Abstract
Study design:
Retrospective study.
Objectives:
To determine the potential impact of rehabilitation care on associated symptoms and functional improvements of paraplegic patients with metastatic spinal cord compression.
Setting:
CMN Propara, Montpellier (France).
Measures:
Demographics, Functional Independence Measure (FIM), Frankel Modified Score and Visual Analog Scale (VAS) for pain, intercurrent adverse medical events and neurological outcome, duration of stay, survival time, rehospitalization in a non-Spinal Cord Injury unit, number of contracts defining the patients rehabilitation goals, number of contracts defining the patients duration of stay within the rehabilitation center.
Results:
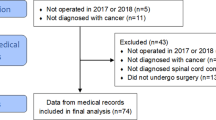
We reviewed the charts of 26 patients. The initial neurological profile was paraplegia or paraparesis for 24 patients and quadriparesis for 2 patients. Regarding functional improvements: four patients demonstrated a poor functional evolution, five patients showed no functional improvements or very slight improvements and all the other patients showed an increase in their overall functional aptitudes. At the end of the stay, 14 patients were urinary independent. Our study reports 52 rehospitalizations in an another unit and 101 outpatient visits during their rehabilitation stay in a physical medicine and rehabilitation (PM&R) center. For the 14 patients who were deceased at the time of data collection, the median survival rate post-paraplegia was 12.7 months. A total of 12 of the 14 patients spent more than a third of their remaining survival time in a rehabilitation center.
Discussion:
Compared to the patients' life expectancy, their stay in a rehabilitation center is too long and prevents them from spending time with family and loved ones. The occurrence rate of the associated symptoms is high because of both cancer-related disorders and neurological disorders caused by the spinal cord lesion. PM&R professionals are faced with patients affected by chronic pain and fatigue as well as frequent rehospitalizations, short stays and outpatient stays, in the primary oncology unit. This study focuses on the need to privilege the patients' comfort over their functional rehabilitation.
Similar content being viewed by others
Introduction
A dearth of relevant literature clearly shows that metastatic paraplegia is a pathology that has been ignored by Physical Medicine and Rehabilitation (PM&R) specialists in the past 30 years.1, 2, 3, 4, 5, 6 Only 10–14% of patients with a spinal epidural metastasis are admitted into a PM&R center.7 The teams are usually not used to dealing with chronic pain, fatigue and other symptoms related to metastatic paraplegia. However, some articles highlight the positive impact of PM&R care for these patients if a contract is clearly established early on listing the rehabilitation goals and the duration of stay. Consequently, this contract is a determining factor for the high rate of patients being able to return home and avoid a long-term hospitalization after a stay in a PM&R center.4 Unfortunately most studies focused on prognosis. Taking into account associated symptoms and functional improvements we conducted a retrospective study to determine if the patients' comfort should prevail on the functional improvements or if these two aspects could be efficiently managed by the PM&R teams.
Methods
The study reports the experience of four PM&R centers that have treated, in the past 5 years (2003–2007), patients with spinal metastases. The corresponding author went to all four centers to supervise the data collection. We collected data on: cancer evolution, history of spinal metastasis, neurological and functional impact, pain, urinary disorders, rehabilitation contract, adverse medical events during the stay, average duration of stay and survival rate. Furthermore, we collected data on rehospitalizations, short-stays or outpatient visits in the primary oncology unit whereas the patients were treated in the PM&R center. The pain was assessed using a Visual Analog Scale (VAS). The analgesics listed in our study were classified using the WHO classification.8 The Functional Independence Measure scale (FIM) is an 18-item, seven level ordinal scale with scores ranging from 1 (total assist) to 7 (complete independence). The patients were classified according to the American Spinal Injury Association (ASIA) standards.9
Results
Demographics data
In total, 26 patients, 21 men and 5 women, were included in the study. The mean age upon admission into a rehabilitation center was 57 years (E: 16–82, s.d.: 15).
Clinical data
Primary cancer locations were: breast, lung and prostate (Figure 1). In 25 cases, the diagnosis of metastasis was made after the diagnosis of the primary cancer. The time delay for the onset of the first symptoms after a known primary cancer was 21 months (E: 0–101, s.d.: 31 months). The first symptoms of spinal metastasis, in a context of a known primary cancer, were back pain (nine cases), neurological pain (four cases), sudden motor deficit (six cases) or progressive motor deficit (seven cases).
In most cases surgery was the first-line treatment for spinal metastasis (Figure 2).
Regarding the intercurrent adverse medical events that occurred during the patients stay, we collected the ones that required a transfer to another unit or major therapeutic treatment (Table 1). Pain complaints were collected using the VAS. The evolution of the VAS pain score during the patients stay is reported in Figure 3 and classified according to a VAS pain score at 0, less than 5 and more than 5 on a scale of 0–10. We were unable to collect the VAS pain scores for 12 patients at the beginning of their stay and for 9 patients at the end of their stay. If the VAS score collection was not done for these patients, the prescriptions showed, however, that analgesics were administered to 19 patients at the beginning of the stay and 12 patients at the end of the stay (Figure 4). No patients had an intrathecal morphine pump implanted. Seven patients constantly used morphine (class 3) as an analgesic during their entire rehabilitation stay.
Our study reports 52 rehospitalizations in an another unit and 101 outpatient visits during the patients rehabilitation stay in a PM&R center.
Neurological data
The topography of the metastasis was thoracic in 22 patients, cervical in 3 patients and thoracocervical in 1 patient.
The initial neurological profile was paraplegia or paraparesis for 24 patients and quadriparesis for 2 patients. Upon admission to the PM&R center, 13 patients were ASIA A or B and 11 were ASIA C or D (Figure 5). We did not have any relevant data for two patients. For 10 patients, we reported no changes in their ASIA classification by the end of their first stay and 2 patients progressed from an ASIA B to D.
Functional data
For seven patients, a rehabilitation care contract was established. It defined the rehabilitation goals, wheelchair recommendations, independence in daily life activities, abilities to manage their transfers, gait status, bladder and sphincter evaluation and finally pressure ulcer treatment. Four patients had a contract that defined the duration of stay. For three patients, the duration of stay was 30–60 days and for one patient it was 90 days. The average duration of stay in a rehabilitation center after a diagnosis of spinal metastasis was 161 days (E: 8–461, s.d.: 115). Six patients came back for a subsequent stay <90 days.
We looked at the progression of the patients' functional status as defined by the differential between FIM scores at the end of the stay and upon admission in the PM&R center. Four patients demonstrated a poor functional evolution, four patients demonstrated no improvements or very slight improvements in their functional abilities (positive differential <10 points) and all other patients demonstrated an increase in their overall functional aptitudes (positive differential>10 points). As seen in Figure 6, the location of the primary cancer did not seem to have an impact on the functional evolution potential and duration of stay.
The data on urinary independence at the beginning of the stay showed that 12 patients proceeded with intermittent self-catheterization, spontaneous miction without residual, imperious miction requiring a penile sheath, miction triggered by bladder tapping. One patient did not have any urinary incontinence. Seven patients had an indwelling catheter. At the end of the stay, 14 patients were urinary independent: 4 with intermittent self-catheterization, 7 with spontaneous miction, 1 with imperious miction and another 1 with miction triggered by bladder tapping. One patient underwent a urinary Bricker derivation. Seven patients still kept an indwelling catheter.
Follow-up
For the 14 patients who were deceased at the time of data collection, the median survival rate post-paraplegia was 12.7 months (E: 3–38, s.d.: 11.2). In Figure 7, duration of hospital stay is benchmarked against the time interval between the onset of their paraplegia and their death. A total of 12 of the 14 patients spent more than a third of their remaining survival time in a rehabilitation center. One patient was lost to follow-up. For the 11 patients who were still alive at the time of data collection, the median survival rate post-paraplegia was 21.1 months (E: 6.5–48, s.d.: 12.1). The ratio ‘duration of hospital stay/survival time’ is shown in Figure 7. Seven patients were transferred to a palliative care unit. One patient was transferred to an intensive care unit where he or she died. No data were available for the four other patients. Finally, 14 of the 26 patients (53%) were able to return home.
Discussion
Not much information is available on PM&R care for patients with metastatic paraplegia. Our study underlines the need for standardized admission criteria and rehabilitation care contracts defining the rehabilitation goals and duration of stay. This contract is very useful in providing the patients with realistic rehabilitation goals and potential functional outcomes.
Associated symptoms
Besides the side effects triggered by anticancer therapeutics, the symptoms associated to metastatic paraplegia do not seem to differ from those associated to trauma spinal cord injury (SCI). It is more the context surrounding these associated symptoms that differs from classic SCI patient care.
The symptoms can be common to trauma SCI but aggravated by cancer (primary cancer and spinal metastasis): back pain or neuropathic pain, transit disorders, static disorders of the spine, upper and lower respiratory tract infections and skin disorders. The symptoms can also be cancer related: appetite disorders, fatigue, psychological disorders, adverse effects of first-line treatments. The McKinley prospective study reports urinary tract infections (UTIs) for 14 patients, respiratory tract complications for 6 patients and other symptoms for 6 other patients: pressure ulcers, gastroparesis, intestinal obstruction, nervous breakdown, hyponatremia and neutropenia.4
Pain
For the patients in our study the pain was constant from the beginning to the end of their rehabilitation stay. We collected data on the intensity of their pain or on the amount of analgesics prescribed. We reported pain as the most frequent first symptom (more than half of the patients). The Helweg-Larsen study reports that 88% of their 153 patients demonstrated pain at the time of the spinal metastasis diagnosis when the locomotor disorders appeared.10 Pain can be a telltale sign of spinal metastasis sometimes several months before the first neurological symptoms, median time interval being 40 days.10 This elapsed time between the onset of pain, and the diagnosis of spinal metastasis has a damaging psychological impact on the patients by reminding them that specialists took a long time to correlate their pain to a spinal metastasis, especially before the onset of the first neurological symptoms.
In the McKinley study patients demonstrated at first a fluctuating pain that rapidly became persistent4 once the neurological disorders had set in. The pain can be caused by existing lesions (bone destruction, vertebral instability, spinal cord compression, SCC) or sometimes by recurrent or extensive lesions or even spinal nerve root compression (SNRC), in all cases this pain alters considerably the quality of life of these patients and impairs the assessment of their potential functional progresses. The nighttime recrudescence of the pain is shown to be a warning sign of metastatic relapse.11 In a large cohort of 398 patients, Bach et al.12 estimated at 83% the percentage of patients suffering from SCC pain before the onset of the first neurological symptoms. After that the pain is classified into SCC pain (36%) and SNRC pain (47%).
The pain can be spontaneous but also triggered by the rehabilitation exercises provided by the PM&R teams and thus be a major obstacle to the patients functional progresses. Therefore our study demonstrates the need for PM&R teams to elaborate a specific rehabilitation program for patients with metastatic paraplegia different from the one designed for trauma SCI patients. To alleviate back pain, opioids as well as steroidal and nonsteroidal anti-inflammatory drugs are limited by their classic adverse effects (transit disorders, sleepiness…)13 but are still largely prescribed. In the Guo cohort, 93% of patients were taking opioid drugs at the end of their rehabilitation stay.6 For SNRC pain neuropathic analgesics are efficient but limited due to their potential adverse hematological reactions.14 The validated efficacy of the γ-aminobutyric acid analogs should overcome this limitation.
Morphine is the best option for cancer pain because of its low or lack of toxicity on the organs. Transdermic patches are even better—less impact on transit functions.13 The initial high-dose corticosteroids prescribed to limit the spinal cord edema,15 should be progressively decreased to moderate-term, low-dose corticosteroids to ensure the necessary additional pain relief.15
Transit disorders
Constipation affected all the patients in our study yet this topic is not often addressed in the literature. The management of these transit disorders differs from classic trauma SCI care. For spinal metastasis patients with incomplete paraplegia the efforts produced during a bowel movement (Valsalva maneuver) trigger neuropathic and back pain. Furthermore for these patients who combine potential undernutrition, the adverse effects of opioids and neuropathic analgesics, immobility and vegetative dysfunctions, and constipation should be avoided at all costs.
Urological disorders
These symptoms rarely come first. In fact, they appear alongside the locomotor disorders, 50% of patients have to empty their retentive bladder by indwelling catheterization, intermittent catheterization or self-catheterization. The sole fact that 14 patients kept their urinary independence at the end of their rehabilitation stay shows the positive impact of the training and educational teachings provided by the PM&R staff. Seven patients regained a spontaneous miction and seven others kept a catheter. For some patients with an altered general state, a well-managed indwelling catheter, might be a better solution than problematic self-catheterizations.
Static disorders of the spinal cord
Some patients might benefit from wearing a chest harness to prevent an aggravation of their spinal kyphosis, limit the deformation of the spinal cord, mainly at the junction areas and reduce the intensity of their pain. For some authors this chest harness should not be kept for more than 6 to 10 weeks.16 In regards to the duration of stay in the rehabilitation center and the rehabilitation objectives set, this already seems too long.
Appetite disorders
Mostly they are linked to the primary tumor rather than the spinal metastasis.17 The PM&R staff should not overlook the negative impact of anticancer drugs on the patient's appetite. The literature reports an alteration of the patients' general state and their skin as well as their aptitude to go on with therapy without a proper food intake. The undernutrition is reflected in the albumin levels: for 62% of the patients of the Guo cohort, the levels are below 3.5 g per 100 ml.6 Resorting to a gastrostomy feeding tube should mark the end of the patients stay in a PM&R unit, they should be transferred to a palliative care unit.
Fatigue
We should discard any metabolic disorders and run some blood tests (anemia, leukopenia) before incriminating any other etiology. A third of the patients from the Guo demonstrated hemoglobin levels <10 g per 100 ml but their anemia did not interfere with their survival prognosis.6 Conversely, Huddart18 demonstrated that hemoglobin levels>12 g per 100 ml were a good predictive factor for survival. This fatigue is also due to anticancer therapeutics and undernutrition and is the result of various associated symptoms. The PM&R teams have to take this fatigue into account when establishing the patients' rehabilitation care contract because of its potential impact on their decision to go on with their rehabilitation program.
Psychological impact
The patients are aware of the severity of their cancer but at the same time they tend to undermine the seriousness of their functional prognosis.17 They are afraid of being hospitalized, they have to deal with the suffering of their family and loved ones as well as their fear and anxiety of rupture and death.
Metastatic recurrence is a constant threat but in reality only observed in 8 to 20% of the patients according to the cohorts19, 20 with an average onset time of 7 months after the initial diagnosis. The initial spinal location is identified in 55% of cases and another location is discovered in 45% of cases. The recurrence risk is correlated to the survival rate: 50% of survivors at +2 years and almost all survivors at +3 years develop a recurrence. However, for initial multifocal spinal metastasis, the recurrence risk is not statistically more important (7.1% for one location, 8.1% if there is more than one location).21
The necessary psychological counseling must focus on offering different solutions to the patients and their family through individual therapy, support groups and behavioral techniques.
Adverse side effects for first-line treatments
Corticosteroids are useful in the acute phase of the neurological lesion and for their pain-relief action. This universal consensus is backed up by a high level of proof,22 yet no agreement was reached on the maximum dosage and treatment duration versus toxicity. Sorensen et al.15, in a randomized study (radiotherapy and high dosage of dexamethasone versus radiotherapy alone) reported a significant gait improvement (P=0.046) in 57 patients (SCC) taking corticosteroids. However, the study reported a high level of adverse effects and three patients demonstrated serious ones such as a severe psychosis, gastric perforation and ulcer. The long-term impact of corticosteroids on neurological improvement is still being debated.23 Furthermore, for the post-surgical phase, no data are available on a potential infectious risk linked to the use of corticosteroids.
Radiotherapy has a low morbidity rate. Its pain-relief impact has been demonstrated in 80–90% of patients.24 The side effects of this therapeutic are mainly fatigue and nausea, sometimes digestive disorders when the digestive tract is irradiated.
Chemotherapy is usually used for treating the primary cancer and not the local spinal metastasis. However, PM&R teams must be aware of the potential side effects of the patients chemotherapy protocol and redefine, if necessary, the limits of the rehabilitation care for these patients.
Surgery does not trigger any morbidity besides the major septic risk of a surgical wound in a context of associated radiotherapy, undernutrition and skin weakness. Wound complications (32%) were observed when radiotherapy took place before surgery compared with 12% if surgery was the first-line treatment (P<0.05).25
Rehabilitation care contract
When life expectancy conditions the PM&R care, the contract defines right away the limits of the patients rehabilitation stay. This contract goes beyond the patients' sole judgments and wishes even though their needs and expectations are taken into account. The objectives of the rehabilitation care must be weighed against the emotional and physical loads the family can manage. This contract will also take into account the advice from neurosurgeons and oncologists and the associated symptoms. The goal of this contract is to define the right balance between time left to spend with family and loved ones and time spent on rehabilitation training. A prospective study on the functional outcome of 32 patients over 5 years, suggested that the functional status or functional improvements can last up to 3 months after leaving the rehabilitation center.4 It showed that the functional improvements and the progression rate are identical for metastatic patients and trauma SCI patients in the first 3 months. We are probably faced with a selection bias since the patients were included based on a vital prognosis>3 months and an aptitude to undergo at least 3 h of rehabilitation training per day. A literature analysis led us to assess the evolution rate using the Karnofsky performance status scale (KPSS)26 (Table 2), a so-called Piper fatigue scale27, 28 and the FIM.29 The correlation between the FIM and the KPSS is linear.30 When the KPSS is lower or equal to 40 for a non-walking patient with spinal metastasis on a lung neoplasm, Abrahm16 estimated the survival rate at 4.4 months in average limiting the perspectives of rehabilitation care. This underlines the relevance of drawing the evolution curve according to three criteria—the FIM, the KPSS and the Piper index—and to determine beyond the first month the balance: survival time/PM&R care/time left with family and loved ones (Figure 8). The contract defined in the first month (Table 3) was partly based on the cancer rehabilitation care principles described by Gamble et al.17 and by O'Toole et al.30 The basis of this contract are: early admission into a SCI Unit, explicit care contract, taking fatigue into account, clearly identifying the role of each PM&R professional, involving the patients and their family in all decisions. We need to be pragmatic in our therapeutic choices.
Beyond the first month, based on the patients' status, new rehabilitation training aspects can be added to the original contract (Table 3).
Conclusion
Thanks to the recent therapeutic advances for treating primary cancer, patients are living longer. There is a suggested higher incidence of lung, prostate and breast cancers,31 leading to complications such as metastatic SCC. In this epidemiological context PM&R teams will be treating more and more patients with metastatic paraplegia in the future. Most patients demonstrate some functional improvements and our literature review underlines the need for a compromise between the time spent in a PM&R center and the time left to spend with family and loved ones. The key to a successful rehabilitation program is the patient's active participation, PM&R teams can achieve that by setting up reasonable rehabilitation goals, taking the patients' associated symptoms into consideration and privileging their comfort. However, to this day, no study can demonstrate the impact of rehabilitation in controlling the morbidity and increasing the patients' life expectancy.
Even though we are aware of the main limits of a retrospective study: incomplete patient charts, memory bias, missing data and study selection bias. The aim of this retrospective study was really to set up a rationale for a controlled prospective study based on the same reading tool.
References
Eriks IE, Angenot EL, Lankhorst GJ . Epidural metastatic spinal cord compression: functional outcome and survival after inpatient rehabilitation. Spinal Cord 2004; 42: 235–239.
Hacking HG, Van As HH, Lankhorst GJ . Factors related to the outcome of inpatient rehabilitation in patients with neoplastic epidural spinal cord compression. Paraplegia 1993; 31: 367–374.
Murray PK . Functional outcome and survival in spinal cord injury secondary to neoplasia. Cancer 1985; 55: 197–201.
McKinley WO, Conti-Wyneken AR, Vokac CW, Cifu DX . Rehabilitative functional outcome of patients with neoplastic spinal cord compressions. Arch Phys Med Rehabil 1996; 77: 892–895.
McKinley WO, Huang ME, Brunsvold KT . Neoplastic versus traumatic spinal cord injury: an outcome comparison after inpatient rehabilitation. Arch Phys Med Rehabil 1999; 80: 1253–1257.
Guo Y, Young B, Palmer JL, Mun Y, Bruera E . Prognostic factors for survival in metastatic spinal cord compression: a retrospective study in a rehabilitation setting. Am J Phys Med Rehabil 2003; 82: 665–668.
McKinley WO, Seel RT, Hardman JT . Nontraumatic spinal cord injury: incidence, epidemiology, and functional outcome. Arch Phys Med Rehabil 1999; 80: 619–623.
WHO. Traitement de la Douleur Cancéreuse. WHO (OMS), WHO Press: Geneva, Switzerland, 1997.
Marino RJ, Barros T, Biering-Sorensen F, Burns SP, Donovan WH et al. International standards for neurological classification of spinal cord injury. J Spinal Cord Med 2003; 26 (Suppl 1): S50–S56.
Helweg-Larsen S, Sorensen PS . Symptoms and signs in metastatic spinal cord compression: a study of progression from first symptom until diagnosis in 153 patients. Eur J Cancer 1994; 30A: 396–398.
Kienstra GE, Terwee CB, Dekker FW, Canta LR, Borstlap AC et al. Prediction of spinal epidural metastases. Arch Neurol 2000; 57: 690–695.
Bach F, Larsen BH, Rohde K, Borgesen SE, Gjerris F et al. Metastatic spinal cord compression. Occurrence, symptoms, clinical presentations and prognosis in 398 patients with spinal cord compression. Acta Neurochir (Wien) 1990; 107: 37–43.
Vrionis FD, Small J . Surgical management of metastatic spinal neoplasms. Neurosurg Focus 2003; 15: E12.
Spinazze S, Caraceni A, Schrijvers D . Epidural spinal cord compression. Crit Rev Oncol Hematol 2005; 56: 397–406.
Sorensen S, Helweg-Larsen S, Mouridsen H, Hansen HH . Effect of high-dose dexamethasone in carcinomatous metastatic spinal cord compression treated with radiotherapy: a randomised trial. Eur J Cancer 1994; 30A: 22–27.
Abrahm JL . Assessment and treatment of patients with malignant spinal cord compression. J Support Oncol 2004; 2: 377–388, 391; discussion 391–373, 398, 401.
Gamble GL, Brown PS, Kinney CL, Maloney FP . Cardiovascular, pulmonary, and cancer rehabilitation. 4. Cancer rehabilitation: principles and psychosocial aspects. Arch Phys Med Rehabil 1990; 71 (4-S): S244–S247.
Huddart RA, Rajan B, Law M, Meyer L, Dearnaley DP . Spinal cord compression in prostate cancer: treatment outcome and prognostic factors. Radiother Oncol 1997; 44: 229–236.
Chamberlain MC, Kormanik PA . Epidural spinal cord compression: a single institution's retrospective experience. Neuro-oncol 1999; 1: 120–123.
van der Sande JJ, Boogerd W, Kroger R, Kappelle AC . Recurrent spinal epidural metastases: a prospective study with a complete follow up. J Neurol Neurosurg Psychiatry 1999; 66: 623–627.
Helweg-Larsen S . Clinical outcome in metastatic spinal cord compression. A prospective study of 153 patients. Acta Neurol Scand 1996; 94: 269–275.
Klimo Jr P, Kestle JR, Schmidt MH . Treatment of metastatic spinal epidural disease: a review of the literature. Neurosurg Focus 2003; 15: E1.
Gale J, Mead GM, Simmonds PD . Management of spinal cord and cauda equina compression secondary to epidural metastatic disease in adults with malignant germ cell tumours. Clin Oncol (R Coll Radiol) 2002; 14: 481–490.
Tong D, Gillick L, Hendrickson FR . The palliation of symptomatic osseous metastases: final results of the Study by the Radiation Therapy Oncology Group. Cancer 1982; 50: 893–899.
Ghogawala Z, Mansfield FL, Borges LF . Spinal radiation before surgical decompression adversely affects outcomes of surgery for symptomatic metastatic spinal cord compression. Spine 2001; 26: 818–824.
Mor V, Laliberte L, Morris JN, Wiemann M . The Karnofsky performance status scale. An examination of its reliability and validity in a research setting. Cancer 1984; 53: 2002–2007.
Piper BF, Dibble SL, Dodd MJ, Weiss MC, Slaughter RE et al. The revised piper fatigue scale: psychometric evaluation in women with breast cancer. Oncol Nurs Forum 1998; 25: 677–684.
Dittner AJ, Wessely SC, Brown RG . The assessment of fatigue: a practical guide for clinicians and researchers. J Psychosom Res 2004; 56: 157–170.
Granger CV, Hamilton BB, Linacre JM, Heinemann AW, Wright BD . Performance profiles of the functional independence measure. Am J Phys Med Rehabil 1993; 72: 84–89.
O'Toole DM, Golden AM . Evaluating cancer patients for rehabilitation potential. West J Med 1991; 155: 384–387.
Cowap J, Hardy JR, A'Hern R . Outcome of malignant spinal cord compression at a cancer center: implications for palliative care services. J Pain Symptom Manage 2000; 19: 257–264.
Acknowledgements
We thank B Clément for the quality of her translation and C Gilbert for her quality work in bibliographical research. This study was presented in part during the National Congress of the French Society of Physical Medicine and Rehabilitation (SOFMER) in Rouen (France), 19–21 October 2006 and in Saint-Malo (France), 4–6 October 2007.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fattal, C., Gault, D., Leblond, C. et al. Metastatic paraplegia: care management characteristics within a rehabilitation center. Spinal Cord 47, 115–121 (2009). https://doi.org/10.1038/sc.2008.75
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sc.2008.75
Keywords
This article is cited by
-
Functional status of patients with metastatic spinal cord compression
Supportive Care in Cancer (2018)
-
Understanding the Role of Rehabilitation Medicine in the Care of Patients with Tumor Causing Spinal Cord Dysfunction
Current Physical Medicine and Rehabilitation Reports (2017)
-
Neoplastic myelopathies and traumatic spinal cord lesions: an Italian comparison of functional and neurological outcomes
Spinal Cord (2011)
-
Survival after rehabilitation for spinal cord injury due to tumor: a 12-year retrospective study
Journal of Neuro-Oncology (2011)
-
Critères de prise en charge en médecine physique et de réadaptation et pathologies cancéreuses
Oncologie (2010)