Summary
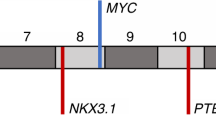
Loss of heterozygosity at chromosome arm 16q is a frequent event in human prostate cancer. In this study, loss of heterozygosity at 16q was studied in 44 prostate cancer patients exhibiting various clinical features. Fifteen polymorphic polymerase chain reaction (PCR) markers were used to identify the separately deleted areas and the findings were compared with clinicopathological variables and 5-year survival of the patients. The results indicated that there are at least three independently deleted regions at 16q. Allelic losses at the central and distal areas were associated significantly with aggressive behaviour of the disease (16q24.1–q24.2, P< 0.01, and 16q24.3–qter, P< 0.05), and the central area of deletion was further significantly associated with poorly differentiated tumour cells (P< 0.05) and with recurrent (P< 0.01) growth of the tumour. During the follow-up period, 28% of the patients initially with M0 disease developed distant metastases. Of the patients showing allelic loss at 16q24.1–q24.2, distant metastasis were found in 45% during the 5-year follow-up period, and 31% of the patients showing loss at 16q21.1 also developed distant metastases. After the 5-year follow-up period, 14 (32%) of the patients remained alive, whereas 19 (43%) had died because of their prostate cancer. The overall survival rate of the patients showing allelic loss at 16q21.1 or 16q24.1–q24.2 was significantly lower than that of the patients with retained heterozygosity.
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Bergerheim, U. S., Kunimi, K., Collins, V. P. & Ekman, P. (1991). Deletion mapping of chromosomes 8, 10, and 16 in human prostatic carcinoma. Genes Chroms Cancer 3: 215–220.
Brendler, C. B., Isaacs, J. T., Follansbee, A. L. & Walsh, P. C. (1984). The use of multiple variables to predict response to endocrine therapy in carcinoma of the prostate: a preliminary report. J Urol 131: 694–700.
Canzian, F., Salovaara, R., Hemminki, A., Kristo, P., Chadwick, R. B., Aaltonen, L. A. & de la Chapelle, A. (1996). Semiautomated assessment of loss of heterozygosity and replication error in tumors. Cancer Res 56: 3331–3337.
Carter, B. S., Ewing, C. M., Ward, W. S., Treiger, B. F., Aalders, T. W., Schalken, J. A., Epstein, J. I. & Isaacs, W. B. (1990). Allelic loss of chromosomes 16q and 10q in human prostate cancer. Proc Natl Acad Sci USA 87: 8751–8755.
Cher, M. L., Bova, G. S., Moore, D. H., Small, E. J., Carroll, P. R., Pin, S. S., Epstein, J. I., Isaacs, W. B. & Jensen, R. H. (1996). Genetic alterations in untreated metastases and androgen-independent prostate cancer detected by comparative genomic hybridization and allelotyping. Cancer Res 56: 3091–3102.
Chisholm, G. D. (1988). TNM classification of urologic tumours in 1988. Br J Urol 62: 501
Délos, S., Carsol, J-L, Ghazarossian, E., Raynaud, J-P & Martin, P. (1995). Testosterone metabolism in primary cultures of human prostate epithelial cells and fibroblasts. J Steroid Biochem Mol Biol 55: 375–383.
Dib, C., Faure, S., Fizames, C., Samson, D., Drout, N., Vignal, A., Millasseau, P., Marc, S., Hazan, J., Seboun, E., Lathrop, M., Gyapay, G., Morrissette, J. & Weissenbach, J. (1996). A comprehensive genetic map of the human genome based on 5,264 microsatellites. Nature 380: 152–154.
Durocher, F., Morrisette, J., Labrie, Y., Labrie, F. & Simard, J. (1995). Mapping of the HSD17B2 gene encoding type II 17β-hydroxysteroid dehydrogenase close to D16S422 on chromosome 16q24.1–q24.2. Genomics 25: 724–726.
Elo, J. P., Akinola, L. A., Poutanen, M., Vihko, P., Kyllönen, A-P, Lukkarinen, O. & Vihko, R. (1996). Characterization of 17β-hydroxysteroid dehydrogenase isoenzyme expression in benign and malignant human prostate. Int J Cancer 66: 37–41.
Elo, J. P., Härkönen, P., Kyllönen, A-P, Lukkarinen, O., Poutanen, M., Vihko, R. & Vihko, P. (1997). Loss of heterozygosity at 16q24.1–q24.2 is significantly associated with metastatic and aggressive behavior of prostate cancer. Cancer Res 57: 3356–3359.
Isaacs, J. T. (1994). Role of androgens in prostatic cancer. Vitam Horm 49: 433–502.
Joos, S., Bergerheim, U. S. R., Pan, Y., Matsuyama, H., Bentz, M., du Manoir, S. & Lichter, P. (1995). Mapping of chromosomal gains and losses in prostate cancer by genomic hybridization. Genes Chromosomes Cancer 14: 267–276.
Kaupmann, K., Becker-Follmann, J., Scherer, G., Jockusch, H. & Starzinski-Powitz, A. (1992). The gene for cell adhesion molecule M-cadherin maps to mouse chromosome 8 and human chromosome 16q24.2–qter and is near the E-cadherin (Uvomorulin) locus in both species. Genomics 14: 488–490.
Klein, H., Bressel, M., Kastendieck, H. & Voigt, K-D (1991). Biochemical endocrinology of prostate cancer. Endocrine Dependent Tumors, Voigt K-D, Knabbe K (eds) 131–163, Raven Press: New York
Labrie, Y., Durocher, F., Lachance, Y., Turgeon, C., Simard, J., Labrie, C. & Labrie, F. (1995). The human type 2 17β hydroxysteroid dehydrogenase gene encodes two alternatively spliced mRNA species. DNA Cell Biol 14: 849–861.
Latil, A., Cussenot, O., Fournier, G., Driouch, K. & Lidereau, R. (1997). Loss of heterozygosity at chromosome 16q in prostate adenocarcinoma: identification of three independent regions. Cancer Res 57: 1058–1062.
Lee, S. W. (1996). H-cadherin, a novel cadherin with growth inhibitory functions and diminished expression in human breast cancer. Nature Med 2: 776–782.
Pullman, W. E. & Bodmer, W. F. (1992). Cloning and characterization of a gene that regulates cell adhesion. Nature 356: 529–532.
Ray, R. & Miller, D. M. (1991). Cloning and characterization of a human c-myc promoter-binding protein. Mol Cell Biol 11: 2154–2161.
Suzuki, H., Komiya, A., Emi, M., Kuramochi, H., Shiraishi, T., Yatani, R. & Shimazaki, J. (1996). Three distinct commonly deleted regions of chromosome arm 16q in human primary and metastatic prostate cancer. Genes Chroms Cancer 17: 225–233.
Umbas, R., Isaacs, W. B., Bringuier, P. P., Schaafsma, H. E., Karthaus, H. F. M., Oosterhof, G. O. N., Debruyne, F. M. J. & Schalken, J. A. (1994). Decreased E-cadherin expression is associated with poor prognosis in patients with prostate cancer. Cancer Res 54: 3929–3933.
Visakorpi, T., Kallioniemi, A. H., Syvänen, A-C, Hyytinen, E. R., Karhu, R., Tammela, T., Isola, J. J. & Kallioniemi, O. P. (1995). Genetic changes in primary and recurrent prostate cancer by comparative genomic hybridization. Cancer Res 55: 342–347.
Vogelstein, B. & Kinzler, K. W. (1992). p53 Function and dysfunction. Cell 70: 523–526.
Werle, E., Schneider, C., Völker, M. & Fiehn, W. (1994). Convenient single-step, one tube purification of PCR products for direct sequencing. Nucleic Acids Res 22: 4354–4355.
Wright, D. K. & Manos, M. M. (1990). ample preparation from paraffin-embedded tissues. PCR Protocols: A Guide to Methods and Applications, Innis MA, Gelfand JJ, White TJ (eds) 153–158, Academic Press: San Diego
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Elo, J., Härkönen, P., Kyllönen, A. et al. Three independently deleted regions at chromosome arm 16q in human prostate cancer: allelic loss at 16q24.1–q24.2 is associated with aggressive behaviour of the disease, recurrent growth, poor differentiation of the tumour and poor prognosis for the patient. Br J Cancer 79, 156–160 (1999). https://doi.org/10.1038/sj.bjc.6690025
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6690025
Keywords
This article is cited by
-
Allelic imbalance and biochemical outcome after radical prostatectomy
Prostate Cancer and Prostatic Diseases (2006)
-
17β-Estradiol upregulates and activates WOX1/WWOXv1 and WOX2/WWOXv2 in vitro: potential role in cancerous progression of breast and prostate to a premetastatic state in vivo
Oncogene (2005)
-
Integration of high-resolution array comparative genomic hybridization analysis of chromosome 16q with expression array data refines common regions of loss at 16q23–qter and identifies underlying candidate tumor suppressor genes in prostate cancer
Oncogene (2004)