Abstract
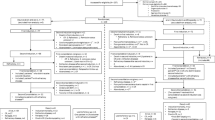
In this trial, acute myeloid leukemia patients (pts) aged 61–80 years received MICE (mitoxantrone, etoposide and cytarabine) induction chemotherapy in combination with different schedules of granulocyte colony-stimulating factor administration. Pts in complete remission were subsequently randomized for two cycles of consolidation therapy: mini-ICE regimen (idarubicin, etoposide and cytarabine) given according to either an intravenous (i.v.) or a ‘non-infusional’ schedule. Among the 346 pts randomized for the second step, 331 pts received consolidation-1 and 182 consolidation-2. A total of 290 events (255 relapses, 35 deaths in first CR) have been reported. The median follow-up was 4.4 years. No significant differences were detected in terms of disease-free survival (median 9 vs 10.4 months, P=0.15, hazard ratio (HR) =1.18, 95% confidence interval (CI) 0.94–1.49) – primary end point – and survival (median 15.7 vs 17.8 months, P=0.19, HR=1.17, 95% CI 0.92–1.50). In the ‘non-infusional’ arm grade 3–4 vomiting (10 vs 2%; P=0.001) and diarrhea (10 vs 4%; P=0.03) were higher than in the ‘i.v.’ arm, whereas time to platelet recovery >20 × 109/l (median: 19 vs 23 days; P=0.02) and duration of hospitalization (mean: 15 vs 27 days; P<0.0001) was shorter. The ‘non-infusional’ consolidation regimen resulted in an antileukemic effect similar to the intravenous regimen, which was less myelosuppressive and associated with less hospitalization days.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Archimbaud E, Jehn U, Thomas X, De Cataldo F, Fillet G, Belhabri A et al. Multicenter randomized phase II trial of idarubicin vs mitoxantrone, combined with VP-16 and cytarabine for induction/consolidation therapy, followed by a feasibility study of autologous peripheral blood stem cell transplantation in elderly patients with acute myeloid leukemia. Leukemia 1999; 13: 843–849.
Zittoun RA, Mandelli F, Willemze R, de Witte T, Labar B, Resegotti L et al. For the European Organization for Research and Treatment of Cancer (EORTC) and the Gruppo Italiano Malattie Ematologiche Maligne dell'Adulto (GIMEMA) Leukemia Cooperative Groups. Autologous or allogeneic bone marrow transplantation compared with intensive chemotherapy in acute myelogenous leukemia. New Engl J Med 1995; 332: 217–223.
Cassileth PA, Harrington DP, Appelbaum F, Lazarus HM, Rowe JM, Paietta E et al. Chemotherapy compared with autologous or allogeneic bone marrow transplantation in the management of acute myeloid leukemia in first remission. N Engl J Med 198; 339: 1649–1656.
Burnett AK, Goldstone AH, Stevens R, Hann IM, Rees JK, Gray RG et al. Randomised comparison of addition autologous bone-marrow transplantation to intensive chemotherapy for acute myeloid leukaemia in first remission: results of MRC AML 10 trial. Medical Research Council Adult and Children's Leukaemia Working Parties. Lancet 1998; 351: 700–708.
Lowenberg B, Suciu S, Archimbaud E, Haak H, Stryckmans P, de Cataldo R et al. Mitoxantrone versus daunorubicin in induction-consolidation chemotherapy – the value of low-dose cytarabine for maintenance of remission, and an assessment of prognostic factors in acute myeloid leukaemia in the elderly: final report. European Organization for the Research and Treatment of Cancer and the Dutch-Belgian Hemato-Oncology Cooperative Hovon Group. J Clin Oncol 1998; 16: 872–881.
Lowenberg B, Suciu S, Archimbaud E, Ossenkoppele G, Verhoef GE, Vellenga E et al. Use of recombinant Granulocyte-Macrophage Colony-Stimulating Factor during and after remission induction chemotherapy in patients aged 61 years and older: final report of AML-11, a randomized phase III study of the Leukemia Cooperative Group of the European Organisation for the Research and Treatment of Cancer and the Dutch Belgian Hemato-Oncology Cooperative Group. Blood 1997; 90: 2951–2961.
Hiddemann W, Kern W, Schoch C, Fonatsch C, Heinecke A, Wormann B et al. Management of acute myeloid leukaemia in elderly patients. J Clin Oncol 1999; 17: 3569–3576.
Goldstone AH, Burnett AK, Wheatley K, Smith AG, Hutchinson M, Clark RE, on behalf of the Medical Research Council Adult Leukaemia Working Party. Attempts to improve treatment outcomes in acute myeloid leukaemia (AML) in older patients: the results of the United Kingdom Medical Research Council AML 11 trial. Blood 2001; 98: 1302–1311.
Ruutu T, Almqvist A, Hallman H, Honkanen T, Jarvenpaa E, Jarventie G et al. Oral induction and consolidation of acute myeloid leukemia with etoposide, 6-thioguanine, and idarubicin (ETI) in elderly patients: a randomized comparison with 5-day TAD. Leukemia 1994; 8: 11–15.
Amadori S, Suciu S, Jehn U, Stasi R, Thomas X, Marie JP et al. Use of glycosylated recombinant human G-CSF (lenograstim) during and/or after induction chemotherapy in patients 61 years of age and older with acute myeloid leukemia: final results of AML-13, a randomized phase-3 study. Blood 2005; 106: 27–34.
Speth PA, van de Loo FA, Linssen PC, Wessels HM, Haanen C . Plasma and human leukemic cell pharmacokinetics of oral and intravenous 4-demethoxydaunomycin. Clin Pharmacol Ther 1986; 40: 643–649.
Thomas X, Suciu S, Marie JP, Leone G, Broccia G, Dryfus F et al. Feasibility and outcome of autologous stem cell transplantation planned after CR and first consolidation in AML patients aged 61–70 years: Results of the prospective EORTC-GIMEMA AML-13 study. Blood 2002; 100 (Suppl 11): 859a.
Yates J, Glidewell O, Wiernik P, Cooper MR, Steinberg D, Dosik H et al. Cytosine arabinoside with daunorubicin or adriamycin for therapy of acute myelocytic leukemia: a CALGB study. Blood 1982; 60: 454–462.
Mitelman F (ed). ISCN 1995: An International System for Human Cytogenetic Nomenclature. S. Karger: Basel, Switzerland, 1995.
Bennett JM, Catovsky D, Daniel MT, Flandrin G, Galton DA, Gralnick HR et al. Proposed revised criteria for the classification of the acute myeloid leukemia: a report of the French-American-British Cooperative Group. Ann Intern Med 1985; 103: 620–625.
Bennett JM, Catovsky D, Daniel MT, Flandrin G, Galton DA, Gralnick HR et al. Proposal for the recognition of minimally differentiated acute myeloid leukaemias (AML-M0). Br J Haematol 1991; 78: 325–329.
Kalbfleisch JD, Prentice RL . The Survival Analysis of Failure Time Data. Second edn. Wiley-Interscience: New Jersey, US, 2002.
Gray RJ . A class of K-sample tests for comparing the cumulative incidence of a competing risk. Annals Stat 1988; 16: 1141–1154.
Brinker H . Population-based age-and sex-specific incidence rates in the 4 main types of leukaemia. Scan J Haematol 1982; 29: 241–249.
Ost A, Lindstrom P, Christenson B, Gyllenhammer H, Engstedt L . Acute leukaemia in a defined geographic area-incidence, clinical history and prognosis. Scan J Haematol 1984; 33: 160–170.
Sorensen JT, Gerald K, Bodensteiner D, Holmes FF . Effect of age on survival in acute leukaemia. Cancer 1993; 72: 1602–1606.
Anderson JE, Kopecky KJ, Willman CL, Head D, O'Donnell MR, Luthardt FW et al. Outcome after induction chemotherapy for older patients with acute myeloid leukemia is not improved with mitoxantrone and etoposide compared to cytarabine and daunorubicin: a Southwest Oncology Group study. Blood 2002; 100: 3869–3876.
Acknowledgements
We acknowledge St Jude Children's Research Hospital for providing of a SAS macro allowing the computation of the cumulative incidences of relapse and of death in CR1, and their comparisons (Gray test).
We thank the Cytogeneticists of the different institutions and the members of the cytogenetic committee: A Bernheim (Institut Gustave Roussy, Villejuif), M Mancini (University La Sapienza, Rome), D Olde-Weghuis (Radboud University Nijmegen Medical Center, Nijmegen) and A Hagemeijer (UZ Gasthuisberg, Leuven). We thank the former data managers of this study: G Solbu, M Dardenne.This work was supported in part by grants from the National Cancer Institute (grant numbers 2U10-CA11488-25 through 2U10-CA11488-36). Its contents are solely the responsibility of the authors and do not represent the official views of the National Cancer Institute (Bethesda, MA, USA). This work was also supported by grants from Deutsche Krebshilfe (Germany). Pharmacia-Upjohn provided an educational grant and Zavedos free of charge. The list of EORTC-GIMEMA members who participated in this study is indicated in the appendix (‘Supplementary Information’).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Leukemia website (http://www.nature.com/leu)
Supplementary information
Rights and permissions
About this article
Cite this article
Jehn, U., Suciu, S., Thomas, X. et al. Non-infusional vs intravenous consolidation chemotherapy in elderly patients with acute myeloid leukemia: final results of the EORTC-GIMEMA AML-13 randomized phase III trial. Leukemia 20, 1723–1730 (2006). https://doi.org/10.1038/sj.leu.2404356
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2404356