Abstract
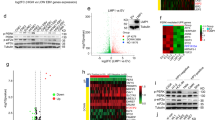
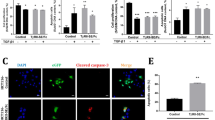
The tumor-suppressor gene PTEN encodes a multifunctional phosphatase that is mutated in a variety of human cancers. PTEN inhibits the phosphatidylinositol 3-kinase pathway and downstream functions, including activation of Akt/protein kinase B (PKB), cell survival, and cell proliferation in tumor cells carrying mutant- or deletion-type PTEN. In such tumor cells, enforced expression of PTEN decreases cell proliferation through cell-cycle arrest at G1 phase accompanied, in some cases, by induction of apoptosis. More recently, the tumor-suppressive effect of PTEN has been reported in ovarian and thyroid tumors that are wild type for PTEN. In the present study, we examined the tumor-suppressive effect of PTEN in human colorectal cancer cells that are wild type for PTEN. Adenoviral-mediated transfer of PTEN (Ad-PTEN) suppressed cell growth and induced apoptosis significantly in colorectal cancer cells (DLD-1, HT29, and SW480) carrying wtPTEN than in normal colon fibroblast cells (CCD-18Co) carrying wtPTEN. This suppression was induced through downregulation of the Akt/PKB pathway, dephosphorylation of focal adhesion kinase (FAK) and mitogen-activated protein kinase (MAPK) and cell-cycle arrest at the G2/M phase, but not the G1 phase. Furthermore, treatment of human colorectal tumor xenografts (HT-29, and SW480) with Ad-PTEN resulted in significant (P=0.01) suppression of tumor growth. These results indicate that Ad-PTEN exerts its tumor-suppressive effect on colorectal cancer cells through inhibition of cell-cycle progression and induction of cell death. Thus Ad-PTEN may be a potential therapeutic for treatment of colorectal cancers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Steck PA et al. Identification of a candidate tumour suppressor gene, MMAC1, at chromosome 10q23.3 that is mutated in multiple advanced cancers. Nat Genet 1997; 15: 356–362.
Li J et al. PTEN, a putative protein tyrosine phosphatase gene mutated in human brain, breast, and prostate cancer. Science 1997; 275: 1943–1947.
Rasheed BK et al. PTEN gene mutations are seen in high-grade but not in low-grade gliomas. Cancer Res 1997; 57: 4187–4190.
Wang SI et al. Somatic mutations of PTEN in glioblastoma multiforme. Cancer Res 1997; 57: 4183–4186.
Tashiro H et al. Mutations in PTEN are frequent in endometrial carcinoma but rare in other common gynecological malignancies. Cancer Res 1977; 57: 3935–3940.
Cairns P et al. Frequent inactivation of PTEN/MMAC1 in primary prostate cancer. Cancer Res 1997; 57: 4997–5000.
Yokomizo A et al. PTEN/MMAC1 mutations identified in small cell, but not in non-small cell lung cancers. Oncogene 1998; 17: 475–479.
Nelen MR et al. Germline mutations in the PTEN/MMAC1 gene in patients with Cowden disease. Hum Mol Genet 1997; 6: 1383–1387.
Liaw D et al. Germline mutations of the PTEN gene in Cowden disease, an inherited breast and thyroid cancer syndrome. Nat Genet 1997; 16: 64–67.
Marsh DJ et al. Germline mutations in PTEN are present in Bannayan–Zonana syndrome. Nat Genet 1997; 16: 333–334.
Maehama T, Dixon JE . The tumor suppressor, PTEN/MMAC1, dephosphorylates the lipid second messenger, phosphatidylinositol 3,4,5-triphosphate. J Biol Chem 1998; 273: 13375–13378.
Myers MP et al. P-TEN, the tumor suppressor from human chromosome 10q23, is a dual-specificity phosphatase. Proc Natl Acad Sci USA 1997; 94: 9052–9057.
Tamura M et al. Inhibition of cell migration, spreading, and focal adhesions by tumor suppressor PTEN. Science 1998; 280: 1614–1617.
Gu J, Tamura M, Yamada KM . Tumor suppressor PTEN inhibits integrin- and growth factor-mediated mitogen-activated protein (MAP) kinase signaling pathways. J Cell Biol 1998; 143: 1375–1383.
Stambolic V et al. Negative regulation of PKB/Akt-dependent cell survival by the tumor suppressor PTEN. Cell 1998; 95: 29–39.
Cheney IW et al. Suppression of tumorigenicity of glioblastoma cells by adenovirus-mediated MMAC1/PTEN gene transfer. Cancer Res 1998; 58: 2331–2334.
Li D M, Sun H . PTEN/MMAC1/TEP1 suppresses the tumorigenicity and induces G1 cell cycle arrest in human glioblastoma cells. Proc Natl Acad Sci USA 1998; 95: 15406–15411.
Furnari FB, Huang HJ, Cavenee WK . The phosphoinositol phosphatase activity of PTEN mediates a serum-sensitive G1 growth arrest in glioma cells. Cancer Res 1998; 58: 5002–5008.
Davies MA et al. Adenoviral transgene expression of MMAC/PTEN in human glioma cells inhibits Akt activation and induces anoikis. Cancer Res 1998; 58: 5285–5290.
Minaguchi T et al. Growth suppression of human ovarian cancer cells by adenovirus-mediated transfer of the PTEN gene. Cancer Res 1999; 59: 6063–6067.
Weng LP et al. Transient ectopic expression of PTEN in thyroid cancer cell lines induces cell cycle arrest and cell type-dependent cell death. Hum Mol Genet 2001; 10: 251–258.
Li J et al. The PTEN/MMAC1 tumor suppressor induces cell death that is rescued by the AKT/protein kinase B oncogene. Cancer Res 1998; 58: 5667–5672.
Dicuonzo G et al. Colorectal carcinomas and PTEN/MMAC1 gene mutations. Clin Cancer Res 2001; 7: 4049–4053.
Gottschalk AR et al. p27Kip1 is required for PTEN-induced G1 growth arrest. Cancer Res 2001; 61: 2105–2111.
Weng LP, Brown JL, Eng C . PTEN coordinates G(1) arrest by down-regulating cyclin D1 via its protein phosphatase activity and up-regulating p27 via its lipid phosphatase activity in a breast cancer model. Hum Mol Genet 2001; 10: 599–604.
Taylor V et al. 5′ phospholipid phosphatase SHIP-2 causes protein kinase B inactivation and cell cycle arrest in glioblastoma cells. Mol Cell Biol 2000; 20: 6860–6871.
Nakamura N et al. Forkhead transcription factors are critical effectors of cell death and cell cycle arrest downstream of PTEN. Mol Cell Biol 2000; 20: 8969–8982.
Dijkers PF et al. Forkhead transcription factor FKHR-L1 modulates cytokine-dependent transcriptional regulation of p27(KIP1). Mol Cell Biol 2000; 20: 9138–9148.
Lu Y et al. The PTEN/MMAC1/TEP tumor suppressor gene decreases cell growth and induces apoptosis and anoikis in breast cancer cells. Oncogene 1999; 18: 7034–7045.
Tamura M, Gu J, Takino T, Yamada KM . Tumor suppressor PTEN inhibition of cell invasion, migration, and growth: differential involvement of focal adhesion kinase and p130Cas. Cancer Res 1999; 59: 442–449.
Gu J et al. Shc and FAK differentially regulate cell motility and directionality modulated by PTEN. J Cell Biol 1999; 146: 389–403.
Peng CY et al. Mitotic and G2 checkpoint control: regulation of 14-3-3 protein binding by phosphorylation of Cdc25C on serine-216. Science 1997; 277: 1501–1505.
Dalal SN, Schweitzer CM, Gan J, DeCaprio JA . Cytoplasmic localization of human cdc25C during interphase requires an intact 14-3-3 binding site. Mol Cell Biol 1999; 19: 4465–4479.
Georgescu MM, Kirsch KH, Akagi T, Shishido T, Hanafusa H . The tumor-suppressive activity of PTEN is regulated by its carboxyl-terminal region. Proc Natl Acad Sci USA 1999; 96: 10182–10187.
Furnari FB, Lin H, Huang HS, Cavenee WK . Growth suppression of glioma cells by PTEN requires a functional phosphatase catalytic domain. Proc Natl Acad Sci USA 1997; 94: 12479–12484.
Robertson GP et al. In vitro loss of heterozygosity targets the PTEN/MMAC1 gene in melanoma. Proc Natl Acad Sci USA 1998; 95: 9418–9423.
Cheney IW et al. Adenovirus-mediated gene transfer of MMAC1/PTEN to glioblastoma cells inhibits S phase entry by the recruitment of p27Kip1 into cyclin E/CDK2 complexes. Cancer Res 1999; 59: 2318–2323.
Stewart A et al. PI3 kinase blockade by Ad-PTEN inhibits invasion and induces apoptosis in RGP and metastatic melanoma cells. Mol Med 2002; 8: 451–461.
Dunphy WG, Kumagai A . The cdc25 protein contains an intrinsic phosphatase activity. Cell 1991; 67: 189–196.
Kastan MB et al. Participation of p53 protein in the cellular response to DNA damage. Cancer Res 1991; 51: 6304–6311.
Sebastian B, Kakizuka A, Hunter T . Cdc25M2 activation of cyclin-dependent kinases by dephosphorylation of threonine-14 and tyrosine-15. Proc Natl Acad Sci USA 1993; 90: 3521–3524.
Saeki T et al. Tumor suppressive effects by adenovirus-mediated mda-7 gene transfer in non-small cell lung cancer cells in vitro. Gene Therapy 2000; 35: 2015–2057.
Saeki T et al. Inhibition of human lung cancer growth following adenovirus mediated mda-7 gene expression in vivo. Oncogene 2002; 21: 4558–4566.
Acknowledgements
We thank Dr Jack Roth for his critical comments, Don Norwood for editorial assistance, and Peggy James for preparation of this manuscript. This work was supported in part by the Texas Higher Education Coordinating Board ATP/ARP grant (003657-0078-2001; RR), by a Career Development award from the University of Texas SPORE in Lung Cancer (P50 CA70907; RR), by the University of Texas MD Anderson Cancer Center Institutional Grant (RR), by the KECK Foundation Fund for Human Cancer Gene Prevention and Therapy (RR), by BESCT Lung Cancer Program Grant (DAMD17-01-1-0689), by TARGET Lung Cancer grant (DAMD17-01-0706), by the University of Texas MD Anderson Cancer Center Support Grant (CA 16672), and by a sponsored research agreement with Introgen Therapeutics, Inc.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Saito, Y., Swanson, X., Mhashilkar, A. et al. Adenovirus-mediated transfer of the PTEN gene inhibits human colorectal cancer growth in vitro and in vivo. Gene Ther 10, 1961–1969 (2003). https://doi.org/10.1038/sj.gt.3302100
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302100
Keywords
This article is cited by
-
Saccharomyces cerevisiae inhibits growth and metastasis and stimulates apoptosis in HT-29 colorectal cancer cell line
Comparative Clinical Pathology (2019)
-
Synergistic tumor suppression by adenovirus-mediated ING4/PTEN double gene therapy for gastric cancer
Cancer Gene Therapy (2016)
-
The function, mechanisms, and role of the genes PTEN and TP53 and the effects of asbestos in the development of malignant mesothelioma: a review focused on the genes' molecular mechanisms
Tumor Biology (2014)
-
Role of SALL4 in the progression and metastasis of colorectal cancer
Journal of Biomedical Science (2013)
-
Enhanced tumor suppression by adenoviral PTEN gene therapy combined with cisplatin chemotherapy in small-cell lung cancer
Cancer Gene Therapy (2013)