1991, 54, 659.
K. Briviba, L. O. Klotz and H. Sies, Toxic and signaling effects of photochemically generated singlet oxygen in biological systems, Biol. Chem. 1997, 378, 1259–1265.
M. J. Davies, Singlet oxygen-mediated damage to proteins and its consequences, Biochem. Biophys. Res. Commun. 2003, 305, 761–770.
M. J. Davies, Reactive species formed on proteins exposed to singlet oxygen, Photochem. Photobiol. Sci. 2004, 3, 17–25.
A. W. Girotti, Photosensitized oxidation of membrane lipids: reaction pathways, cytotoxic effects, and cytoprotective mechanisms, J. Photochem. Photobiol., B 2001, 63, 103–113.
I. Andreu, I. M. Morera, F. Boscá, L. Sánchez, P. Camps and M. A. Miranda, Cholesterol-diaryl ketone stereoisomeric dyads as models for clean type I and type II photooxygenation mechanisms, Org. Biomol. Chem. 2008, 6, 860–867.
E. R. Stadtman, Oxidation of free amino acids and amino acid residues in proteins by radiolysis and by metal-catalysed reactions, Annu. Rev. Biochem. 1993, 62, 797–821.
W. M. Garrison, Reaction mechanisms in the radiolysis of peptides, polypeptides and proteins, Chem. Rev. 1987, 87, 381–398.
P. U. Giacomoni, Sun Protection in Man, Comprehensive Series in Photosciences, Elsevier, Amsterdam, 2001, vol.3,.
R. C. Straight and J. D. Spikes, Photosensitized oxidation of biomolecules. in Polymers and Biopolymers, ed. A. A. Frimer and O. Singlet, CRC Press, Boca Raton, FL, 1985, pp. 91–143.
A. Wright, W. A. Bubb, C. L. Hawkins and M. J. Davies, Singlet oxygen mediated protein oxidation: evidence for the formation of reactive side-chain peroxides on tyrosine residues, Photochem. Photobiol. 2002, 76, 35–46.
V. V. Agon, W. A. Bubb, A. Wright, C. L. Hawkins and M. J. Davies, Sensitizer-mediated photooxidation of histidine residues: evidence for the formation of reactive side-chain peroxides, Free Radical Biol. Med. 2006, 40, 698–710.
J. E. Huyett, P. E. Doan, R. Gurbiel, A. L. P. Houseman, M. Sivaraja, D. B. Goodin and B. M. Hoffman, Compound ES of cytochrome c peroxidase contains a Trp π-cation radical: characterization by CW and pulsed Q-band ENDOR spectroscopy, J. Am. Chem. Soc. 1995, 117, 9033–9041.
R. W. Redmond and J. N. Gamlin, A compilation of singlet oxygen yields from biologically relevant molecules, Photochem. Photobiol. 1999, 70, 391–475.
A. J. Lewis and D. E. Furst, Nonsteroidal Anti-Inflammatory Drugs: Mechanisms and Clinical Uses, Marcel Dekker, New York, 2nd edn, 1994.
F. Boscá, M. L. Marín and M. A. Miranda, Photoreactivity of the nonsteroidal anti-inflammatory 2-arylpropionic acids with photosensitizing side effects, Photochem. Photobiol. 2001, 74, 637–655.
G. M. J. Beijersbergen van Henegouwen, Phototoxicity of drugs and other xenobiotics, J. Photochem. Photobiol., B 1991, 10, 183–210.
M. A. Miranda, J. V. Castell, D. Hernández, M. J. Gómez-Lechón, F. Boscá, I. M. Morera and Z. Sarabia, Drug-photosensitized protein modifications: identification of the reactive sites and elucidation of the reaction mechanisms with tiaprofenic acid/albumin as model system, Chem. Res. Toxicol. 1998, 11, 172–177.
M. C. Jiménez, U. Pischel and M. A. Miranda, Photoinduced processes in naproxen-based chiral dyads, J. Photochem. Photobiol., C 2007, 8, 128–142.
A. Catalfo, G. Bracchitta, G. de Guidi, Role of aromatic amino acid tryptophan UVA-photoproducts in the determination of drug photosensitization mechanism: a comparison between methylene blue and naproxen, Photochem. Photobiol. Sci. 2009, 8, 1467–1475.
I. Vayá, R. Pérez-Ruiz, V. Lhiaubet-Vallet, M. C. Jiménez and M. A. Miranda, Drug-protein interactions assessed by fluorescence measurements in the real complexes and in model dyads, Chem. Phys. Lett. 2010, 486, 147–153.
I. Vayá, M. C. Jiménez and M. A. Miranda, Excited state interactions in flurbiprofen-tryptophan dyads, J. Phys. Chem. B 2007, 111, 9363–9371.
I. Vayá, P. Bonancía, M. C. Jiménez, D. Markovitsi, T. Gustavsson and M. A. Miranda, Excited state interactions between flurbiprofen and tryptophan in drug-protein complexes and in model dyads. Fluorescence studies from the femtosecond to the nanosecond time domains, Phys. Chem. Chem. Phys. 2013, 15, 4727–4734.
A. G. Griesbeck, J. Neudörfl, A. de Kiff, Photoinduced electron-transfer chemistry of the bielectrophoric N-phthaloyl derivatives of the amino acids tyrosine, histidine and tryptophan, Beilstein J. Org. Chem. 2011, 7, 518–524.
B. Giese, M. Wang, J. Gao, M. Stoltz, P. Müller and M. Graber, Electron relay race in peptides, J. Org. Chem. 2009, 74, 3621–3625.
M. Cordes, A. Köttgen, C. Jasper, O. Jacques, H. Boudebous and B. Giese, Influence of amino acid side chains on long-distance electron transfer in peptides: electron hopping via “stepping stones”, Angew. Chem., Int. Ed. 2008, 47, 3461–3463.
B. Abraham and L. A. Kelly, Photooxidation of amino acids and proteins mediated by novel 1,8-naphthalimide derivatives, J. Phys. Chem. B 2003, 107, 12534–12541.
E. Cadenas, Biochemistry of oxygen toxicity, Annu. Rev. Biochem. 1989, 58, 79–110.
D. de la Peña, C. Martí, S. Nonell, L. A. Martínez and M. A. Miranda, Time-resolved near infrared studies on singlet oxygen production by the photosensitizing 2-arylpropionic acids, Photochem. Photobiol. 1997, 65, 828–832.
B. A. Kerwin and R. L. Remmele, Protect from light: photodegradation and protein biologics, J. Pharm. Sci. 2007, 96, 1468–1479.
P. Kang and C. S. Foote, Synthesis of a C-13, N-15 labeled imidazole and characterization of the 2,5-endoperoxide and its decomposition, Tetrahedron Lett. 2000, 41, 9623–9626.
I. Saito, T. Matsuura, M. Nakagawa and T. Hino, Peroxidic intermediates in photosensitized oxygenation of tryptophan derivatives, Acc. Chem. Res. 1977, 10, 346–352.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Dedicated to the memory of Prof. Nicholas J. Turro.
Electronic supplementary information (ESI) available: Additional X-ray structures, photodegradation kinetics, UV, fluorescence and transient absorption data (8 pages). CCDC 662964, 662965, 662957, 662958, 662959 and 662960. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c3pp50252j
Rights and permissions
About this article
Cite this article
Vayá, I., Andreu, I., Jiménez, M.C. et al. Photooxygenation mechanisms in naproxen–amino acid linked systems. Photochem Photobiol Sci 13, 224–230 (2014). https://doi.org/10.1039/c3pp50252j
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c3pp50252j