Abstract
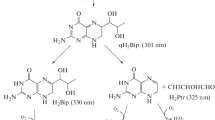
7,8-Dihydrobiopterin (H2Bip) and 7,8-dihydroneopterin (H2Nep) belong to a class of heterocyclic compounds present in a wide range of living systems. H2Bip accumulates in the skin of patients suffering from vitiligo, whereas H2Nep is secreted by human macrophages when the cellular immune system is activated. We have investigated the photochemical reactivity of both compounds upon UV-A irradiation (320-400 nm), the chemical structures of the products and their thermal stability. The study was performed in neutral aqueous solutions. The reactions were followed by UV/Visible spectrophotometry and HPLC and the products were analyzed by means of electrospray ionization mass spectrometry and 1H-NMR. Excitation of H2Bip and H2Nep leads to the formation, in each case, of two main isomeric dimers. The latter compounds undergo a thermal process that may consist in a retro [2 + 2]-cycloaddition and hydrolysis to yield the reactant (H2Bip or H2Nep) and a product that has incorporated a molecule of H2O.
Similar content being viewed by others
References
D. J. Brown, The Chemistry of Heterocyclic Compounds, John Wiley & Sons, New York, 1988, pp. 1–42.
W. Pfleiderer, Chemistry and Biology of Pteridines and Folates, Plenum Press, New York, 1993, pp. 1–16.
T. J. Kappock and J. P. Caradonna, Pterin-dependent amino acid hydroxylases Chem. Rev. 1996 96 2659–2756.
E. R. Werner, G. Werner-Felmayr, D. Fuchs, A. Hausen, G. Reibnegger, J. J. Yim, W. Pfleiderer and H. Wachter, Tetrahydrobiopterin biosynthetic activities in human macrophages, fibroblasts, THP-1 and T24 cells. GTP-cyclohydrolase I is stimulated by interferon gamma, and 6-pyrovyl tetrahydropterin synthase and sepiapterinreductase are constitutively present J. Biol. Chem. 1990 265 3189–3192.
C. Huber, J. R. Batchelor, D. Fuchs, A. Hausen, A. Lang, D. Niederwieser, G. Reibnegger, P. Swetly, J. Troppmair and H. Wachter, Immune response-associated production of neopterin. Release from macrophages primarily under control of interferon-gamma J. Exp. Med. 1984 160 310–316.
C. Huber, D. Fuchs, A. Hausen, R. Margreiter, G. Reibnegger, M. Spielberger and H. Wachter, Pteridines as a new marker to detect human T cells activated by allogeneic or modified self major histocompatibility complex (MHC) determinants J. Immunol. 1983 130 1047–1050.
D. Fuchs, G. Weiss and H. Wachter, The role of neopterin as a monitor of cellular immune activation in transplantation, inflammatory, infectious, and malignant diseases Crit. Rev. Clin. Lab. Sci. 1992 29 307–341.
D. Fuchs, G. Weiss and H. Wachter, Neopterin, biochemistry and clinical use as a marker for cellular immune reactions Int. Arch. Allergy Immunol. 1993 101 1–6.
D. Fuchs, A. Hausen, G. Reibnegger, E. R. Werner, M. P. Dierich and H. Wachter, Neopterin as marker for activated cell mediated immunity Immunol. Today 1988 9 150–155.
C. A. Nichol, G. K. Smith and D. S. Duch, Biosynthesis and metabolism of tetrahydrobiopterin and molybdopterin Annu. Rev. Biochem. 1985 54 729–764.
K. U. Schallreuter, G. Büttner, M. R. Pittelkow, J. M. Wood, N. N. Swanson and C. Korner, Regulation of melanin biosynthesis in the human epidermis by tetrahydrobiopterin Biochem. Biophys. Res. Commun. 1994 204 43–48.
K. U. Schallreuter, J. Moore, J. M. Wood, W. D. Beazley, E. M. Peters, L. K. Marles, S. C. Behrens-Williams, R. Dummer, N. Blau, B. Thöny, Epidermal H2O2 accumulation alters tetrahydrobiopterin (6BH4) recycling in vitiligo: Identification of a general mechanism in regulation of all 6BH4-dependent processes? J. Invest. Dermatol. 2001 116 167–174.
K. U. Schallreuter, J. M. Wood, M. R. Pittelkow, M. Gütlich, K. R. Lemke, W. Rödl, N. N. Swanson, K. Hitzemann and I. Ziegler, Regulation of melanin biosynthesis in the human epidermis by tetrahydrobiopterin Science 1994 263 1444–1446.
H. Rokos, W. D. Beazley and K. U. Schallreuter, Oxidative stress in vitiligo: Photo-oxidation of pterins produces H2O2 and pterin-6-carboxylic acid Biochem. Biophys. Res. Commun. 2002 292 805–811.
A. H. Thomas, C. Lorente, A. L. Capparelli, C. G. Martínez, A. M. Braun and E. Oliveros, Singlet oxygen (1?g) production by pterin derivatives in aqueous solutions Photochem. Photobiol. Sci. 2003 2 245–250.
M. L. Dántola, M. Vignoni, C. González, C. Lorente, P. Vicendo, E. Oliveros and A. H. Thomas, Electron transfer processes induced by the triplet state of pterins in aqueous solutions Free Radical Biol. Med. 2010 49 1014–1022.
M. Vignoni, F. M. Cabrerizo, C. Lorente and A. H. Thomas, New Results on the Photochemistry of Biopterin and Neopterin in Aqueous Solution Photochem. Photobiol. 2009 85 365–373.
K. Ito and S. Kawanishi, Photoinduced hydroxylation of deoxyguanosine in DNA by pterins: Sequence specificity and mechanism Biochemistry 1997 36 1774–1781.
G. Petroselli, M. L. Dántola, F. M. Cabrerizo, A. L. Capparelli, C. Lorente, E. Oliveros and A. H. Thomas, Oxidation of 2’-Deoxyguanosine 5’-Monophosphate Photoinduced by Pterin: Type I versus Type II Mechanism J. Am. Chem. Soc. 2008 130 3001–3011.
M. Vignoni, F. M. Cabrerizo, C. Lorente, C. Claparols, E. Oliveros and A. H. Thomas, Photochemistry of dihydrobiopterin in aqueous solution Org. Biomol. Chem. 2010 8 800–810.
M. Vignoni, M. P. Serrano, E. Oliveros and A. H. Thomas, Photodimerization of 7,8-dihydroneopterin in aqueous solution under UV-A irradiation Photochem. Photobiol. 2011 87 51–55.
Y. Wang, Y. Li, Y. Wu and H. Yan, Mechanism of dihydroneopterin aldolase NMR, equilibrium and transient kinetic studies of the Staphylococcus aureus and Escherichia coli enzymes FEBS J. 2007 274 2240–2252.
M. L. Dántola, A. H. Thomas, E. Oliveros and C. Lorente, Visible-light photochemistry of 6-formyl-7,8-dihydropterin in aqueous solution J. Photochem. Photobiol., A 2010 209 104–110.
M. Vignoni, M. L. Salum, R. Erra-Balsells, A. H. Thomas and F. M. Cabrerizo, 1H NMR characterization of the intermediate formed upon UV-A excitation of biopterin, neopterin and 6-hydroxymethylpterin in O2-free aqueous solutions Chem. Phys. Lett. 2010 484 330–332.
B. H. Jennings, S. C. Pastra and J. L. Wellington, Photosensitized dimerization of thymine Photochem. Photobiol. 1970 11 215–226.
B. H. Jennings, S. Pastra-Landis and J. W. Lerman, Photosensitized dimerization of uracil Photochem. Photobiol. 1972 15 479–491.
M Poe and K. Hoogsteen, 5,6,7,8-tetrahydrofolic acid. Conformation of the tetrahydropyrazine ring J. Biol. Chem. 1978 253 543–546.
A. R. Katritzky, N. G. Akhmedov, E. M. Myshakin, A. K. Verma and C. D. Hall, Low-temperature 1H and 13C NMR Spectra of N-Substituted 1,2,3,4-Tetrahydropyrazino[1,2-a]indoles Magn. Reson. Chem. 2005 43 351–358.
T. L. Gilchrist, Heterocyclic Chemistry, 3rd edn, Prentice Hall, 1997.
Author information
Authors and Affiliations
Corresponding authors
Additional information
This article is published as part of a themed issue in honour of Professor Kurt Schaffner on the occasion of his 80th birthday.
Electronic supplementary information (ESI) available. See DOI: 10.1039/c2pp05363b
Rights and permissions
About this article
Cite this article
Vignoni, M., Lorente, C., Cabrerizo, F.M. et al. Characterization and reactivity of photodimers of dihydroneopterin and dihydrobiopterin. Photochem Photobiol Sci 11, 979–987 (2012). https://doi.org/10.1039/c2pp05363b
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c2pp05363b