Abstract
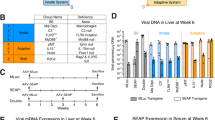
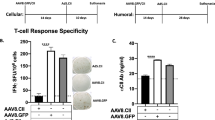
Adeno-associated virus (AAV) is widely considered a promising vector for therapeutic gene delivery. This promise is based on previous studies assessing AAVs safety and toxicity, ability to infect nondividing cells, elicit a limited immune response and provide long-term gene expression. However, we now find that earlier studies underappreciated the degree of AAV immunogenicity as well as the extent to which genetic background, through regulation of immune responsiveness, influences the duration of gene expression and thereby the effectiveness of AAV-mediated gene therapy. We evaluated antibody responses in 12 mouse strains to AAV serotype 2 (AAV2) and AAV2-expressed transgene products including green fluorescent protein (GFP), human α1-antitrypsin and murine interleukin-10. As expected, all immunocompetent mice administered AAV2 developed serologic evidence of immune responsiveness to the virus. However, a previously unidentified serologic prozone effect was observed suggesting that the concentrations of anti-AAV2 antibodies may have historically been subject to marked underestimation. Furthermore, strains with genetic predisposition to autoimmunity (eg, NOD, NZW, MRL-lpr) specifically imparted a functionally deleterious immune response to AAV-delivered transgene products. These findings suggest that more thorough studies of anti-AAV immunity should be performed, and that genetic predisposition to autoimmunity should be considered when assessing AAV efficacy and safety in humans.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Berns KI, Giraud C . Biology of adeno-associated virus. Curr Top Microbiol Immunol 1996; 218: 1–23.
Berns KI . The Gordon Wilson Lecture. From basic virology to human gene therapy. Trans Am Clin Climatol Assoc 1999; 110: 75–85.
Kay MA, Glorioso JC, Naldini L . Viral vectors for gene therapy: the art of turning infectious agents into vehicles of therapeutics. Nat Med 2001; 7: 33–40.
Stedman H et al. Phase I clinical trial utilizing gene therapy for limb girdle muscular dystrophy: alpha-, beta-, gamma-, or delta-sarcoglycan gene delivered with intramuscular instillations of adeno-associated vectors. Hum Gene Ther 2000; 11: 777–790.
Wagner JA et al. Safety and biological efficacy of an adeno-associated virus vector-cystic fibrosis transmembrane regulator (AAV-CFTR) in the cystic fibrosis maxillary sinus. Laryngoscope 1999; 109: 266–274.
Kay MA et al. Evidence for gene transfer and expression of factor IX in haemophilia B patients treated with an AAV vector. Nat Genet 2000; 24: 257–261.
Marshall E . Gene therapy. Viral vectors still pack surprises. Science 2001; 294: 1640.
Donsante A et al. Observed incidence of tumorigenesis in long-term rodent studies of rAAV vectors. Gene Therapy 2001; 8: 1343–1346.
Blacklow NR, Hoggan MD, Rowe WP . Serologic evidence for human infection with adenovirus-associated viruses. J Natl Cancer Inst 1968; 40: 319–327.
Erles K, Sebokova P, Schlehofer JR . Update on the prevalence of serum antibodies (IgG and IgM) to adeno-associated virus (AAV). J Med Virol 1999; 59: 406–411.
Moskalenko M et al. Epitope mapping of human anti-adeno-associated virus type 2 neutralizing antibodies: implications for gene therapy and virus structure. J Virol 2000; 74: 1761–1766.
Hildinger M et al. Hybrid vectors based on adeno-associated virus serotypes 2 and 5 for muscle-directed gene transfer. J Virol 2001; 75: 6199–6203.
Xiao W et al. Gene therapy vectors based on adeno-associated virus type 1. J Virol 1999; 73: 3994–4003.
Halbert CL, Standaert TA, Wilson CB, Miller AD . Successful readministration of adeno-associated virus vectors to the mouse lung requires transient immunosuppression during the initial exposure. J Virol 1998; 72: 9795–9805.
Manning WC et al. Transient immunosuppression allows transgene expression following readministration of adeno-associated viral vectors. Hum Gene Ther 1998; 9: 477–485.
Xiao W et al. Route of administration determines induction of T-cell-independent humoral responses to adeno-associated virus vectors. Mol Ther 2000; 1: 323–329.
Ge Y, Powell S, Van Roey M, McArthur JG . Factors influencing the development of an anti-factor IX (FIX) immune response following administration of adeno-associated virus-FIX. Blood 2001; 97: 3733–3737.
Chirmule N et al. Humoral immunity to adeno-associated virus type 2 vectors following administration to murine and nonhuman primate muscle. J Virol 2000; 74: 2420–2425.
Beck SE et al. Repeated delivery of adeno-associated virus vectors to the rabbit airway. J Virol 1999; 73: 9446–9455.
Bennett J, Anand V, Acland GM, Maguire AM . Cross-species comparison of in vivo reporter gene expression after recombinant adeno-associated virus-mediated retinal transduction. Methods Enzymol 2000; 316: 777–789.
Brockstedt DG et al. Induction of immunity to antigens expressed by recombinant adeno-associated virus depends on the route of administration. Clin Immunol 1999; 92: 67–75.
Sarukhan A, Soudais C, Danos O, Jooss K . Factors influencing cross-presentation of non-self antigens expressed from recombinant adeno-associated virus vectors. J Gene Med 2001; 3: 260–270.
Goudy K et al. Adeno-associated virus vector mediated interleukin-10 gene delivery prevents type 1 diabetes in NOD mice. Proc Natl Acad Sci USA 2001; 98: 13913–13918.
Carpenter AB . Enzyme-linked immunoassays. In: Rose NR, de Macario EC, Folds JD, Lane HC, Nakamura RM (eds). Manual of Clinical Laboratory Immunology. ASM Press: Washington, DC, 1997, pp 20–29.
Murphy ED . Lymphoproliferation (lpr) and other single-locus models for murine lupus. In: Gershwin ME, Merchant B (eds). Immunologic Defects in Laboratory Animals. London: Plenum Press: New York, 1981, pp 143–173.
Jurado RL, Campbell J, Martin PD . Prozone phenomenon in secondary syphilis. Has its time arrived? Arch Intern Med 1993; 153: 2496–2498.
Chirmule N et al. Immune responses to adenovirus and adeno-associated virus in humans. Gene Therapy 1999; 6: 1574–1583.
Hernandez YJ et al. Latent adeno-associated virus infection elicits humoral but not cell-mediated immune responses in a nonhuman primate model. J Virol 1999; 73: 8549–8558.
Zaiss AK et al. Differential activation of innate immune responses by adenovirus and adeno-associated virus vectors. J Virol 2002; 76: 4580–4590.
Song S et al. Sustained secretion of human alpha-1-antitrypsin from murine muscle transduced with adeno-associated virus vectors. Proc Natl Acad Sci USA 1998; 95: 14384–14388.
Lee HC et al. Remission in models of type 1 diabetes by gene therapy using a single-chain insulin analogue. Nature 2000; 408: 483–488.
Liu Y et al. Strain-based genetic differences regulate the efficiency of systemic gene delivery as well as expression. J Biol Chem 2002; 277: 4966–4972.
Ito K, Takeuchi Y, Kato S . Strain-dependent antibody response induced by DNA immunization. Immunol Lett 2000; 74: 245–250.
Blaikie L, Basketter DA . Strain variation in the IgG1 antibody response to proteins administered intranasally in the mouse. Food Chem Toxicol 1999; 37: 897–904.
Fields PA et al. Risk and prevention of anti-factor IX formation in AAV-mediated gene transfer in the context of a large deletion of F9. Mol Ther 2001; 4: 201–210.
Lo WD et al. Adeno-associated virus-mediated gene transfer to the brain: duration and modulation of expression. Hum Gene Ther 1999; 10: 201–213.
Cordier L et al. Muscle-specific promoters may be necessary for adeno-associated virus-mediated gene transfer in the treatment of muscular dystrophies. Hum Gene Ther 2001; 12: 205–215.
Liu DW et al. Recombinant adeno-associated virus expressing human papillomavirus type 16 E7 peptide DNA fused with heat shock protein DNA as a potential vaccine for cervical cancer. J Virol 2000; 74: 2888–2894.
Manning WC et al. Genetic immunization with adeno-associated virus vectors expressing herpes simplex virus type 2 glycoproteins B and D. J Virol 1997; 71: 7960–7962.
Xin KQ et al. A novel recombinant adeno-associated virus vaccine induces a long-term humoral immune response to human immunodeficiency virus. Hum Gene Ther 2001; 12: 1047–1061.
Anand V et al. A deviant immune response to viral proteins and transgene product is generated on subretinal administration of adenovirus and adeno-associated virus. Mol Ther 2002; 5: 125–132.
Zolotukhin S et al. Recombinant adeno-associated virus purification using novel methods improves infectious titer and yield. Gene Therapy 1999; 6: 973–985.
Hauswirth WW, Lewin AS, Zolotukhin S, Muzyczka N . Production and purification of recombinant adeno-associated virus. Methods Enzymol 2000; 316: 743–761.
Acknowledgements
These studies were funded by the Juvenile Diabetes Research Foundation, The National Institutes of Health, and Endowments from The Powel Gene Therapy Center and Sebastian Family Professorship.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zhang, Y., Powers, M., Wasserfall, C. et al. Immunity to adeno-associated virus serotype 2 delivered transgenes imparted by genetic predisposition to autoimmunity. Gene Ther 11, 233–240 (2004). https://doi.org/10.1038/sj.gt.3302144
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302144
Keywords
This article is cited by
-
Local and systemic responses following intravitreous injection of AAV2-encoded modified Volvox channelrhodopsin-1 in a genetically blind rat model
Gene Therapy (2016)
-
Immune responses to adeno-associated virus type 2 encoding channelrhodopsin-2 in a genetically blind rat model for gene therapy
Gene Therapy (2011)
-
Effect of viral dose on neutralizing antibody response and transgene expression after AAV1 vector re-administration in mice
Gene Therapy (2008)
-
Efficient and persistent transduction of exocrine and endocrine pancreas by adeno-associated virus type 8
Journal of Biomedical Science (2007)
-
Long-term expression and repeated administration of AAV type 1, 2 and 5 vectors in skeletal muscle of immunocompetent adult mice
Gene Therapy (2006)