Abstract
Glial cell-line-derived neurotrophic factor (GDNF) and neurturin (NTN) protect retinal ganglion cells (RGCs) from axotomy-induced apoptosis. It is likely that neuroprotection by GDNF or NTN in the adult central nervous system (CNS) involves indirect mechanisms and independent signal transduction events. Extracellular glutamate is a trigger of apoptosis in injured RGCs, and glutamate transporter levels can be upregulated by GDNF. Therefore, GDNF may indirectly protect RGCs by enhancing glutamate uptake in the retina. We studied the upregulation of the glutamate transporters GLAST-1 and GLT-1 by GDNF and NTN, and the intracellular pathways required for GDNF/NTN neuroprotection. GDNF required phosphoinositide-3 kinase (PI3K) and Src activity to upregulate GLAST-1 and GLT-1. NTN required PI3K activity to upregulate GLAST-1 and did not affect GLT-1 levels. PI3K activity was also important for GDNF and NTN neuroprotection following optic nerve transection. However, GDNF also required Src and mitogen-activated protein kinase activity to prevent RGC apoptosis. RNA interference demonstrated that the upregulation of GLAST-1 by GDNF and NTN is required to rescue RGCs. Thus, additional independent signal transduction events, together with the upregulation of GLT-1 by GDNF, differentiate the biological activity of GDNF from NTN. Furthermore, the upregulation of the glial glutamate transporter GLAST-1 by both factors is an indirect neuroprotective mechanism in the CNS.
Similar content being viewed by others
Main
Optic nerve transection is a widely used model of neuronal apoptosis in the injured adult central nervous system (CNS). Approximately 90% of retinal ganglion cells (RGCs) undergo apoptosis within the first 2 weeks after optic nerve transection.1 The triggers for RGC apoptosis after axotomy include the loss of trophic support, glutamate excitotoxicity, reactive oxygen species toxicity, and inflammatory responses by surrounding glia.1
Glial cell-line-derived neurotrophic factor (GDNF) has previously been shown to reduce apoptosis and enhance the survival of axotomized RGCs.2, 3, 4 GDNF signals through the ligand binding receptor GFRα-1 and the tyrosine kinase receptor Ret. Ret is expressed in the inner nuclear layer and ganglion cell layer of the mammalian retina.5 GFRα-1 is also localized to RGCs; however, the majority of GFRα-1 immunoreactivity in the retina is localized to Müller cells,4 suggesting that retinal glia are a major target for GDNF. GDNF signal transduction through the phosphoinositide-3 kinase (PI3K), mitogen-activated protein kinase (MAPK), and Src kinase pathways has been shown to prevent apoptosis, promote neuronal development and differentiation, and promote axonal regeneration in the CNS.6
Neurturin (NTN), a relative of GDNF, also promotes the survival of injured RGCs, producing enhanced effects when delivered in combination with GDNF.4 NTN transduces signals through Ret by binding to the high affinity receptor GFRα-2. Interestingly, the pattern of GFRα-2 mRNA expression suggests that retinal Müller glia, in addition to RGCs, may express NTN receptors.5 Given these findings, it is also possible that glia play a role in NTN-mediated survival responses after RGC axotomy. Similar to GDNF, signal transduction by NTN via the PI3K, MAPK, and Src pathways is known to contribute to antiapoptotic functions and regeneration in the CNS.6
The activation of N-methyl-D-aspartate (NMDA) or AMPA-kainate receptors has been shown to cause the degeneration of RGCs.7, 8 The NMDA receptor antagonist MK-801 attenuates the loss of RGCs by NMDA excitotoxicity.9 Furthermore, MK-801 and the AMPA receptor antagonist DNQX have been shown to protect RGCs from apoptosis after optic nerve injury.10, 11, 12, 13 These findings indicate that glutamate-dependent activation of NMDA and AMPA-kainate receptors is an important trigger of apoptotic cell death in injured adult RGCs.
Glutamate is removed from the extracellular space and transported into glial and neuronal cells by several classes of excitatory amino-acid transporters, including GLAST-1 and GLT-1. In the mammalian retina, GLAST-1 is localized to Müller glia and nerve fiber layer astrocytes.14, 15, 16 GLAST-1 inhibition or deficiency has been shown to increase glutamate levels in the eye and promote RGC degeneration.17 GLT-1 has been localized to cone photoreceptors and cone bipolar cells in the mammalian retina.18 GLT-1-deficient mice show increased neuronal damage after retinal ischemia,19 suggesting that GLT-1 plays an important role in CNS neuronal apoptosis.
Only 13–14% of RGCs express Ret,20 the signal transducing kinase receptor for GDNF and NTN. Furthermore, GDNF and BDNF responsive cells are not separate populations, and the majority of RGCs express the BDNF receptor TrkB.21 It is therefore surprising that GDNF rescues more than 14% of RGCs after axotomy and has additive effects with BDNF.4 RGCs are known to be directly responsive to BDNF,22 and therefore GDNF may prevent apoptosis by additional indirect mechanisms. One such mechanism may be the upregulation of glutamate transporters in surrounding glial cells, thereby enhancing extracellular glutamate uptake and reducing its excitotoxic effects on injured RGCs. In the present study, we examined the intracellular pathways involved in GDNF and NTN neuroprotection and determined whether the modulation of GLAST-1 is neuroprotective.
Results
GDNF and NTN modulate GLAST-1 levels in vitro
We first examined the intracellular pathways that upregulate GLAST-1 expression in response to GDNF or NTN treatment. Retinal Müller glia express the glutamate transporter GLAST-1,14, 15, 16 the glutamate processing enzyme glutamine synthetase (GS),14 and GDNF/NTN receptors.4, 5 We initially examined the regulation of GLAST-1 and GS in the rMC-1 Müller cell line because the culture of primary Müller cells leads to phenotypic changes that include the loss of vimentin, glial-fibrillary acidic protein (GFAP), and GS expression within a few days in culture.23, 24
rMC-1 cells were not immunoreactive for the neuronal marker, protein gene product 9.5 (PGP9.5, Figure 1b). GLAST-1 immunoreactivity (Figure 1c) in rMC-1 cells colocalized with vimentin (Figure 1d), an intermediate filament that is expressed by Müller glia. rMC-1 cells also expressed GFRα-1 (Figure 1e) and GFRα-2 (Figure 1f), the high affinity receptors for GDNF and NTN, respectively. We did not observe any decreases in GLAST-1, GS, vimentin, GFAP, GFRα-1, or GFRα-2 protein levels over the course of several months of passaging rMC-1 cells.
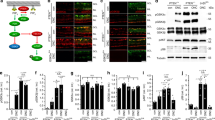
Cultured rMC-1 Müller cells are responsive to GDNF and Neurturin. (a–f) Confocal micrographs of immunofluorescently stained rMC-1 cells. (a) Negative control with primary antibody omitted. (b) Additional control demonstrating that rMC-1 cells were not immunoreactive for PGP9.5 and neuronal marker in the CNS. (c) The glutamate transporter GLAST-1 was colocalized with the intermediate filament vimentin (d), a characteristic marker of Müller glia. rMC cells also expressed GFRα-1 (e) and GFRα-2 (f), the high affinity receptors for GDNF and NTN, respectively. (g, h) Increased phosphorylation of Akt, MAPK, and Src in rMC-1 cells following treatment with GDNF (g) or NTN (h). Western blots directed against phosphorylated (p) or total Akt, MAPK, or Src at different time points after administration of 200 ng/ml of GDNF or NTN. Either growth factor caused a rapid increase in the phosphorylation of existing pools of Akt, MAPK, and Src, without affecting total protein levels. (i, j) Specific pharmacological inhibitors blocked phosphorylation of intracellular substrates. Western blots showing relative levels of phosphorylated (p) and total Akt, MAPK, or Src. Combined administration of GDNF (g) or NTN (h) with specific PI3K inhibitor LY294002 (LY) reduced Akt phosphorylation. The specific MEK inhibitor PD98059 (PD) reduced MAPK phosphorylation, whereas the Src inhibitor PP2 reduced the phosphorylation of Src. Scale bar (a–f)=50 μm
Next, we determined whether neurotrophic factor treatment could induce phosphorylation of intracellular kinases that are known to be involved in GDNF/NTN signal transduction. PI3K, MAPK, and Src are activated by phosphorylation at key residues, a feature that can be assessed in western blots. Although it is difficult to assess PI3K activity directly, the PI3K-dependent phosphorylation of Akt (protein kinase B (PKB)) at Serine 473 is indicative of PI3K activity and is required for Akt activation. Similarly, the MEK-dependent phosphorylation of MAPK at Threonine 202/Tyrosine 204 is required for MAPK activation. Lastly, Src phosphorylation at Tyrosine 416 is a hallmark of enzyme activation. We used phospho-specific antisera to detect the activation of Akt (Ser 473), MAPK (Thr 202/Tyr 204), and Src (Tyr 416) in western blots, to assay for PI3K, MAPK, and Src activation following GDNF or NTN administration. Both GDNF and NTN treatment increased the phosphorylation of Akt, MAPK, and Src in rMC-1 cells (Figure 1g and h). Interestingly, NTN appeared to induce a more prolonged increase in Akt and MAPK phosphorylation compared to GDNF. The levels of total Akt, MAPK, or Src were not altered by GDNF or NTN, demonstrating that increases in phospho-Akt, -MAPK, and -Src were due to phosphorylation of existing intracellular pools (Figure 1g and h). We next examined the effects of a PI3K inhibitor (LY294002; 20 μM),25 a MEK inhibitor (PD98059; 20 μM),26 and a Src inhibitor (PP2; 2 μM)27 on the phosphorylation of Akt, MAPK, and Src (see Materials and Methods for inhibitor specificity). LY294002 reduced the phosphorylation of Akt, which is known to be activated by PI3K. PD98059 reduced phosphorylation of MAPK, the downstream target of MEK, and PP2 reduced Src phosphorylation in rMC-1 cells (Figure 1i and j). Again, total Akt, MAPK, and Src levels remained constant, indicating that the phosphorylation of existing intracellular pools was being modulated.
We then looked at the effects of GDNF or NTN on GLAST-1 and GS levels in rMC-1 cells. The regulation of GLAST-1 and GS by GDNF or NTN was examined at 12 and 24 h after administration. GLAST-1 levels in rMC-1 cells treated with GDNF or NTN increased within 12 h (Figure 2a). This was noticeably different from that of BDNF treatment. A time course showed that GLAST-1 upregulation occurred between 4 and 12 h after GDNF or NTN treatment (Figure 2b and c). In contrast to GLAST-1, GS levels were not altered by either GDNF or NTN (Figure 2b and c). Therefore, in addition to GDNF, NTN treatment increases GLAST-1 levels in a Müller cell line, independent of GS. Combined administration of neurotrophic factors further increased GLAST-1 expression in an additive manner (Figure 2d).
GDNF and NTN increase GLAST-1 levels in rMC-1 Müller cells. Western blots showing relative levels of GLAST-1 or GS in lysates of treated rMC-1 cells. (a) Both GDNF and NTN increased GLAST-1 levels within 12 h of administration. The effects of BDNF are shown for comparison. (b, c) Time courses of GLAST-1 upregulation after GDNF (b) or NTN (c) treatment. Neither factor affected GS levels in rMC-1 cells. (d) Combined administration of neurotrophic factors had enhanced effects on GLAST-1 expression, as shown in the quantification below. Asterisks (*) denote P<0.01 between selected groups and BDNF, GDNF, or NTN alone
The enhanced effects of combined growth factor treatments on GLAST-1 levels suggest that these factors act through independent mechanisms. Therefore, we were interested in determining whether this effect was dependent on different intracellular signaling cascades. Control administration of the PI3K inhibitor (LY294002), the MEK inhibitor (PD98059), or the Src inhibitor (PP2) in the absence of neurotrophic factors did not affect GLAST-1 levels at 24 h after treatment (Figure 3a). Next, we tested whether these inhibitors could block the GDNF- or NTN-dependent signal transduction events that upregulate GLAST-1. LY294002 or PP2 treatment abolished the GDNF-dependent upregulation of GLAST-1 at 24 h (Figure 3b) or 48 h (Figure 3c); however, only LY294002 inhibited GLAST-1 upregulation by NTN (Figure 3d and e). Our findings point to a common involvement of PI3K in GLAST-1 upregulation by GDNF and NTN, with the additional involvement of Src in GDNF-dependent processes.
Western blots showing GLAST-1 levels at 24 or 48 h after various treatments. (a) Control administration of PD98059 (PD), LY294002 (LY), or PP2 did not have any effect on GLAST-1 levels at 24 h after treatment. (b, c) GDNF upregulated GLAST-1 levels at 24 h (b) and 48 h (c), and this effect was abolished by the specific PI3K inhibitor LY294002 (LY) and the Src inhibitor PP2. Averaged densitometry values for three separate experiments are shown on the right. Asterisks (*) denote P<0.01 between selected groups and GDNF treatment alone. (d, e) NTN also upregulated GLAST-1 levels at 24 h (d) and 48 h (e) after treatment. The effect of NTN was antagonized by combined administration with the PI3K inhibitor LY294002 (LY). Averaged densitometry values are for three separate experiments and are shown on the right. Asterisks (*) denote P<0.05 between selected groups and NTN treatment alone. Combined administration of the MEK inhibitor PD98059 (PD) with GDNF or NTN had no effect on GLAST-1 levels, suggesting that MAPK signaling is not involved in these processes
Differential regulation of GLAST-1 and GLT-1 by GDNF and NTN
In order to expand on our in vitro findings, we examined the role that GDNF and NTN play in the regulation of GLAST-1 in vivo. In the mammalian retina, GLAST-1 is localized to nerve fiber layer astrocytes in addition to Müller glia.14, 15, 16 GLAST-1 western blots showed several bands at approximately 66 kDa as described previously.28, 29, 30 These bands represent different glycosylated forms of GLAST-128, 29, 30 and have been shown to have no functional significance on transporter activity.29 At 5 days after intraocular GDNF administration, retinas had approximately twofold higher GLAST-1 levels compared to controls (Figure 4). This effect was antagonized by LY294002 and PP2 (Figure 4), indicating that the upregulation of GLAST-1 by GDNF in vivo is dependent on PI3K and Src activity. Intraocular delivery of NTN also enhanced GLAST-1 expression; however, none of the inhibitors used in the present study were effective in abolishing this response (Figure 4). Control injection of LY294002, PD98059, or PP2 had no effect of GLAST-1 levels at 5 days after intraocular injection (Figure 4), suggesting that the effects of kinase inhibition were specific to signal transduction by the coadministered neurotrophic factors. These results suggest that GDNF increases GLAST-1 expression through PI3K and Src activation, whereas the effects of NTN in vivo are not dependent on a single intracellular pathway.
Effects of kinase inhibitors on GDNF or NTN dependent increases in GLAST-1 levels in vivo. (Above) Control GLAST-1 western blot at 5 days after treatment with the PI3K inhibitor (LY294002), the MEK inhibitor (PD98059), or the Src inhibitor (PP2) showing no effect of inhibitors alone on GLAST-1 levels. (Below) GLAST-1 western blots and corresponding densitometry measurements of four separate experiments showing relative GLAST-1 levels in retinal homogenates after various treatments. The results expressed relative to the normal retina standard (±S.E.M.), which was set as a value of 1. GDNF and NTN significantly increased GLAST-1 levels in the retina at 5 days after the initial intraocular injection. Control (vehicle-treated retinas) had similar levels of GLAST-1 to normal untreated retinas. Both LY294002 (LY) and PP2 significantly reduced the effects of GDNF, which however did not affect GLAST-1 levels after NTN delivery. Asterisks (*) denote P<0.01 between selected groups and GDNF treatment
The glutamate transporter GLT-1 is expressed by cone bipolar cells in the mammalian retina.18 As these cells synapse on RGCs and participate in glutamate reuptake, we studied the effects of GDNF and NTN on the expression of GLT-1 in vivo. GLT-1 western blots showed large bands at approximately 70 kDa.31, 32 Similar to GLAST-1, GLT-1 is differentially glycosylated, having no apparent functional significance.31 GDNF increased GLT-1 levels by approximately threefold compared to controls (Figure 5), an effect that was inhibited by coadministration of LY294002 or PP2 (Figure 5). NTN, however, did not significantly alter the levels of GLT-1 in vivo (Figure 5). Control intraocular delivery of pharmacological inhibitors alone did not affect GLT-1 levels in the retina (Figure 5). Thus, GDNF but not NTN increases the levels of the glutamate transporter GLT-1 in the retina, dependent on activation of PI3K and Src.
Effects of GDNF or NTN on GLT-1 levels in vivo. (Above) Control GLT-1 western blot at 5 days after treatment with the PI3K inhibitor (LY294002), the MEK inhibitor (PD98059), or the Src inhibitor (PP2). Inhibitors alone did not affect GLT-1 levels. (Below) GLT-1 western blots and corresponding densitometry measurements of four separate experiments (±S.E.M.) showing relative GLT-1 levels in retinal homogenates after various treatments. GDNF but not NTN significantly increased GLT-1 levels after 5 days. LY294002 (LY) and PP2 significantly reduced the effects of GDNF, demonstrating a dependence on the PI3K and Src kinase pathways, respectively. Asterisks (*) denote P<0.01 between selected groups and GDNF treatment
GS is localized to retinal Müller glia and nerve fiber layer astrocytes14 that convert glutamate to glutamine. Previous findings suggest that GLAST-1 and GS are co-regulated;33 however, we found that neither GDNF nor NTN altered the levels of GS after intraocular administration (Figure 6), showing that GS may be regulated independently of GLAST-1 in the adult mammalian retina. Control delivery of pharmacological inhibitors alone did not affect GS levels at 5 days.
Western immunoblotting for GS after intraocular injection of GDNF or NTN. (Above) Control experiments showing that the MEK inhibitor PD98059, the PI3K inhibitor LY294002 (LY), and the Src inhibitor PP2 did not affect GS levels at 5 days after intraocular injection. (Below) Neither GDNF nor NTN had any significant effect on GS levels in the retina when administered alone or in combination with LY294002 (LY), PD98059 (PD), or PP2
Intracellular pathways involved in GDNF or NTN neuroprotection
It is likely that the additive RGC survival observed following combined administration of GDNF and NTN in vivo6 is a function of independent intracellular signaling by these two factors. In order to test this hypothesis, we examined the role that the PI3K, MAPK, and Src kinase pathways play in GDNF and NTN neuroprotection after optic nerve transection.
Both GDNF and NTN induced phosphorylation of Akt/PKB, which is activated downstream of PI3K. This effect was specifically blocked by the PI3K inhibitor LY294002 and was not affected by the MEK inhibitor PD98059 or the Src inhibitor PP2 (Figure 7). MAPK phosphorylation was selectively inhibited by PD98059 and was not affected by LY294002 or PP2 (Figure 7). The Src kinase inhibitor PP2 reduced Src phosphorylation after intraocular GDNF of NTN delivery, and LY294002 had no effect (Figure 7). MAPK pathway inhibition by the MEK inhibitor PD98059 also reduced Src phosphorylation after GDNF of NTN treatment (Figure 7). The downstream activation of Src by MAPK and cross-talk between these pathways has been shown elsewhere.34, 35 There were no changes in total Akt, MAPK, or Src levels at 4 h after trophic factor delivery (Figure 7), indicating that the increased levels of phosphorylated Akt, MAPK, and Src were due to activation of existing intracellular pools. These findings show that at the concentrations used in our in vivo studies, LY294002, PD98059, and PP2 were selective for their specific intracellular kinases as described previously.25, 26, 27
Western immunoblots showing the phosphorylation of Akt, MAPK, and Src in retinal homogenates after intraocular injection of GDNF or NTN and pharmacological inhibitors. Phospho-specific antibodies used in these studies detect phosphorylation at key residues that are known to be indicative of kinase activation. Samples were processed 4 h after intraocular injections. Both GDNF and NTN increased the phosphorylation (p) of Akt, MAPK, and Src after intraocular injection. Combined administration of the PI3K inhibitor LY294002 (LY) with GDNF of NTN specifically inhibited phosphorylation of the downstream target Akt without affecting MAPK or Src phosphorylation. The MEK inhibitor PD98059 (PD) specifically inhibited the phosphorylation of MAPK (arrowheads) without affecting Akt phosphorylation. The Src specific inhibitor PP2 reduced Src phosphorylation (arrowheads) without affecting Akt or MAPK phosphorylation. Src phosphorylation was also reduced by the MEK inhibitor PD98059, most likely an effect of cross-talk between the MAPK and Src cascades, which has been reported previously in many cell types. Total Akt, MAPK, and Src were not affected by treatments, demonstrating that phosphorylation events occurred in existing intracellular pools
We then used these selective PI3K, MAPK, and Src pathway inhibitors to uncover the intracellular cascades required for GDNF or NTN neuroprotection in the adult retina. The effects of PI3K, MAPK, and Src inhibitors on RGC survival were quantified at 14 days postaxotomy, the time point at which approximately 90% of RGCs have undergone apoptosis. Similar to previous findings,2, 3, 4 GDNF administration increased RGC survival by approximately two- to threefold over that of control vehicle injections (Figure 8c, Figure 9). Pharmacological inhibition of the Src kinase pathway (PP2), the PI3K pathway (LY294002), or MAPK activation (PD98059) significantly diminished the neuroprotective effects of GDNF by differing degrees (Figure 8, Figure 9). Administration of either PP2, LY294002, or PD98059 alone did not affect RGC survival in comparison to control vehicle treatment, indicating that these effects were specific to blockade of neurotrophic factor-dependent signal transduction (Figure 9). These results indicate that the GDNF-mediated rescue of axotomized RGCs is dependent on multiple intracellular pathways.
Epifluorescence micrographs of flatmounted retinas showing fluorogold labeled RGCs at 14 days after optic nerve transection. (a) Retrogradely labeled RGCs at 1 day after axotomy. No significant RGC death is observed at this time point, and fascicles of labeled RGC axons are visible. (b) At 14 days after optic nerve transection, the majority of RGCs have degenerated and a few remaining RGCs (arrows), as well as transcellularly labeled microglia (arrowheads), are apparent. (c) GDNF administration enhanced the survival of RGCs. (d) This survival promoting effect was diminished by co-administration of the PI3K pathway inhibitor LY294002 (‘LY’). Similar to GDNF, the neuroprotective effects of NTN (e) were abolished by PI3K inhibition (f). Scale bar=50 μm
Mean RGC densities (±S.E.M.) at 14 days after axotomy. Samples were taken at different retinal eccentricities (inner, middle, outer). Intraocular administration of PP2 (Src inhibitor), LY294002 (‘LY’; PI3K inhibitor), or PD98059 (‘PD’; MAPK inhibitor) had no significant effect on cell survival compared to control vehicle treatment. The administration of GDNF or NTN resulted in an approximate two- to threefold increase in RGC survival after 14 days. Combined injection PP2, LY294002, or PD98059 with GDNF significantly reduced cell survival compared to retinas that received GDNF alone. Co-treatment with LY294002 significantly antagonized the effects of NTN, whereas PP2 had no effect. Intraocular administration of PD98059 had no significant effect on NTN-dependent cell survival with the exception of samples taken in the outer retina (&hashP<0.05). Asterisks (*) denote a significant difference from the corresponding GDNF or NTN treatment (P<0.001)
In agreement with previous findings,6 NTN had similar neuroprotective effects to GDNF at 14 days postaxotomy (Figure 8e, Figure 9). Unlike GDNF, however, RGC survival after NTN treatment was only reduced by the PI3K inhibitor LY294002 (Figure 9) and was not affected by PP2 or PD98059. These findings show that the survival promoting effects of NTN are dependent on PI3K signal transduction and, unlike GDNF, appear to be independent of MAPK and Src activation.
Involvement of GLAST-1 in GDNF and NTN neuroprotection
Given that the neuroprotective effects of GDNF and NTN on axotomized RGCs were dependent on similar intracellular pathways to those required for the regulation of GLAST-1, we were unable to determine if GLAST-1 upregulation plays a neuroprotective role based on differences in signal transduction alone. In order to address this issue, we used RNA interference to prevent the increase in GLAST-1 levels after GDNF or NTN delivery to the retina and also examined the effect on RGC survival after axotomy.
Intraocular injection of short interfering RNAs (siRNAs) directed against GLAST-1 antagonized the upregulation of GLAST-1 by GDNF or NTN (Figure 10). A control siRNA directed against firefly luciferase showed no effect. Coadministration of GLAST-1 siRNAs resulted in significantly lower RGC densities at 14 days postaxotomy, in comparison to treatments of GDNF or NTN alone or together with the control siRNA (Figure 10). The reduction in RGC survival was most apparent in the outer retina, where RGC densities were reduced to near control levels at 14 days.
GLAST-1 upregulation plays a role in the rescue of axotomized RGCs by GDNF and NTN. Two different siRNAs directed against GLAST-1 diminished the upregulation of GLAST-1 in vivo by GDNF or NTN, as shown by western blot, at 5 days postaxotomy. A control siRNA directed against firefly luciferase (Luc siRNA) had no effect. RGC densities (±S.E.M.) at 14 days postaxotomy were significantly lower (*P<0.001) in comparison to GDNF or NTN treatment when GLAST-1 siRNA was coadministered. RGC densities in the outer retina were most greatly affected by GLAST-1 siRNA delivery
Discussion
We have described in detail, the intracellular signal transduction pathways that regulate GLAST-1 levels, GLT-1 levels, and RGC survival in the mammalian retina, after GDNF or NTN treatment. In addition, we have shown that the upregulation of GLAST-1 by GDNF or NTN contributes to the rescue of axotomized RGCs undergoing apoptosis.
Upregulation of GLAST-1 and GLT-1 by GDNF and neurturin
In the present study, we show that GLAST-1 levels in rMC-1 cells are increased by GDNF or NTN. This process is highly dependent on PI3K activation, with additional modulation by Src in the case of GDNF. Our findings also translated to the mammalian retina where both GDNF and NTN enhanced GLAST-1 expression. Although the importance of PI3K and Src signaling in vivo mirrored that of GDNF in rMC-1 cells, no single inhibitor was effective in abolishing the upregulation of GLAST-1 by NTN in vivo. A number of factors may account for this discrepancy. In the retina, nerve fiber layer astrocytes in addition to Müller cells express GLAST-114, 15, 16 and may express GFRα-2.5 It is possible that GLAST-1 expression in astrocytes is regulated in a different manner to that in Müller glia, confounding the effects of NTN administration. Alternatively, NTN may induce the secretion of additional factors that alter GLAST-1 levels in the mammalian retina, where redundant signaling pathways may exist. Overall, our findings show that both GDNF and NTN increase GLAST-1 expression and produce enhanced effects when administered together, possibly through additional GDNF-dependent Src activity.
GLT-1 expressing bipolar cells have previously been shown to express Ret,5 suggesting that they are an alternate neuronal target for GDNF in the mammalian retina. Of interest is the present finding that GDNF but not NTN increases GLT-1 levels in the retina through PI3K and Src activation. It is unclear whether this contributes to RGC rescue or to enhanced effects observed after combined delivery of these two factors.6 This question is difficult to answer because the location of bipolar cell bodies in the inner nuclear layer makes it difficult to transfect these cells with intraocular siRNA treatments. Our attempts to knock down GLT-1 were unsuccessful. However, a recent study has demonstrated that the upregulation of GLT-1 by β-lactam antibiotics is neuroprotective,36 suggesting that the upregulation of GLT-1 by GDNF may contribute to the rescue of injured RGCs.
Src is critical for GDNF activity in vivo
The present study points to a key role for Src in the regulation of GLAST-1 and GLT-1 levels by GDNF. In addition, the neuroprotective effects of GDNF in adult mammalian RGCs were Src dependent, demonstrating that Src is involved in GDNF neuroprotection in vivo. Unlike GDNF, the modulation of GLAST-1 by NTN was not affected by Src inhibition. Furthermore, the neuroprotective effects of NTN in vivo did not require Src activity. Thus, in the injured mammalian CNS, GDNF- but not NTN-mediated neuronal survival may be dependent on Src kinase pathways. Src signaling events may therefore be the factor that accounts for the additive cell rescue observed when GDNF is delivered in combination with NTN.4
Indirect neuroprotective effects of GDNF and NTN in the adult CNS
In the present study, we show that the MAPK pathway was required for axotomized RGC survival after GDNF treatment. Nicole et al.37 reported that GDNF reduces the conductance of the NMDA receptor through activation of the MAPK pathway, protecting cultured cells against glutamate excitotoxicity. This may be an additional MAPK-dependent survival mechanism of GDNF after optic nerve transection. Alternatively, our results suggest that Src activation, which is required for GDNF-mediated rescue of axotomized RGCs, is reduced by MAPK inhibition. Src may therefore be activated via MAPK-dependent signal transduction in the adult retina, accounting for the loss of neuroprotection after PD98059 delivery with GDNF.
The importance of GLAST-1 in maintaining normal retinal function and preventing the degeneration of RGCs has been established previously.38 However, it was previously unclear whether the upregulation of GLAST-1 by growth factors plays a physiological role in neuroprotection after CNS injury. In order to parse out the possible antiapoptotic effects of GLAST-1 upregulation, we used siRNAs in vivo to prevent the increases in GLAST-1 expression observed after GDNF or NTN delivery. These experiments demonstrate that the upregulation of GLAST-1 contributes to the survival promoting effects of both GDNF and NTN. Our findings also show that RGC survival in the outer retina was most dependent on GLAST-1 modulation by neurotrophic factors. This may be a consequence of the higher susceptibility of larger RGCs to glutamate excitotoxicity.7 The density of larger α-RGCs is known to increase toward the outer or peripheral retina,39 where we observed the most pronounced decrease in RGC density after GLAST-1 siRNA co-treatment.
In conclusion, the PI3K pathway is a key component in GLAST-1 upregulation and CNS neuroprotection by GDNF and NTN. Furthermore, the involvement of Src kinase and MAPK kinase signaling in GDNF neuroprotection, together with the additional upregulation of GLT-1, appear to distinguish the biological effects of GDNF from NTN in vivo. These key differences may be the biological basis for the enhanced neuroprotective effects observed after the combined delivery of both of these neurotrophic factors or for the additive effects observed after co-delivery of GDNF or NTN with BDNF in vivo. The present findings also implicate the upregulation of GLAST-1 as a key component of GDNF and NTN neuroprotection in the injured adult CNS. This mechanism accounts for the greater rate of RGC neuroprotection in relation to receptor expression in vivo.
Materials and Methods
Animals
Adult female Sprague–Dawley rats (225–250 g; Charles River) that were housed in a Level B pathogen-free environment were used for all experiments. Experimental procedures were carried out according to the guidelines of the Canadian Council on Animal Care and the German guidelines for animal care, supervised by the local authorities in Braunschweig.
Optic nerve transection and intraocular injections
The left optic nerve of each rat was completely transected within the orbit of the eye as described previously.3, 4 During surgery, animals were anesthetized with intraperitoneal injections of 7% chloral hydrate (420 mg/kg). The superior orbital contents were resected and the eye was rotated temporally by applying traction to the extraocular muscles. Care was taken not to damage the lacrimal gland during surgery. The meninges surrounding the optic nerve were cut longitudinally to avoid damaging the retinal vessels, and the optic nerve was exposed. The optic nerve was gently lifted out of the meningeal sheath and transected within 1.5 mm of the back of the eye. Immediately after transecting the nerve, a small piece of gelfoam soaked in 3% Fluorogold was applied to the end of the optic nerve stump to retrogradely label RGCs.
Following the completion of the surgical procedure, animals received an intraocular injection of an experimental or control solution. A pulled glass micropipette was used to deliver up to 4 μl of the desired solution into the vitreous chamber of the eye. Injections were made posterior to the limbus of the eye to ensure that the anterior ocular structures were not damaged. Care was also taken not to damage the lens.
Control injections consisted of 4 μl of 50 : 50 PBS (in which growth factors were dissolved) and DMSO (in which pharmacological inhibitors were dissolved). Experimental injections used in the present study were as follows: a 2 μl solution containing 0.5 μg of GDNF, or a 2 μl solution containing 0.5 μg of NTN, or 2 μl of 20 mM PD98059 (MAPK pathway inhibitor), or 2 μl of 20 mM LY294002 (PI3K pathway inhibitor), or 2 μl of 20 mM PP2 (Src kinase pathway inhibitor), or a combination of a growth factor and a pharmacological inhibitor (2 μl of each). See below for the specificity of these inhibitors. For assaying RGC survival at 14 days postaxotomy, the animals received two intraocular injections during the course of RGC degeneration: an initial injection at day 0 and a second injection at day 5 postaxotomy, the time point when RGCs begin to undergo delayed apoptosis after axotomy.
In order to determine which intracellular pathways are required for GDNF- or NTN-mediated regulation of GLAST-1, GLT-1, or GS in vivo, the animals received intraocular injections of GDNF or NTN alone or in combination with pathway inhibitors at days 0 and 3. The retinas were then extracted at 5 days after the initial intraocular injection and processed for western immunoblotting directed against GLAST-1, GLT-1, and GS. Additional retinas were used to determine if the pharmacological inhibitors used in this study decreased the phosphorylation (activation) of specific intracellular messengers that are activated by GDNF or NTN. The animals received intraocular injections of GDNF or NTN alone or in combination with pathway inhibitors, and the retinas were extracted at 4 h after injection and processed for immunoblotting directed against phosphorylated isoforms of MAPK, Akt/PKB (a downstream messenger of the PI3K cascade), and Src.
In order to determine if GLAST-1 upregulation plays a role in the rescue of RGCs by GDNF or NTN, we administered combined intraocular injections of GDNF or NTN and siRNAs directed against GLAST-1. RGC survival was quantified at 14 days postaxotomy. GLAST-1 siRNAs were generated using the Silencer siRNA construction kit (Ambion, Austin, TX, USA) according to the manufacturer's protocol. Sequences used in the present study were: GLAST-1(123) 5′-AGAGGAGAUUUCGCUUUCU-3′, GLAST-1(176) 5′-AAAGCCGUAUCAGCUGAUU-3′. The animals received intraocular injections containing 1 μg of GDNF or NTN (dissolved in 2 μl of sterile PBS) and 2 μl of an siRNA solution containing 2 μg of GLAST-1 siRNA complexed in Lipofectamine/Opti-MEM (Invitrogen, Burlington, ON, Canada). Control injections had Firefly Luciferase siRNA (5′-CGUACGCGGAAUACUUCGA-3′) substituted for GLAST-1 siRNA. A separate group of animals that received identical treatments was used for western blotting to determine if GLAST-1 levels were reduced by intraocular GLAST-1 siRNA delivery. For these experiments, retinal lysates were processed at 5 days postaxotomy, the time point when RGCs begin to die.
Tissue preparation and RGC survival assessment
In order to assess RGC survival in different treatment groups, animals were killed using an overdose of anesthetic (7% chloral hydrate) at 14 days after optic nerve transection. Their eyes were enucleated, the cornea and lens were removed, and the remaining eye cups were fixed in a solution of 4% paraformaldehyde in PBS containing 2% sucrose for 1.5 h at room temperature. Eye cups were then rinsed 15 min in PBS, and the neural retina was dissected away from the sclera. Retinas were flatmounted in 50 : 50% glycerol : PBS. Surviving RGCs labeled with Fluorogold were quantified using a Zeiss Axiovert epifluorescence microscope. RGC densities were determined within a grid of 85 000 mm2 size, using a × 20 objective lens, and expressed as the number of RGCs per square millimeter (±S.E.M.). RGC densities were sampled from each quadrant of the flatmounted retina. Within each quadrant, three samples were taken at different retinal eccentricities (inner, mid-periphery, outer). Thus, a total of 12 samples from each retina were examined. Samples were grouped by retinal eccentricity and statistically significant differences between groups were detected using Tukey's post hoc test, following an analysis of variance at P<0.05.
Cell culture
rMC-1 cells were cultured in DMEM/F12 containing 1% glutamine and 0.5% fetal calf serum. Cells were passaged at 5-day intervals, when they had reached near confluence. For determining the effects of growth factors on the phosphorylation of intracellular messengers, or the expression of GLAST-1 or GS, rMC-1 cells were seeded in six-well plates, at a density of 100 000 cells/well. Low cell densities were used in order to minimize the effects of endogenous growth factors produced by the cells themselves.
In vitro experiments
Prior to performing in vitro experiments, rMC-1 cells were plated and cultured for 24 h in normal medium and then serum deprived for 24 h. Cells were then cultured for 5 days in the presence of 100 ng/ml of succinyl-Concanavalin-A to promote differentiation. Our preliminary experiments demonstrated that cell division in rMC-1 cells could be inhibited for approximately 5–7 days under these conditions (not shown).
rMC-1 cells were rinsed 2 × with serum-free DMEM/F12, serum deprived for 6 h, and then treated with 200 ng/ml of GDNF or NTN in DMEM/F12 containing 0.5% fetal calf serum and 1% glutamine. At different time points after trophic factor administration, the cells were rinsed with PBS and lysates were made for western blotting. GDNF or NTN-treated rMC-1 cell lysates were extracted at 1, 2, 4, 12, or 24 h after the addition of trophic factors to the media. Additional groups of rMC-1 cells were treated with 200 ng/ml of GDNF or NTN alone or in combination with 20 μM PD98059 or 20 μM LY294002 or 2 μM PP2, and cell lysates were prepared at 24 or 48 h after treatment. These samples were used to assess the effects of pharmacological inhibitors on GDNF- or NTN-mediated alterations in GLAST-1 or GS levels in rMC-1 cells. In order to verify the effectiveness of pharmacological inhibitors, rMC-1 cells were treated with GDNF or NTN alone or in combination with one of the three inhibitors used in this study, and cell lysates for western blotting against phosphorylated substrates were made at 2 h after treatment.
Immunofluorescent staining of rMC-1 cells
rMC-1 cells were cultured on glass coverslips in 12 well plates. Coverslips were rinsed in PBS, fixed in 3 : 1 methanol : acetone for 30 s, rinsed in PBS, and then rinsed in distilled water. Immunocytochemistry was then performed on the fixed cells. Coverslips were placed in 12-well plates and immersed in primary antibody solutions overnight at 4°C. Primary antibodies consisted of guinea-pig anti-GLAST-1 (1 : 500, Chemicon, Temecula, CA, USA), rabbit-anti-GFRα-1 (1 : 500; Transduction Laboratories, Lexington, KY, USA), rabbit-anti-GFRα-2 (1 : 1000; Santa Cruz Biotechnology, Santa Cruz, CA, USA), mouse-anti-PGP9.5 (1 : 400; Ultraclone, UK), and goat-anti-Vimentin (1 : 200; Sigma, Oakville, ON, Canada). Primary antisera were diluted in PBS+3% normal serum. Secondary antisera (affinity purified FITC or Texas Red-conjugated goat-anti-rabbit, goat-anti-mouse, donkey-anti-goat, donkey-anti-guinea-pig; 1 : 500; Jackson Immunoresearch, West Grove, PA, USA) were diluted in PBS+3% normal serum. Following primary antibody incubation, coverslips were rinsed 3 × 15 min in PBS and incubated with the secondary antibody solution for 2 h. The coverslips were then rinsed 3 × 15 min and mounted using Vectashield mounting medium (Vector Labs, Burlingame, CA, USA). Immunostained cells were then imaged using a Zeiss laser scanning confocal microscope (LSM 510) and single or double label images were captured.
Specificity of pharmacological kinase inhibitors
The specificity of the pharmacological inhibitors used in this study has been demonstrated previously.25, 26, 27 These inhibitors have been widely used to study signal transduction in vitro and in vivo. LY294002 is a specific inhibitor of PI3K with an IC50 of 1.4 μM.25 LY294002 does not have any inhibitory effects on other protein kinases, including protein tyrosine kinases (Src kinase, EGF receptor), serine/threonine kinases (MAP kinase, PKC, PKA, S6 kinase), lipid kinases (PI4K, DAG kinase), or kidney ATPase.25 PD98059 is a specific inhibitor of the activation of MAPK.26 PD98059 inhibits MEK1(MAPKK1), the upstream kinase that activates MAPK, with an IC50 of 4 μM.26 PD98059 is a unique inhibitor because it does not inhibit activated MEK1 but rather prevents any additional activation by interacting with inactive MEK1. PD98059 was shown to have no inhibitory effect on 18 other serine/threonine kinases, including MAPK, JNK, PKA, PKC, Cdk2, phosphorylase kinase, and S6 kinase.26 PP2 is a tyrosine kinase inhibitor that is selective for Src.27 PP2 inhibits Src with an IC50 of 1–5 μM27, 40 and has a greater than 100-fold selectivity for Src family kinases over other tyrosine kinases, including ZAP-70, JAK-2, and the EGF receptor.
Western blotting
Following experimental manipulations, rMC-1 cells, cultured in six-well plates, were rinsed 2 × with PBS. Cells were then lysed in 100 μl of SDS lysis buffer containing 2% SDS, 0.3% DTT, 10% glycerol in 40 mM Tris-Cl (pH 6.8), sodium orthovanadate (1 mM), sodium pyrophosphate (2.5 mM), PMSF (1 mM), and a protease inhibitor cocktail (P2714; Sigma). Cell lysates were sonicated, heated to 90°C for 8 min, centrifuged (13 000 r.p.m., 12 min, 4°C), and the remaining protein samples were frozen at −20°C.
For in vivo experiments, animals were killed, and the retinas were quickly extracted in ice-cold PBS. Each retina was placed in 200 μl of warmed SDS lysis buffer. The retinas were then sonicated and the remaining solutions were heated to 90°C for 8 min, centrifuged (13 000 r.p.m., 12 min, 4°C), and the protein samples were separated from the pelleted debris. Protein samples were then frozen at −20°C for future use.
The protein concentration of each of the samples was assayed with the BCA assay kit (Pierce Biotechnology Inc., Rockford, IL, USA), according to the manufacturer's protocol. Total protein fractions were then separated by SDS-polyacrylamide gel electrophoresis (10% acrylamide) and immunoblotted after semidry electrotransfer to nitrocellulose membranes. Protein samples from experimental retinas were assayed together on the same gel along with a pooled retinal preparation (four normal retinas total) that was used as a standard. This standard was run in two separate lanes on every gel and the densitometry values for these two lanes were averaged. Loading was verified by visualizing protein bands with Ponceau S dye and reprobing selected blots with antisera directed against β-tubulin (mouse monoclonal; Sigma). All blots were blocked in 5% milk in TBS-T (Tris-buffered saline containing 0.1% Tween 20) for 1 h at room temperature. Blots were then incubated overnight at 4°C, with gentle shaking. All primary antibodies were dissolved in 1% milk in TBS-T. Primary antisera for examining the regulation of glutamate transporters or GS consisted of guinea-pig-anti-GLAST-1 (1 : 2000, Chemicon), guinea-pig-anti-GLT-1 (1 : 1000, Chemicon), or mouse-anti-GS (1 : 2000, Chemicon).
The PI3K-dependent phosphorylation of Akt (PKB) at Serine 473 is indicative of Akt activation. Similarly, the MEK-dependent phosphorylation of MAPK at Threonine 202/Tyrosine 204 is required for MAPK activation and phosphorylation of Src at Tyrosine 416 is necessary for enzyme activity. We used phospho-specific antisera to detect the activation of Akt (Ser 473), MAPK (Thr 202/Tyr 204), and Src (Tyr 416). Primary antisera for analyzing the effects of growth factors and pathway inhibitors on kinase phosphorylation consisted of the following: rabbit-anti-phospho-MAPK Thr202/Tyr204 (1 : 1000, Cell Signalling Technology), goat-anti-MAPK (Santa Cruz Biotechnology), rabbit-anti-phospho-Akt Ser473 (1 : 500, Cell Signalling Technology), rabbit-anti-Akt (1 : 1000, Cell Signalling Technology), rabbit-anti-phospho-src Tyr416 (1 : 250, Cell Signalling Technology), and rabbit-anti-src (1 : 500, Abcam, Cambridge, UK). The blots were washed for 3 × 15 min in TBS-T and incubated in a 1 : 2000 dilution of secondary antisera (horseradish peroxidase conjugated, crossreacted against rat serum antigens; Jackson Immunoresearch) dissolved in 5% milk in TBS-T. Afterward, the membranes were rinsed 3 × 15 min in TBS-T. Chemiluminescent immunoreactive complexes were visualized using a BioRad Fluor-S Max multi-imager system equipped with a 50 mm f1.4 Nikon lens. Protein levels were quantified by densitometry (BioQuant Image Analysis, Nashville, TN, USA). Densitometry values were normalized against the tubulin loading controls and then normalized against the value for the pooled normal retina standard run on each gel. GLAST-1, GLT-1, and GS western blots for in vitro and in vivo experiments were performed on three separate experiments so that a semiquantitative analysis could be performed. The results were expressed as mean densitometry measurements ±S.D. Statistically significant differences between different groups were detected using Tukey's post hoc test following an analysis of variance at P<0.05.
Abbreviations
- GDNF:
-
glial cell-line-derived neurotrophic factor
- NTN:
-
neurturin
- BDNF:
-
brain-derived neurotrophic factor
- GLAST-1:
-
L-glutamate/L-aspartate transporter
- GLT-1:
-
L-glutamate transporter
- GS:
-
glutamine synthetase
- PI3K:
-
phosphoinositide-3 kinase
- MAPK:
-
mitogen-activated protein kinase
- Src:
-
sarcoma
- RGC:
-
retinal ganglion cell
References
Koeberle PD, Bahr M . Growth and guidance cues for regenerating axons: where have they gone? J Neurobiol 2004; 59: 162–180.
Klocker N, Braunling F, Isenmann S, Bahr M . In vivo neurotrophic effects of GDNF on axotomized retinal ganglion cells. Neuroreport 1997; 8: 3439–3442.
Koeberle PD, Ball AK . Effects of GDNF on retinal ganglion cell survival following axotomy. Vision Res 1998; 38: 1505–1515.
Koeberle PD, Ball AK . Neurturin enhances the survival of axotomized retinal ganglion cells in vivo: combined effects with glial cell line-derived neurotrophic factor and brain-derived neurotrophic factor. Neuroscience 2002; 110: 555–567.
Jomary C, Thomas M, Grist J, Milbrandt J, Neal MJ, Jones SE . Expression patterns of neurturin and its receptor components in developing and degenerative mouse retina. Invest Ophthalmol Vis Sci 1999; 40: 568–574.
Airaksinen MS, Saarma M . The GDNF family: signalling, biological functions and therapeutic value. Nat Rev Neurosci 2002; 3: 383–394.
Dreyer EB, Pan ZH, Storm S, Lipton SA . Greater sensitivity of larger retinal ganglion cells to NMDA-mediated cell death. Neuroreport 1994; 5: 629–631.
Vorwerk CK, Kreutz MR, Bockers TM, Brosz M, Dreyer EB, Sabel BA . Susceptibility of retinal ganglion cells to excitotoxicity depends on soma size and retinal eccentricity. Curr Eye Res 1999; 19: 59–65.
Levy DI, Lipton SA . Comparison of delayed administration of competitive and uncompetitive antagonists in preventing NMDA receptor-mediated neuronal death. Neurology 1990; 40: 852–855.
Schuettauf F, Naskar R, Vorwerk CK, Zurakowski D, Dreyer EB . Ganglion cell loss after optic nerve crush mediated through AMPA-kainate and NMDA receptors. Invest Ophthalmol Vis Sci 2000; 41: 4313–4316.
Russelakis-Carneiro M, Silveira LC, Perry VH . Factors affecting the survival of cat retinal ganglion cells after optic nerve injury. J Neurocytol 1996; 25: 393–402.
Yoles E, Muller S, Schwartz M . NMDA-receptor antagonist protects neurons from secondary degeneration after partial optic nerve crush. J Neurotrauma 1997; 14: 665–675.
Kikuchi M, Tenneti L, Lipton SA . Role of p38 mitogen-activated protein kinase in axotomy-induced apoptosis of rat retinal ganglion cells. J Neurosci 2000; 20: 5037–5044.
Derouiche A, Rauen T . Coincidence of L-glutamate/L-aspartate transporter (GLAST) and glutamine synthetase (GS) immunoreactions in retinal glia: evidence for coupling of GLAST and GS in transmitter clearance. J Neurosci Res 1995; 42: 131–143.
Lehre KP, Davanger S, Danbolt NC . Localization of the glutamate transporter protein GLAST in rat retina. Brain Res 1997; 744: 129–137.
Rauen T, Rothstein JD, Wassle H . Differential expression of three glutamate transporter subtypes in the rat retina. Cell Tissue Res 1996; 286: 325–336.
Vorwerk CK, Naskar R, Schuettauf F, Quinto K, Zurakowski D, Gochenauer G et al. Depression of retinal glutamate transporter function leads to elevated intravitreal glutamate levels and ganglion cell death. Invest Ophthalmol Vis Sci 2000; 41: 3615–3621.
Rauen T, Kanner BI . Localization of the glutamate transporter GLT-1 in rat and macaque monkey retinae. Neurosci Lett 1994; 169: 137–140.
Harada T, Harada C, Watanabe M, Inoue Y, Sakagawa T, Nakayama N et al. Functions of the two glutamate transporters GLAST and GLT-1 in the retina. Proc Natl Acad Sci USA 1998; 95: 4663–4666.
Lindqvist N, Peinado-Ramonn P, Vidal-Sanz M, Hallbook F . GDNF, Ret, GFRalpha1 and 2 in the adult rat retino-tectal system after optic nerve transection. Exp Neurol 2004; 187: 487–499.
Jelsma TN, Friedman HH, Berkelaar M, Bray GM, Aguayo AJ . Different forms of the neurotrophin receptor trkB mRNA predominate in rat retina and optic nerve. J Neurobiol 1993; 24: 1207–1214.
Johnson JE, Barde YA, Schwab M, Thoenen H . Brain-derived neurotrophic factor supports the survival of cultured rat retinal ganglion cells. J Neurosci 1986; 6: 3031–3038.
Guidry C . Isolation and characterization of porcine Muller cells. Myofibroblastic dedifferentiation in culture. Invest Ophthalmol Vis Sci 1996; 37: 740–752.
McGillem GS, Guidry C, Dacheux RF . Antigenic changes of rabbit retinal Muller cells in culture. Invest Ophthalmol Vis Sci 1998; 39: 1453–1461.
Vlahos CJ, Matter WF, Hui KY, Brown RF . A specific inhibitor of phosphatidylinositol 3-kinase, 2-(4-morpholinyl)-8-phenyl-4H-1-benzopyran-4-one (LY294002). J Biol Chem 1994; 269: 5241–5248.
Alessi DR, Cuenda A, Cohen P, Dudley DT, Saltiel AR . PD 098059 is a specific inhibitor of the activation of mitogen-activated protein kinase kinase in vitro and in vivo. J Biol Chem 1995; 270: 27489–27494.
Hanke JH, Gardner JP, Dow RL, Changelian PS, Brissette WH, Weringer EJ et al. Discovery of a novel, potent, and Src family-selective tyrosine kinase inhibitor. Study of Lck- and FynT-dependent T cell activation. J Biol Chem 1996; 271: 695–701.
Furuta A, Rothstein JD, Martin LJ . Glutamate transporter protein subtypes are expressed differentially during rat CNS development. J Neurosci 1997; 17: 8363–8375.
Conradt M, Storck T, Stoffel W . Localization of N-glycosylation sites and functional role of the carbohydrate units of GLAST-1, a cloned rat brain L-glutamate/L-aspartate transporter. Eur J Biochem 1995; 229: 682–687.
Schlag BD, Vondrasek JR, Munir M, Kalandadze A, Zelenaia OA, Rothstein JD et al. Regulation of the glial Na+-dependent glutamate transporters by cyclic AMP analogs and neurons. Mol Pharmacol 1998; 53: 355–369.
Raunser S, Haase W, Bostina M, Parcej DN, Kuhlbrandt W . High-yield expression, reconstitution and structure of the recombinant, fully functional glutamate transporter GLT-1 from Rattus norvegicus. J Mol Biol 2005; 351: 598–613.
Rothstein JD, Martin L, Levey AI, Dykes-Hoberg M, Jin L, Wu D et al. Localization of neuronal and glial glutamate transporters. Neuron 1994; 13: 713–725.
Rauen T, Wiessner M . Fine tuning of glutamate uptake and degradation in glial cells: common transcriptional regulation of GLAST1 and GS. Neurochem Int 2000; 37: 179–189.
Chaturvedi LS, Marsh HM, Basson MD . Src and focal adhesion kinase mediate mechanical strain-induced proliferation and ERK1/2 phosphorylation in human H441 pulmonary epithelial cells. Am J Physiol Cell Physiol 2007; 292: C1701–C1713.
Guo J, Wu HW, Hu G, Han X, De W, Sun YJ . Sustained activation of Src-family tyrosine kinases by ischemia: a potential mechanism mediating extracellular signal-regulated kinase cascades in hippocampal dentate gyrus. Neuroscience 2006; 143: 827–836.
Rothstein JD, Patel S, Regan MR, Haenggeli C, Huang YH, Bergles DE et al. Beta-lactam antibiotics offer neuroprotection by increasing glutamate transporter expression. Nature 2005; 433: 73–77.
Nicole O, Ali C, Docagne F, Plawinski L, MacKenzie ET, Vivien D et al. Neuroprotection mediated by glial cell line-derived neurotrophic factor: involvement of a reduction of NMDA-induced calcium influx by the mitogen-activated protein kinase pathway. J Neurosci 2001; 21: 3024–3033.
Vorwerk CK, Naskar R, Schuettauf F, Quinto K, Zurakowski D, Gochenauer G et al. Depression of retinal glutamate transporter function leads to elevated intravitreal glutamate levels and ganglion cell death. Invest Ophthalmol Vis Sci 2000; 41: 3615–3621.
Brooks DE, Komaromy AM, Kallberg ME . Comparative retinal ganglion cell and optic nerve morphology. Vet Ophthalmol 1999; 2: 3–11.
Hanke JH, Gardner JP, Dow RL, Changelian PS, Brissette WH, Weringer EJ et al. Discovery of a novel, potent, and Src family-selective tyrosine kinase inhibitor. Study of Lck- and FynT-dependent T cell activation. J Biol Chem 1996; 271: 695–701.
Acknowledgements
This work was supported by fellowships to PDK from the Alexander von Humboldt Foundation and the Natural Sciences and Engineering Research Council of Canada. We thank VP Sarthy for the rMC-1 cells.
Author information
Authors and Affiliations
Corresponding author
Additional information
Edited by N Bazan
Rights and permissions
About this article
Cite this article
Koeberle, P., Bähr, M. The upregulation of GLAST-1 is an indirect antiapoptotic mechanism of GDNF and neurturin in the adult CNS. Cell Death Differ 15, 471–483 (2008). https://doi.org/10.1038/sj.cdd.4402281
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cdd.4402281
Keywords
This article is cited by
-
N,N-Dimethyl-3β-hydroxycholenamide attenuates neuronal death and retinal inflammation in retinal ischemia/reperfusion injury by inhibiting Ninjurin 1
Journal of Neuroinflammation (2023)
-
Effects of Aerobic Exercise during Pregnancy on Neurobehavioral Performances and Serum Levels of GDNF in Adult Male Mice Offspring
Neurophysiology (2019)
-
Neuritin 1 promotes retinal ganglion cell survival and axonal regeneration following optic nerve crush
Cell Death & Disease (2015)
-
Arundic acid attenuates retinal ganglion cell death by increasing glutamate/aspartate transporter expression in a model of normal tension glaucoma
Cell Death & Disease (2015)
-
Non-viral gene therapy for GDNF production in RCS rat: the crucial role of the plasmid dose
Gene Therapy (2012)