Abstract
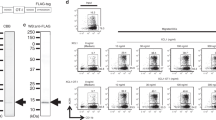
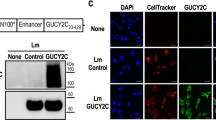
Human telomerase reverse transcriptase (TERT) has been considered a potential tumor-associated antigen for active-specific immunotherapy. However, effective specific tumor antigen-specific immunity has been difficult to induce consistently by various TERT vaccine formulations. New adjuvant strategies have been employed, such as utilizing chemokines to attract T cells and antigen-presenting cells. Chemokine adjuvant strategies may enhance tumor antigen-specific immunity induced by vaccines. Therefore, we utilized chemokine ligand 21 (CCL21) as an adjuvant with a xenogeneic TERT DNA vaccine to induce tumor antigen-specific immunity against TERT-expressing breast cancer. The TERT DNA vaccine consisted of a plasmid containing the COOH terminal end of the TERT (cTERT) gene, encapsulated in multilayered liposomes with hemagglutinating virus of Japan coating. We demonstrated that CCL21 treatment before cTERT DNA vaccine, given intramuscularly, induced significantly higher anti-TERT specific cell-mediated immunity compared to cTERT DNA vaccine alone. Effective tumor antigen-specific immunity was shown both in prophylactic and therapeutic regimens against TS/A murine breast cancer. The study demonstrated that CCL21 administration before cTERT DNA vaccination significantly augmented tumor antigen-specific immunity against breast cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Abbreviations
- APCs:
-
antigen-presenting cells
- CCL21:
-
chemokine ligand 21
- CCL21/cTERT vaccine:
-
cTERT DNA vaccine plus CCL21 24 h before
- hTERT:
-
human TERT
- cTERT:
-
COOH-terminal end of TERT
- DC:
-
dendritic cells
- DTH:
-
delayed-type hypersensitivity
- HVJ:
-
hemagglutinating virus of Japan
- TAA:
-
tumor-associated antigen
- TERT:
-
telomerase reverse transcriptase
- TRP2:
-
tyrosinase-related protein 2
References
Wang RF, Rosenberg SA . Human tumor antigens for cancer vaccine development. Immunol Rev 1999; 170: 85–100.
Nakamura TM, Morin GB, Chapman KB, Weinrich SL, Andrews WH, Lingner J et al. Telomerase catalytic subunit homologs from fission yeast and human. Science 1997; 277: 955–959.
Kim NW, Piatyszek MA, Prowse KR, Harley CB, West MD, Ho PL et al. Specific association of human telomerase activity with immortal cells and cancer. Science 1994; 266: 2011–2015.
Hahn WC, Stewart SA, Brooks MW, York SG, Eaton E, Kurachi A et al. Inhibition of telomerase limits the growth of human cancer cells. Nat Med 1999; 5: 1164–1170.
Liu K, Schoonmaker MM, Levine BL, June CH, Hodes RJ, Weng NP . Constitutive and regulated expression of telomerase reverse transcriptase (hTERT) in human lymphocytes. Proc Natl Acad Sci USA 1999; 96: 5147–5152.
Harada K, Kurisu K, Arita K, Sadatomo T, Tahara H, Tahara E et al. Telomerase activity in central nervous system malignant lymphoma. Cancer 1999; 86: 1050–1055.
Bieche I, Nogues C, Paradis V, Olivi M, Bedossa P, Lidereau R et al. Quantitation of hTERT gene expression in sporadic breast tumors with a real-time reverse transcription-polymerase chain reaction assay. Clin Cancer Res 2000; 6: 452–459.
Vonderheide RH, Hahn WC, Schultze JL, Nadler LM . The telomerase catalytic subunit is a widely expressed tumor-associated antigen recognized by cytotoxic T lymphocytes. Immunity 1999; 10: 673–679.
Vonderheide RH . Telomerase as a universal tumor-associated antigen for cancer immunotherapy. Oncogene 2002; 21: 674–679.
Minev B, Hipp J, Firat H, Schmidt JD, Langlade-Demoyen P, Zanetti M . Cytotoxic T cell immunity against telomerase reverse transcriptase in humans. Proc Natl Acad Sci USA 2000; 97: 4796–4801.
Nair SK, Heiser A, Boczkowski D, Majumdar A, Naoe M, Lebkowski JS et al. Induction of cytotoxic T cell responses and tumor immunity against unrelated tumors using telomerase reverse transcriptase RNA transfected dendritic cells. Nat Med 2000; 6: 1011–1017.
Ayyoub M, Migliaccio M, Guillaume P, Lienard D, Cerottini JC, Romero P et al. Lack of tumor recognition by hTERT peptide 540-548-specific CD8(+) T cells from melanoma patients reveals inefficient antigen processing. Eur J Immunol 2001; 31: 2642–2651.
Vonderheide RH, Schultze JL, Anderson KS, Maecker B, Butler MO, Xia Z et al. Equivalent induction of telomerase-specific cytotoxic T lymphocytes from tumor-bearing patients and healthy individuals. Cancer Res 2001; 61: 8366–8370.
Schroers R, Huang XF, Hammer J, Zhang J, Chen SY . Identification of HLA DR7-restricted epitopes from human telomerase reverse transcriptase recognized by CD4+ T-helper cells. Cancer Res 2002; 62: 2600–2605.
Su Z, Vieweg J, Weizer AZ, Dahm P, Yancey D, Turaga V et al. Enhanced induction of telomerase-specific CD4(+) T cells using dendritic cells transfected with RNA encoding a chimeric gene product. Cancer Res 2002; 62: 5041–5048.
Scardino A, Gross DA, Alves P, Schultze JL, Graff-Dubois S, Faure O et al. HER-2/neu and hTERT cryptic epitopes as novel targets for broad spectrum tumor immunotherapy. J Immunol 2002; 168: 5900–5906.
Gross DA, Graff-Dubois S, Opolon P, Cornet S, Alves P, Bennaceur-Griscelli A et al. High vaccination efficiency of low-affinity epitopes in antitumor immunotherapy. J Clin Invest 2004; 113: 425–433.
Vonderheide RH, Domchek SM, Schultze JL, George DJ, Hoar KM, Chen DY et al. Vaccination of cancer patients against telomerase induces functional antitumor CD8+ T lymphocytes. Clin Cancer Res 2004; 10: 828–839.
Zou W . Immunosuppressive networks in the tumour environment and their therapeutic relevance. Nat Rev Cancer 2005; 5: 263–274.
Zhou WZ, Kaneda Y, Huang S, Morishita R, Hoon D . Protective immunization against melanoma by gp100 DNA-HVJ-liposome vaccine. Gene Therapy 1999; 6: 1768–1773.
Tanaka M, Kaneda Y, Fujii S, Yamano T, Hashimoto K, Huang SK et al. Induction of a systemic immune response by a polyvalent melanoma-associated antigen DNA vaccine for prevention and treatment of malignant melanoma. Mol Ther 2002; 5: 291–299.
Yamano T, Kaneda Y, Huang S, Hiramatsu SH, Hoon DS . Enhancement of Immunity by a DNA Melanoma Vaccine against TRP2 with CCL21 as an Adjuvant. Mol Ther 2006; 13: 194–202.
Gold JS, Ferrone CR, Guevara-Patino JA, Hawkins WG, Dyall R, Engelhorn ME et al. A single heteroclitic epitope determines cancer immunity after xenogeneic DNA immunization against a tumor differentiation antigen. J Immunol 2003; 170: 5188–5194.
Homey B, Muller A, Zlotnik A . Chemokines: agents for the immunotherapy of cancer? Nat Rev Immunol 2002; 2: 175–184.
Cyster JG . Chemokines and cell migration in secondary lymphoid organs. Science 1999; 286: 2098–2102.
Nanni P, De Giovanni C, Landuzzi L, Nicoletti G, Frabetti F, Rossi I et al. Therapy of murine mammary carcinoma metastasis with interferon gamma and MHC gene-transduced tumour cells. Br J Cancer 1996; 74: 1564–1569.
Rodriguez F, Zhang J, Whitton JL . DNA immunization: ubiquitination of a viral protein enhances cytotoxic T-lymphocyte induction and antiviral protection but abrogates antibody induction. J Virol 1997; 71: 8497–8503.
del Guercio MF, Alexander J, Kubo RT, Arrhenius T, Maewal A, Appella E et al. Potent immunogenic short linear peptide constructs composed of B cell epitopes and Pan DR T helper epitopes (PADRE) for antibody responses in vivo. Vaccine 1997; 15: 441–448.
Ishioka GY, Fikes J, Hermanson G, Livingston B, Crimi C, Qin M et al. Utilization of MHC class I transgenic mice for development of minigene DNA vaccines encoding multiple HLA-restricted CTL epitopes. J Immunol 1999; 162: 3915–3925.
Kolquist KA, Ellisen LW, Counter CM, Meyerson M, Tan LK, Weinberg RA et al. Expression of TERT in early premalignant lesions and a subset of cells in normal tissues. Nat Genet 1998; 19: 182–186.
Kusakabe K, Xin KQ, Katoh H, Sumino K, Hagiwara E, Kawamoto S et al. The timing of GM-CSF expression plasmid administration influences the Th1/Th2 response induced by an HIV-1-specific DNA vaccine. J Immunol 2000; 164: 3102–3111.
Barth A, Hoon DS, Foshag LJ, Nizze JA, Famatiga E, Okun E et al. Polyvalent melanoma cell vaccine induces delayed-type hypersensitivity and in vitro cellular immune response. Cancer Res 1994; 54: 3342–3345.
Soiffer R, Lynch T, Mihm M, Jung K, Rhuda C, Schmollinger JC et al. Vaccination with irradiated autologous melanoma cells engineered to secrete human granulocyte-macrophage colony-stimulating factor generates potent antitumor immunity in patients with metastatic melanoma. Proc Natl Acad Sci USA 1998; 95: 13141–13146.
Xiang R, Mizutani N, Luo Y, Chiodoni C, Zhou H, Mizutani M et al. A DNA vaccine targeting survivin combines apoptosis with suppression of angiogenesis in lung tumor eradication. Cancer Res 2005; 65: 553–561.
Shimizu K, Kuriyama H, Kjaergaard J, Lee W, Tanaka H, Shu S . Comparative analysis of antigen loading strategies of dendritic cells for tumor immunotherapy. J Immunother 2004; 27: 265–272.
Vicari AP, Caux C, Trinchieri G . Tumour escape from immune surveillance through dendritic cell inactivation. Semin Cancer Biol 2002; 12: 33–42.
Acknowledgements
We thank the members of Department of Molecular Oncology JWCI and Cedars Sinai Vivarium personnel for their assistance. This work was supported in part by the Komen Breast Center Program and Gonda Laboratory, JWCI.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yamano, T., Kaneda, Y., Hiramatsu, S. et al. Immunity against breast cancer by TERT DNA vaccine primed with chemokine CCL21. Cancer Gene Ther 14, 451–459 (2007). https://doi.org/10.1038/sj.cgt.7701035
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cgt.7701035
Keywords
This article is cited by
-
Nanotechnology in the arena of cancer immunotherapy
Archives of Pharmacal Research (2020)