Abstract
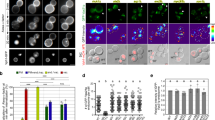
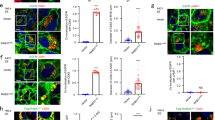
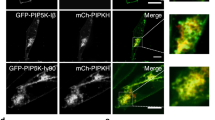
We show here that the GTPase RhoG is involved in caveolar trafficking. Wild-type RhoG moves sequentially to the plasma membrane, intracellular vesicles, and the Golgi apparatus along markers of this endocytic pathway. Such translocation is associated with changes in RhoG GDP/GTP levels and is highly dependent on lipid raft integrity and on the function of the GTPase dynamin2. In addition, the constitutively active RhoGQ61L mutant is preferentially located in endocytic vesicles that can be decorated with markers of the caveola-derived endocytic pathway. RhoGQ61L, but not the analogous Rac1 mutant protein, affects caveola internalization and the subsequent delivery of endocytic vesicles to the Golgi apparatus. The expression of RhoG/Rac1 chimeric proteins and RhoGQ61L effector mutants in cells induces alterations in the internalization of caveolae and severe changes in vesicle structure, respectively. However, the knockdown of endogenous rhoG transcripts using small interfering RNAs does not affect significantly the trafficking of caveola-derived vesicles, suggesting that RhoG function is dispensable for this endocytic process or, alternatively, that its function is compensated by other molecules. Taken together, these observations assign a novel function to RhoG and suggest that caveolar trafficking, as previously shown for other endocytic routes, is modulated by GTPases of the Ras superfamily.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Conner SD, Schmid SL . (2003). Nature 422: 37–44.
Cox D, Chang P, Zhang Q, Reddy PG, Bokoch GM, Greenberg S . (1997). J Exp Med 186: 1487–1494.
Di Guglielmo GM, Le Roy C, Goodfellow AF, Wrana JL . (2003). Nat Cell Biol 5: 410–421.
Ellis S, Mellor H . (2000). Trends Cell Biol 10: 85–88.
Felberbaum-Corti M, Van Der Goot FG, Gruenberg J . (2003). Nat Cell Biol 5: 382–384.
Filippi MD, Harris CE, Meller J, Gu Y, Zheng Y, Williams DA . (2004). Nat Immunol 5: 744–751.
Gauthier-Rouviere C, Vignal E, Meriane M, Roux P, Montcourier P, Fort P . (1998). Mol Biol Cell 9: 1379–1394.
Gonzalez-Gaitan M . (2003). Nat Rev Mol Cell Biol 4: 213–224.
Greenberg S . (1999). J Leukoc Biol 66: 712–717.
Gruenberg J . (2001). Nat Rev Mol Cell Biol 2: 721–730.
Henley JR, Krueger EW, Oswald BJ, McNiven MA . (1998). J Cell Biol 141: 85–99.
Hoppe AD, Swanson JA . (2004). Mol Biol Cell 15: 3509–3519.
Joneson T, McDonough M, Bar-Sagi D, Van Aelst L . (1996). Science 274: 1374–1376.
Katoh H, Negishi M . (2003). Nature 424: 461–464.
Katoh H, Yasui H, Yamaguchi Y, Aoki J, Fujita H, Mori K et al. (2000). Mol Cell Biol 20: 7378–7387.
Li G, Stahl PD . (1993). J Biol Chem 268: 24475–24480.
Massol P, Montcourrier P, Guillemot JC, Chavrier P . (1998). EMBO J 17: 6219–6229.
May V, Schiller MR, Eipper BA, Mains RE . (2002). J Neurosci 22: 6980–6990.
Nichols B . (2003). J Cell Sci 116: 4707–4714.
Nichols BJ . (2002). Nat Cell Biol 4: 374–378.
Nichols BJ, Kenworthy AK, Polishchuk RS, Lodge R, Roberts TH, Hirschberg K et al. (2001). J Cell Biol 153: 529–541.
Nichols BJ, Lippincott-Schwartz J . (2001). Trends Cell Biol 11: 406–412.
Orlandi PA, Fishman PH . (1998). J Cell Biol 141: 905–915.
Parton RG, Richards AA . (2003). Traffic 4: 724–738.
Pelkmans L, Fava E, Grabner H, Hannus M, Habermann B, Krausz E et al. (2005). Nature 436: 78–86.
Pelkmans L, Helenius A . (2002). Traffic 3: 311–320.
Pelkmans L, Zerial M . (2005). Nature 436: 128–133.
Prieto-Sanchez RM, Bustelo XR . (2003). J Biol Chem 278: 37916–37925.
Sabharanjak S, Sharma P, Parton RG, Mayor S . (2002). Dev Cell 2: 411–423.
Simons K, Toomre D . (2000). Nat Rev Mol Cell Biol 1: 31–39.
Torgersen ML, Skretting G, van Deurs B, Sandvig K . (2001). J Cell Sci 114: 3737–3747.
Vignal E, Blangy A, Martin M, Gauthier-Rouviere C, Fort P . (2001). Mol Cell Biol 21: 8022–8034.
Wennerberg K, Ellerbroek SM, Liu RY, Karnoub AE, Burridge K, Der CJ . (2002). J Biol Chem 277: 47810–47817.
Wu WJ, Erickson JW, Lin R, Cerione RA . (2000). Nature 405: 800–804.
Zerial M, McBride H . (2001). Nat Rev Mol Cell Biol 2: 107–117.
Acknowledgements
We thank M Blázquez and Dr P Fort for excellent technical assistance and the supply of anti-RhoG antibodies, respectively. We also thank Dr H Lee for the cloning of human rab cDNAs and the generation of rab point mutants. This work was supported by grants from both the US National Cancer Institute (5-R01-CA73735-08) and the Spanish Ministry of Education and Science (SAF2003-00028). RMP-S and IMB are students of the Molecular and Cellular Cancer Biology graduate program of the CIC and the University of Salamanca and are supported by FPU fellowships (AP2000-3829 and FP2000-6489; Spanish Ministry of Education and Science).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on Oncogene website (http://www.nature.com/onc)
Rights and permissions
About this article
Cite this article
Prieto-Sánchez, R., Berenjeno, I. & Bustelo, X. Involvement of the Rho/Rac family member RhoG in caveolar endocytosis. Oncogene 25, 2961–2973 (2006). https://doi.org/10.1038/sj.onc.1209333
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1209333
Keywords
This article is cited by
-
Mechanisms and functions of endocytosis in T cells
Cell Communication and Signaling (2021)
-
Functions of lipid raft membrane microdomains at the blood–brain barrier
Journal of Molecular Medicine (2009)