Abstract
Proteins are carriers of biological functions and the effects of atmospheric-pressurenon-thermal plasmas on proteins are important to applications such as sterilizationand plasma-induced apoptosis of cancer cells. Herein, we report our detailedinvestigation of the effects of helium-oxygen non-thermal dielectric barrierdischarge (DBD) plasmas on the inactivation of lactate dehydrogenase (LDH) enzymesolutions. Circular dichroism (CD) and dynamic light scattering (DLS) indicate thatthe loss of activity stems from plasma-induced modification of the secondarymolecular structure as well as polymerization of the peptide chains. Raising thetreatment intensity leads to a reduced alpha-helix content, increase in thepercentage of the beta-sheet regions and random sequence, as well as graduallydecreasing LDH activity. However, the structure of the LDH plasma-treated for 300seconds exhibits a recovery trend after storage for 24 h and itsactivity also increases slightly. By comparing direct and indirect plasmatreatments, plasma-induced LDH inactivation can be attributed to reactive species(RS) in the plasma, especially ones with a long lifetime including hydrogenperoxide, ozone and nitrate ion which play the major role in the alteration of themacromolecular structure and molecular diameter in lieu of heat, UV radiation, andcharged particles.
Similar content being viewed by others
Introduction
Owing to advantages such as production of highly reactive species at low temperature andflexible operation1,2, atmospheric-pressure non-thermal plasmas haveattracted much attention in biology and biomedicine3 and applicationsinclude plasma sterilization4,5,6,7,8,9,10,11, living tissuetreatment12, blood coagulation13, cell detachment14,15, induction of apoptosis16,17,18, cellproliferation19, cancer therapy20,21,22,23,24, andso on. However, even though the biological effects of atmospheric-pressure plasmas havebeen investigated and several possible mechanisms have been suggested, systematicverification of these hypotheses is still lacking and the precise mechanism is still notwell understood. To gain further insight, it is necessary to study not only thebiological effects of cells and tissues, but also their interaction with cold plasmas onthe molecular level. Proteins are the main vehicles of biological functions and accountfor 68% of the dry weight of cells and tissues. There have been investigations on theuse of atmospheric-pressure plasmas to modify the secondary structure of proteins inaqueous solutions and inactivate infectious prion proteins under dry conditions. Forexample, discharge plasmas inactivate and induce Heme degradation of horseradishperoxidase in the phosphate buffer (PBS) solution25, inactivates lysozymein an aqueous solution26, activates lipase in the PBS solution27 and inactivates polyphenoloxidase (PPO) and peroxidase (POD) in a modelfood system28. However, in spite of recent progress, the molecularmechanism between plasmas and enzymatic activity is still unclear. In this workdescribed in this paper, a dielectric barrier discharge (DBD) plasma is used to treatlactate dehydrogenase (LDH), an important sugar metabolic enzyme29,30and the mechanism of protein inactivation and effects on cell metabolism areinvestigated.
Results and discussion
Emission spectrometry and mass spectrometry of DBD plasmas
The typical optical spectrum of the helium-oxygen DBD plasma (Fig.1) between 200 and 900 nm is displayed in Fig. 2. The dominant emission lines illustrate the presence of themetastable helium atom He (728.1 nm, 706.5 nm,667.8 nm, 587.5 nm, 501.5 nm,447.1 nm and 388.8 nm), OH radical(306-310 nm) and atomic oxygen (OI: 799.5 nm,777.2 nm, 715.7 nm; OII: 656.5 nm, and578.4 nm). In addition, the detected reactive species associatedwith nitrogen are excited nitrogen molecules between 300 and400 nm.
Figures 3(a) depict the time-averaged mass spectra ofpositive and negative ions obtained at a distance of 5 mm from thebottom of the quartz glass DBD plasma to the orifice of the mass spectrometer.Ions up to 100 amu are detected. The positive mass spectrum in Fig. 3a shows about there are 10 predominant species in thehelium-oxygen plasma, namely O2+,H3O+,H3O+(H2O), N2+,O+, NO+, as well as small portions ofH2O+, N2H+,NO2+. In the negative spectrum in Fig. 3(b), more than 10 species are detected and the main speciesare O−, OH−,H3O−,O2−,(OH)O−,OH−(H2O),NO2−,O3−, andNO3−.
The DBD plasma produces a variety of ions and free radicals in the gas phase.These active ions and free radicals react with water and produce variousbiologically active reactive species (RS) in the liquid phase such as ones witha long lifetime including hydrogen peroxide (H2O2), ozone(O3) and nitrate ion(NO3−) as well as short-lived RS includinghydroxyl radical (OH−), superoxide(O2−) and singlet oxygen31. Ozone is produced in the DBD plasma by the interaction betweenatomic oxygen and oxygen in the gas phase and then spreads into the liquidphase. Oxygen reduction by two electrons forms hydrogen peroxide, while thenitrate ion is produced by the reaction between nitrogen dioxide and water. Inthis study, some known long-lived species in the liquid are measured and thepossible production mechanism is shown in Table 1. RS ishighly reactive which can mediate the bio-macromolecules via the cellmembrane or intercellularly. By comparing the direct treatment of LDH withindirect treatment, the effects of the short-lived RS in the liquid phase can beexplored.
Concentration of RS
The sample solutions were treated by the non-thermal DBD plasma. Since the LDHenzyme is dissolved in PBS, the RS in PBS created by the plasma are expected toaffect the LDH enzyme. Figure 4 shows the RSconcentrations including those of ozone, hydrogen peroxide and nitrate ion fordifferent treatment time. The RS concentrations increase with the plasmaexposure time. The hydrogen peroxide, nitrate ion and ozone concentrationsincrease from 0.077 mg/L to 158.5 mg/L,0.098 mg/L to 25.5 mg/L and 0.34 mg/L to3.6 mg/L, respectively, after 300 s.
The RS concentrations after storage (0 to 24 h) are monitored asshown in Fig. 5 which shows two different trends for thethree kinds of long-lived RS. For ozone and hydrogen peroxide, theconcentrations decrease within 24 h. The concentration of ozonedrops from 3.6 mg/L to 2.9 mg/L whereas that of hydrogenperoxide diminishes from 158 mg/L to 103.5 mg/L.However, the nitrate ion content rises from 25.5 mg/L to27.1 mg/L in the first 3 hours possibly due to oxidationof nitrite ions and then stabilizes in the remaining time.
Inactivation of LDH by plasma treatment
The LDH protease solution is exposed to the helium-oxygen non-thermal dielectricbarrier discharge plasma directly and indirectly and the absolute LDH activityis assessed after treatment for one hour. As shown in Fig.6, the LDH activity decreases steadily with exposure time regardlessof treatment modes revealing that inactivation of the LDH activity increaseswith time. Compared to the direct treatment, reduction of the LDH activity isless in the indirect treatment for the same time. However, as reportedpreviously on lipase treated by a plasma jet27,32, there is nodifference in the enzymatic activity between the direct and indirect treatment.This may be due to the special physical and chemical properties of proteins anddifference in the sensitivity of different proteins for different plasma. In ourstudy, the difference in LDH enzymatic activity between the direct treatment andindirect treatment can be attributed to the difference in the RS in the twomodes. Compared to the indirect treatment, besides the effects of the long-livedRS (hydrogen peroxide, ozone and nitrate ions in the PBS solution), there areshort-lived RS such as OH˙,O2− and O2(1Δg) as well as UV in the direct treatment31,33,34,35,36. Hence, the RS may be responsible for theslightly larger effects in the direct treatment.
To investigate the continuous effects of the LDH activity after plasma treatment,the LDH is treated for 300 s in both the direct and indirect modes.The treated LDH samples are stored at 4 °C and theactivity is monitored at different time points. The change in the enzymeactivity with storage time is shown in Fig. 7. The enzymeactivity declines quickly in the first three hours and then gradually from threeto twelve hours. The results indicate that there are continuous effects on theLDH activity regardless of treatment modes attributable to the long-lived RS(hydrogen peroxide, ozone and nitrate ion) in the PBS created by the DBDplasma. Hydrogen peroxide can oxidize proteins effectively causing the proteinto denature as follows37:

In addition, many amino acids can be modified by ozone38, forexample,

With regard to nitrate ions, conformational change or protein denaturation resultfrom changing the electrical charges39,40. In comparison withthe indirect treatment, the decrease in the LDH activity is smaller in thedirect treatment, suggesting that the short-lived RS (OH˙,O2− and O2(1Δg)) and UV produce only minor effects on theLDH activity. Therefore, the possible mechanism of the continuous effect onprotein by DBD plasma is due to hydrogen peroxide41,42, ozone,and nitrate ions which cause chemical modification of the LDH molecule orbetween the molecules25,43,44. More experiments are needed toclarify the synergistic effects. As shown in Fig. 7, theLDH enzyme activity exhibits an unexpected increase after storage for24 hours. This phenomenon indicates some“re-naturing” of the LDH protease after the DBD plasmatreatment after a while. A possible reason is that after a long time, theinactivation effect is reversible or the functional domains that are hidden inthe interior of the hydrophilic amino acids induced by the DBD plasma areexposed again. This topic will be discussed further later.
CD analysis
Figures 8(a) depict the CD spectra of the LDH after directand indirect treatment for 0-300 s and one hour after the plasmatreatment. The spectra show the typical negative peak at 208 nmcorresponding to the percentage of the α-helical structure in theenzyme and a shoulder at 218 nm indicating the percentage of theβ-sheet structure. Flattening of these two regions with increasingplasma exposure times indicates reduction in the helical structure and largerβ-sheet content. In addition, there is a positive peak at230 nm and the negative peak represents the turn at200 nm. The changes illustrate that the secondary structure of theproteins change with treatment time depending on the concentration of RS in thePBS created by the plasma.
The exact percentages of the different structures are calculated from thecorrected spectra. The ordered secondary structure of LDH mostly consists ofα-helices and turns and the relative content of theβ-sheet structure is smaller. As shown in Fig.8(c), the amounts of turns and α-helix structure decreasedrastically with treatment time while the number of random areas andparticularly amount of β-sheet structures increase strikingly,indicating that the secondary structure of the LDH enzyme is altered by theplasma. In the LDH solution after the direct treatment, the α-helixcontent decreases from 33.20 to 20.60%, whereas the β-sheet contentincreases from 12.30 to 22.70% after treatment for 300 s. In theindirect treatment, the α-helix content decreases from 33.10 to22.60% and the β-sheet content increases from 12.30 to 21.70%. Theloss in the α-helix content and increase in the β-sheetcontent are similar in both treatment modes, indicating that the long-lived RSin the PBS may play primary roles in the structural changes in the LDHenzyme35,45,46.
In a complex plasma, many reactions can cause conformational and secondarystructural changes, but all of them are likely initiated by the inherent RS36. These RS are able to cleave peptide bonds and modify amino acidchains thereby producing secondary structure changes. The sulfur-containingamino acids such as cysteine and aromatic amino acids are particularlysusceptible. Concerning LDH, the active sites are more flexible compared withthe other structure and these amino acids such as Arg-171, His-195, Arg-109 andAsp-168 play a key role in the active sites47 and all areinvolved in the formation of the α-helical structure. Therefore,there is a strong correlation between the enzyme activity andα-helical structure. Oxidation of even one single amino acid in theprotein can affect its function. For example, oxidation of histidine-195 at theactive side of the LDH can result in loss of activity because the active sitescannot combine with the substrate.
Figures 9(a) show the CD spectra after storing at4 °C for different time after plasma treatment for300 s directly and indirectly, respectively. The DBD plasma hascontinuous effects on the secondary structure of LDH protease but the effectsare attenuated after storage and as shown in Fig. 9(c),after storage for 24 h, the second structures exhibit recovery whichcoincides with that of the enzyme activity further demonstrating that the changein enzyme activity is associated with the change of secondary structure inducedby DBD plasma48.
DLS analysis
The hydrodynamic radius and particle distribution of the LDH protease solutionare determined by means of dynamic light scattering by analyzing the combinedbehavior to better understand the effects of atmospheric-pressure plasma on LDH.The hydrodynamic radius of the LDH protease solution in the different treatmentmodes for different treatment time are shown in Figs. 10and 11, respectively. Figures10(a) and 11(a) show that the LDH proteasesolution without the treatment has a mass peak of the contribution ratio of100%, molecular radius of 4.9 nm and molecular weight of140 kDa. As shown in Figs. 10(b) and 11(b), the average hydrodynamic radius increases withtreatment time and the particle distribution also changes. The results indicatethat the DBD plasma can promote molecular aggregation between the LDH moleculesin this concentration range to generate larger and more complexsupramolecules49,50. A possible explanation involves themodification of amino acid, electrostatic interactions and hydrophobicinteractions. Ozone (O3)38 and particularly hydrogenperoxide (H2O2)37 are able to oxidize theamino acid side chains (cysteine) to form protein–proteincross-linkage51,52,53. In the direct treatment for180 s, the hydrodynamic radius and molecular weight increase to15.5 nm and 2056 kDa, respectively (Fig.10 (b)) and these values are larger than those after theindirect treatment (Fig.11 (b)). These differences may beattributed to the effects of UV radiation and short-lived reactive speciescreated by the plasma. Figures 10(c) and 11(c) show two new peaks after plasma treatment for300 s. A possible mechanism is that weak stability of the irregularsupramolecules which further polymerize with increasing treatment time. After ahigh degree of polymerization, the supramolecules disintegrate to two newpolymeric molecules spontaneously54,55.
The DBD plasma continuous effect on the hydrodynamic radius and particledistribution of LDH protease solution are assessed. As shown in Figs. 10 and 11, polymerization increases inthe initial 12 hours and the hydrodynamic radius of the newsupramolecules gradually increases (Figs. 12(a); Figs. 13(a)). Moreover, the DLS spectrum acquired after24 hours shows that there are new small polymers (Fig. 12(c) and 13(c)). A possible explanationis that the supramolecules disintegrate into two new polymeric moleculesspontaneously and this is the major reason for the aforementioned enzymerecovery.
Based on the above results, there is a strong correlation between the loss in theenzyme activity and change in the molecular structure depending on the RSproduced by DBD plasma. The molecular mechanism of LDH inactivation can bedescribed as follows. First, in the molecule, some important amino acids aremodified by the RS and the secondary structure changes, especially the structureof the active center and the active sites lose recognition and catalyticfunctions. Secondly, between the molecules, the peptide chains polymerize toform irregular supramolecules which wrap up the small quantity of active sitesand hence, the LDH is unable to participate in the catalytic reaction.Aggregation of molecules decreases the enzyme activity and also protects theactive sites from being modified by RS. When the supramolecules disintegratespontaneously after storing for a long time, the protected active sites areexposed again causing recovery as shown in Fig. 14. Allin all, the changes in the secondary structure and hydrodynamic radius coincidewith the loss and recovery of the enzyme activity. The results suggest that theenzymatic activity change arises from not only intramolecular chemicalmodification, but also intermolecular aggregation.
pH and temperature
The relationship between the temperature and pH of the LDH protease solution withtreatment time is shown in Fig. 15. The highesttemperature is 24 °C after plasma exposure for300 s and the pH remains at about 7.5 before and after the plasmatreatment because the PBS has the buffering capability. Since the temperatureand pH do not reach values that can cause enzyme inactivation, their effects canbe neglected.
Conclusion
A helium-oxygen non-thermal DBD plasma is employed to treat LDH as a model enzyme inPBS. The concentrations of the long-lived RS in the plasma-treated PBS, forinstance, hydrogen peroxide, nitrate ions and ozone, increase with treatment timebut decrease with storage time except nitrate. The LDH activity decreasessignificantly with plasma treatment time or storage time in the first12 h regardless of treatment modes, but recovers slightly after storingfor 24 h. The CD and DLS results suggest the mechanisms to explain thechange in the LDH activity. It is likely due to modification of the secondarystructure in the molecule and peptide chain polymerization between the molecules asa result of the reactive species created by the DBD plasma. By comparing the directand indirect plasma treatment, the changes in the LDH activity can be attributed tothe RS, especially long-lived ones such as hydrogen peroxide, ozone and nitrate ioninstead of heat, UV radiation and charged particles.
Materials and methods
Discharge Apparatus and Plasma treatment
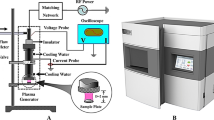
The atmospheric-pressure DBD plasma is depicted in Fig. 1.The DBD plasma reactors consist of a hollow plexiglass as a reactor chamber onwhich there are two air inlet and outlet holes. The high-voltage electrode is a32 mm diameter copper cylinder covered by 1 mm thickquartz glass as the insulating dielectric barrier and the ground electrode is a37 mm diameter copper cylinder. The discharge gap between the bottomof the quartz glass and sample surface is 5 mm. An alternatingcurrent power supply operating in frequencies between 10 and 42 kHzwith variable output voltages between 0 and 50 kV (peak to peak) isused.
Helium (99.99% pure) and oxygen (99.99% pure) were the carrier gases and the flowrates regulated by flow meters were 80 L/h and 10 L/h,respectively. In order to eliminate as much air from the reactor chamber aspossible, the working gas was bled into the chamber for 5 minutesbefore the experiment. The non-thermal DBD plasma was generated at voltage of14 kV (peak to peak) at a frequency of 24 kHz with adischarge power density of about 1 W/cm2. One ml ofthe LDH enzyme solutions in a 35 mm diameter petri dish was treatedby the DBD plasma and they were put on ice and stored in a cool bag in order toavoid unintentional inactivation after the treatment. In addition, because thedepth of plasma penetration was limited, neither charged particles nor reactivespecies generated in the plasma could interact directly with the LDH in thesolution. Hence, we investigated the LDH treated by direct and indirect plasmatreatment to demonstrate the inactivation effects of the reactive speciesinduced by the plasma. In the direct treatment, the plasma was used to treat theLDH enzyme solution whereas in the indirect treatment, the solution without LDHwas first exposed to the plasma followed by introduction of the protein to thetreated solution instantly.
Atomic emission spectrometry and molecular-beam massspectrometry
The optical emission spectra of the helium-oxygen non-thermal dielectric barrierdischarge (DBD) plasmas were acquired on the AvaSpec-2048-8-RM spectrometerequipped with gratings of 2,400 grooves/mm at a spectral rangebetween 200 and 900 nm. A molecular-beam mass spectrometer (MBMS,Hiden EQP mass/energy analyzer HPR 60) was operated in the time-averaged mode.The distance between the bottom of the quartz glass of the DBD plasma andorifice of the mass spectrometer was 5 mm.
Enzymes
LDH (from rabbit muscle, Type V, HM0037, sigma-Aldrich, Shanghai, China) in themitochondria of eukaryotic cells as tetramer can catalyze dehydrogenation oflactate to pyruvate which can take part in the Krebs cycle to provide energy tothe cells or organism. The samples were dissolved in phosphate buffer solution(PBS, pH = 7.5) to an initial concentrations of5 mg/ml and ultrasonically degassed. The enzyme solutions weredivided into small portions of 1 ml, put into 1.5 mlcentrifuge tubes and stored at −20 °C untilusage. These stock solutions were prepared once for the whole study.
Enzyme activity assays
The LDH activity was monitored using the double antibody sandwich method. Thepurified rabbit LDH antibody was used to coat the microtiter plate wells andmake the solid-phase antibody. The LDH antibody was mixed with horseradishperoxidase (HRP) to form the antibody-antigen-enzyme-antibody complex. Afterwashing and addition of tetramethylbenzidine (TMB), the solution turned blue dueto the HRP enzyme-catalyzed reaction. Sulfuric acid was added and the colorchange was monitored spectrophotometrically at 450 nm. Theconcentration of the rabbit LDH was determined by comparing the optical density(OD) with the standard curve. The absorbance was determined with respect to avalue on a microplate reader (Varioskan Flash, Thermo Scientific, Finland). Themeasurement was completed within 15 min after adding the solutionand the LDH liquid samples were diluted five times prior to the measurement. Theenzyme activity was expressed as the absolute one and the error represented thestandard deviation derived from at least six independent measurements.
Circular dichroism spectroscopy
The CD spectra were acquired in the UV range from 200 to 250 nm on aJasco-810-CD spectropolarimeter (Japan Spectroscopic Company, Tokyo, Japan) witha 500 μl sample quartz cup. The LDH enzyme solutionswere diluted from 5 mg/ml to 0.15 mg/ml before the CDspectra were obtained. The quartz sample cup was cleaned by doubly distilledwater and the secondary structure fractions were calculated from the spectrausing the CD analysis software. The absorbance was also measured at25 °C.
Dynamic light scattering (DLS)
The samples were injected into a sample cell (50 μl) inthe Dynapro-MS800 (ATC, England) instrument illuminated by a 25 mWand 750 nm solid-state laser at a fixed scattering angleθ = 90o equipped with adigital autocorrelator. The delay time was linearly spaced to sample the broaddistributions properly. The measurements were performed at25 °C and the data were then collected after temperaturestabilization for 10 to 15 min. Before the test, the samples werefiltered using inorganic membrane filters (whatman, Anatop10 Plus,0.22 μm), centrifuged at 10,000 rpm for10 minutes and degassed while attention was paid to make sure therewere no bubbles in the samples.
The particle size was related to the translational diffusivity (D) and theinstrument calculated the diffusivity (D) of the molecules in the sample basedon autocorrelation of the scattered light intensity. The hydrodynamic radius ofthe molecules was derived from D using a uniform sphere andStokes–Einstein equation,D = kT/6 πηr, wherek is Boltzmann’s constant, T is the absolute temperature,η is the viscosity of the liquid in which the particle is moving,and r is the hydrodynamic diameter56. This equation assumes thatthe particles move independently of each another. Finally, the DynaPro-MS800estimated the molecular weight according to the existing model of molecularradius / molecular weight and the distribution of particles in the solution.
Measurement of RS in PBS induced by plasma
When the plasma reacts with the liquid such as deionized water or PBS, RS areproduced but direct monitoring of RS in the liquid is difficult due to the shorthalf-life and high reactivity. Here, we only measured the concentration of thelong-lived RS such as hydrogen peroxide, nitrate and ozone in theplasma-treated liquid (PBS solution) spectrophotometrically on the PhotoLab6100(WTW, Germany). The test kits were 18789, 09713 and 00607 respectively andthe test methods were according to the WTW company website57.
pH and temperature
For the LDH protease solutions, the temperature was measured by an infraredthermometer (Raytek ST20 XB) and regular thermometer. The pH values weredetermined using a Basic PH meter (PB-10, Sartorius, Germany). The temperatureand pH of the enzyme solutions were measured simultaneously.
Additional Information
How to cite this article: Zhang, H. et al. Effects and Mechanism ofAtmospheric-Pressure Dielectric Barrier Discharge Cold Plasma on LactateDehydrogenase (LDH) Enzyme. Sci. Rep.5, 10031; doi: 10.1038/srep10031 (2015).
References
Mariotti, D. & Sankaran, R. M. Microplasmas for nanomaterials synthesis. J. Phys. D: Appl.Phys. 43, 323001, (2010).
Xian, Y. B. et al. From short pulses to short breaks: exotic plasma bullets via residual electron control.Sci. Rep. 3, 1599, (2013).
Kong, M. G. et al. Plasma medicine: an introductory review. New J.Phys. 11, 115012, (2009).
Moreau, M., Orange, N. & Feuilloley, M. G. J. Non-thermal plasma technologies: new tools for bio-decontamination. J. Biotechadv. 26, 610–617, (2008).
Moman, R. M. & Najmaldeen, H. The bactericidal efficacy of cold atmospheric plasma technology on some bacterial strains. Egypt. Acad. J. biolog. Sci. 2, 43–47 (2010).
Fricke, K. et al. Atmospheric pressure plasma: a high-performance tool for the efficient removal of biofilms. PLoS ONE 7, e42539, (2012).
Cheng, C. et al. Development of a new atmospheric pressure cold plasma jet generator and application in sterilization. Chin. Phys. 15, 1544–1548, (2006).
Cooper, M., Fridman, G., Fridman, A. & Joshi, S. G. Biological responses of Bacillus stratosphericus to Floating Electrode‐Dielectric Barrier Discharge Plasma Treatment. J. Appl. Microbiol. 109, 2039–2048, (2010).
Jie, S. et al. Observation of inactivation of Bacillus sbtilis spores under exposures of oxygen added argon atmospheric pressure plasma jet. Jpn. J. Appl. Phys. 53, 110310, (2014).
Jie, S. et al. Sterilization of Bacillus subtilis Spores Using an Atmospheric Plsama Jet with Argon and Oxygen Mixture Gas. Appl. Phys. Express 5, 036201, (2012).
Cheng, C. et al. Atmospheric pressure plasma jet utilizing Ar and Ar/H2O mixtures and applications to bacteria inactivation. Chin. Phys.B. 23, 075204, (2014).
Dobrynin, D., Fridman, G., Friedman, G., & Fridman, A. Physical and biological mechanisms of direct plasma interaction with living tissue. New J. Phys. 11, 115020, (2009).
Kuo, S. P. et al. Contribution of a portable air plasma torch to rapid blood coagulation as a method of preventing bleeding. New J. Phys. 11, 115016, (2009).
Hoentsch, M., von Woedtke, T., Weltmann, K. D. & Nebe, J. B. Time-dependent effects of low-temperature atmospheric-pressure argon plasma on epithelial cell attachment, viability and tight junction formation in vitro. J. Phys. D: Appl. Phys. 45, 025206, (2012).
Volotskova, O., Stepp, M. A. & Keidar, M. Integrin activation by a cold atmospheric plasma jet. New J.Phys. 14, 053019, (2012).
Tuhvatulin, A. I. et al. Non-thermal Plasma Causes p53-Dependent Apoptosis in Human Colon Carcinoma Cells. Acta Naturae 4, 82–87 (2012).
Panngom, K. et al. Preferential killing of human lung cancer cell lines with mitochondrial dysfunction by nonthermal dielectric barrier discharge plasma. Cell death & disease 4, e642, (2013).
Ahn, H. J. et al. Atmospheric-pressure plasma jet induces apoptosis involving mitochondria via generation of free radicals. PLoS ONE 6, e28154, (2011).
Kalghatgi, S. et al. Effects of non-thermal plasma on mammalian cells. PLoS ONE 6, e16270, (2011).
Partecke, L. I. et al. Tissue Tolerable Plasma (TTP) induces apoptosis in pancreatic cancer cells in vitro and in vivo. BMC cancer 12, 473, (2012).
Volotskova, O. et al. Targeting the cancer cell cycle by cold atmospheric plasma. Sci. Rep. 2, 636, (2012).
Joh, H. M. et al. Effect of additive oxygen gas on cellular response of lung cancer cells induced by atmospheric pressure helium plasma jet. Sci. Rep. 4, 6638, (2014).
Keidar, M. et al. Cold atmospheric plasma in cancer therapy a). Phys. Plasmas. 20, 057101, (2013).
Keidar, M. et al. Cold plasma selectivity and the possibility of a paradigm shift in cancer therapy. Br. J. Cancer 105, 1295–1301, (2011).
Ke, Z. & Huang, Q. Inactivation and Heme Degradation of Horseradish Peroxidase Induced by Discharge Plasma. Plasma Process. Polym. 10, 731–739, (2013).
Takai, E., Kitano, K., Kuwabara, J. & Shiraki, K. Protein Inactivation by Low-temperature Atmospheric Pressure Plasma in Aqueous Solution. Plasma Process. Polym. 9, 77–82, (2012).
Li, H. P. et al. Manipulation of Lipase Activity by the Helium Radio-Frequency, Atmospheric‐Pressure Glow Discharge Plasma Jet. Plasma Process. Polym. 8, 224–229, (2011).
Surowsky, B., Fischer, A., Schlueter, O. & Knorr, D. Cold plasma effects on enzyme activity in a model food system.Innov. Food Sci. Emerg. Technol. 19, 146–152, (2013).
Zewe, V. & Fromm, H. J. Kinetic studies of rabbit muscle lactate dehydrogenase. J.Biol. Chem. 237, 1668–1675 (1962).
Fritz, P. J. Rabbit muscle lactate dehydrogenase 5; a regulatory enzyme. Science 150, 364–366, (1965).
Priya Arjunan, K. & Morss Clyne, A. Hydroxyl Radical and Hydrogen Peroxide are Primarily Responsible for Dielectric Barrier Discharge Plasma‐Induced Angiogenesis. Plasma Process. Polym. 8, 1154–1164, (2011).
Li, G. et al. Genetic effects of radio-frequency, atmospheric-pressure glow discharges with helium. Appl. Phys. lett. 92, 221504, (2008).
Khoroshilova, E. V., Repeyev, Y. A. & Nikogosyan, D. N. UV protolysis of aromatic amino acids and related dipeptides and tripeptides. J. Photochem. Photobiol. B, Biol. 7, 159–172, (1990).
Xiong, Z. et al. How deep can plasma penetrate into a biofilm? Appl. Phys. Lett. 98, 221503, (2011).
Bernard, C. et al. Validation of cold plasma treatment for protein inactivation: a surface plasmon resonance-based biosensor study. J. Phys. D: Appl.Phys. 39, 3470–3478, (2006).
Davies, K. J., Delsignore, M. E. & Lin, S. W. Protein damage and degradation by oxygen radicals. II. Modification of amino acids. J. Biol. Chem. 262, 9902–9907 (1987).
Nordberg, J. & Arnér, E. S. Reactive oxygen species, antioxidants and the mammalian thioredoxin system. Free Radic. Biol. Med. 31, 1287–1312, (2001).
Cataldo, F. Ozone degradation of ribonucleic acid (RNA). Polym. Degrad. Stab. 89, 274–281, (2005).
Dill, K. A. Dominant forces in protein folding. Biochemistry 29, 7133–7155, (1990).
O'Brien, E. P., Dima, R. I., Brooks, B. & Thirumalai, D. Interactions between hydrophobic and ionic solutes in aqueous guanidinium chloride and urea solutions: lessons for protein denaturation mechanism. J. AM. CHEM. SOC. 129, 7346–7353, (2007).
Cabiscol, E., Tamarit, J. & Ros, J. Oxidative stress in bacteria and protein damage by reactive oxygen species. Int. Microbiol. 3, 3–8 (2000).
Roberts, C. R., Roughley, P. J. & Mort, J. S. Degradation of human proteoglycan aggregate induced by hydrogen peroxide. Protein fragmentation, amino acid modification and hyaluronic acid cleavage. Biochem. J. 259, 805–811 (1989).
Laroussi, M. & Leipold, F. Evaluation of the roles of reactive species, heat and UV radiation in the inactivation of bacterial cells by air plasmas at atmospheric pressure. Int J Mass Spectrom 233, 81–86, (2004).
Deng, X. T., Shi, J. J. & Kong, M. G. Protein destruction by a helium atmospheric pressure glow discharge: Capability and mechanisms. J. Appl. Phys. 101, 074701, (2007).
Attri, P. et al. Effects of atmospheric-pressure non-thermal plasma jets on enzyme solutions. J Korean Phys Soc. 60, 959–964, (2012).
Takai, E. et al. Degeneration of amyloid-ß fibrils caused by exposure to low-temperature atmospheric-pressure plasma in aqueous solution. Appl. Phys. Lett. 104, 023701, (2014).
Sass, C. et al. Characterization of rabbit lactate dehydrogenase-M and lactate dehydrogenase-H cDNAs. Control of lactate dehydrogenase expression in rabbit muscle. J. Biol. Chem. 264, 4076–4081 (1989).
Pankaj, S. K., Misra, N. N. & Cullen, P. J. Kinetics of tomato peroxidase inactivation by atmospheric pressure cold plasma based on dielectric barrier discharge. Innov. Food Sci.Emerg. Technol. 19, 153–157, (2013).
Moradian-Oldak, J., Leung, W. & Fincham, A. G. Temperature and pH-dependent supramolecular self-assembly of amelogenin molecules: a dynamic light-scattering analysis. J. Struct.biol. 122, 320–327, (1998).
Jans, H. et al. Dynamic light scattering as a powerful tool for gold nanoparticle bioconjugation and biomolecular binding studies. Anal.Chem. 81, 9425–9432, (2009).
Moradian-Oldak, J. et al. Self-assembly properties of recombinant engineered amelogenin proteins analyzed by dynamic light scattering and atomic force microscopy. J. Struct. biol. 131, 27–37, (2000).
Berlett, B. S. & Stadtman, E. R. Protein oxidation in aging, disease and oxidative stress. J.Biol. Chem. 272, 20313–20316 (1997).
Stadtman, E. R. & Berlett, B. S. Reactive oxygen-mediated protein oxidation in aging and disease. Chem. Res. Toxicol. 10, 485–494 (1997).
Guo, H. & Karplus, M. Solvent influence on the stability of the peptide hydrogen bond: a supramolecular cooperative effect. J. Phys. Chem. 98, 7104–7105, (1994).
Puorger, C. et al. Infinite kinetic stability against dissociation of supramolecular protein complexes through donor strand complementation. Structure 16, 631–642, (2008).
Song, D. & Forciniti, D. Effects of cosolvents and pH on protein adsorption on polystyrene latex: a dynamic light scattering study. J. Colloid Interface Sci. 221, 25–37, (2000).
Shen, J. et al. Characteristics of DC Gas‐Liquid Phase Atmospheric‐Pressure Plasma and Bacteria Inactivation Mechanism. Plasma Process. Polym. (2014), doi: 10.1002/ppap.201400129.
Acknowledgements
This work was jointly supported by the National Natural Science Foundation of Chinaunder Grant No. 11035005, No. 50876101 and No. 11005126, Hefei Institutes ofPhysical Science, Chinese Academy of Sciences (CASHIPS) Dean Funds No. YZJJ201331,City University of Hong Kong Applied Research Grant (ARG) No. 9667085, CityUniversity of Hong Kong Strategic Research Grant (SRG) No. 7004188, as well as HongKong Research Grants Council (RGC) General Research Funds (GRF) No. CityU112212.
Author information
Authors and Affiliations
Contributions
W.X. and C.C. led the project and supervised all experiments. H.Z., Z.X., J.S., X.L.,L.D. J.M., Y.L., Q.S. and Z.Z conducted experiments and measurements. W.X., C.C. andP.C. co-led data analysis and physical interpretations. All authors discussed theresults. H.Z, W.X., C.C. and P.C. co-wrote the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0International License. The images or other third party material in this article areincluded in the article’s Creative Commons license, unless indicatedotherwise in the credit line; if the material is not included under the CreativeCommons license, users will need to obtain permission from the license holder toreproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Zhang, H., Xu, Z., Shen, J. et al. Effects and Mechanism of Atmospheric-Pressure Dielectric Barrier Discharge Cold Plasmaon Lactate Dehydrogenase (LDH) Enzyme. Sci Rep 5, 10031 (2015). https://doi.org/10.1038/srep10031
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep10031
This article is cited by
-
Polymerization Process from the Cold Atmospheric Pressure Plasma Treatment of l-Cysteine Powder by Addition of DMSO and H2O2
Plasma Chemistry and Plasma Processing (2023)
-
Dielectric Barrier Discharge Cold Atmospheric Plasma Treatment of Egg White Protein: Insights into the Functional, Rheological, and Structural Properties
Food and Bioprocess Technology (2023)
-
Exploring the Role of Various Feed Gases in Cold Plasma Technology: A Comprehensive Review
Food and Bioprocess Technology (2023)
-
Mechanisms of bacterial inhibition and tolerance around cold atmospheric plasma
Applied Microbiology and Biotechnology (2023)
-
Degradation of carbamazepine by high-voltage direct current gas–liquid plasma with the addition of H2O2 and Fe2+
Environmental Science and Pollution Research (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.