Abstract
Plasticizers increase the flexibility of plastics. As environmental leachates they lead to increased water and soil pollution, as well as to serious harm to human health. This study was set out to explore various web applications to predict the toxicological properties of plasticizers. Web-based tools (e.g., BOILED-Egg, LAZAR, PROTOX-II, CarcinoPred-EL) and VEGA were accessed via an 5th–10th generation computer in order to obtain toxicological predictions. Based on the LAZAR mutagenicity assessment was only bisphenol F predicted as mutagenic. The BBP and DBP in RF; DEHP in RF and XGBoost; DNOP in RF and XGBoost models were predicted as carcinogenic in the CarcinoPred-EL web application. From the bee predictive model (KNN/IRFMN) BPF, di-n-propyl phthalate, diallyl phthalate, dibutyl phthalate, and diisohexyl phthalate were predicted as strong bee toxicants. Acute toxicity for fish using the model Sarpy/IRFMN predicted 19 plasticizers as strong toxicants with LC50 values of less than 1 mg/L. This study also considered plasticizer effects on gastrointestinal absorption and other toxicological endpoints.
Similar content being viewed by others
Introduction
In everyday life, plastics are utilized for various applications and provide a variety of benefits to consumers, including medical treatment, and technological breakthroughs1. At the same time, plasticizers are emerging contaminants of food products from a variety of food-packaging. They cause health risks to organisms and humans2,3. Environmental concerns include the build-up of waste in landfills, as well as contaminations of ground, coastal and increasingly also open ocean waters. Chemical compounds which are used in soft, flexible, and transparent plastic production can contaminate aquatic and marine environments if they are soluble and hydrophilic. Volatile plasticizers may cause atmospheric pollution and affect the respiration in human and other organisms. Plasticizers as emerging contaminants were detected in environmental partitions of the geo-, atmo-, hydro- and bio-sphere, such as in dust, air, wastewater, drinking water, organisms and food4. Plasticizers are extensively disseminated into the sea, posing toxic effects to marine organisms5 and were found in aerosols above the East China Sea due to fossil fuel burning6. Plasticizers can cause adverse effects on the human endocrine systems by consumption of microplastics from various food sources that are contaminated with plasticizers. Plasticizers may readily be absorbed and dispersed in lipids if they are lipophilic, resulting in body malfunctions, age-related bioaccumulation, and bio-magnification throughout trophic webs7.
Bisphenols (Bishydroxy-aryl alkanes) are phenol derivatives. BPA alone exceeds 3.8 million tons of production8,9. It may be found in a variety of daily items such as water pipelines, computer parts, toys, and thermal papers. There is rising evidence that BPA is detrimental for human health8,10. It also has oxidative and mutagenic potential with the ability to cause DNA methylation and oxidative stress11. BPA is further carcinogenic, mutagenic, immunotoxic and hepatotoxic. Besides, BPA is suggested to be neurotoxic and teratogenic, however, with inconsistent evidence as yet12. BPA exposure enhances the risk of heart diseases, obesity, and diabetes in humans13. BPA poses a significant hazard to potable water and food safety14. The above examples resulted in substantial research into exposure-associated health issues related to phthalates and plasticizers generally in humans. During lactation, maternal oral exposure to BPA was increasing the probability of mammary carcinogenesis in a dimethylbenzanthracene (DMBA)-induced rat model6. The pharmacological effect of BPA in breast cancer cells is mediated through G protein-coupled estrogen receptor 1 (GPER). As a result, GPER-mediated signaling is considered as one of the transduction pathways via which BPA may promote cancer development15. BPA was demonstrated to impact the mammary gland, brain, and reproductive system in rat after fetal and postnatal exposure. It also promotes the growth of hormone-dependent cancers16.
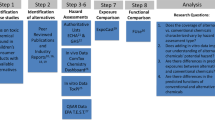
Followed by customer complaints, BPA was removed from numerous plastic items, resulting in the development and usage of bisphenol alternatives with equally toxic outcomes17. In 2000, Fiege and co-workers reported the potential toxicities of bisphenol A derivatives8. This holds particularly for analogues such as bisphenol F (BPF) and bisphenol S (BPS). BPF is used to make electrical insulators, epoxy resins, and food packages5. Both analogues, BPS and BPF were detected in consumer products of daily use such as paper, and also in products from the food industry18,19. Non-toxic or less hazardous chemicals should replace toxic compounds to minimize negative impacts on the environment and the public, according to government laws. Several chemical substitutes, on the other hand, are still untested before being sold and might be hazardous or even more damaging than the original compounds20. This also holds for example for perfluorinated compounds used as pesticides21. The flame-retardants BPS and BPF, being structural analogues of BPA, may have similar physiological effects compared to BPA. In situ and in vitro screening of compounds for toxicological effects is a costly process in terms of material, infrastructure and human resources22. In this situation web applications can provide inexpensive alternatives to predict the toxicological properties of test chemicals (Fig. 1)23. These methods can also offer priority criteria for toxicological assessments that should later be tested in vitro. Animal testing will be reduced and replaced, which is both costly and ethically difficult24. The use of in silico techniques like QSAR would minimize the use of animals in chemical toxicity testing and improve the efficacy of pollutant risk evaluation for human and environmental health25. An important toxicological endpoint here is mutagenicity. Since several mutations are induced by chemicals, it is worthwhile to develop alternative screening methods, such as robust in silico methods to predict chemically induced mutagenicity26. In this study, we used the VEGA platform to predict fish and honey bee acute toxicity. We evaluated plasticizer toxicological endpoints and demonstrated applicability and acceptability of web tools and the VEGA software platform.
Pictorial representation of in silico analysis of various predictive toxicity analysis (ChemBioDraw Ultra 14—https://scistore.cambridgesoft.com/chembiodraw/).
Materials and methods
Data collection of plasticizer chemical structures
Test compound names and abbreviations were retrieved from ChemSpider (S.Table 1). Two-dimensional molecular plasticizer structures were designed using Marvin 17.21.0, ChemAxon and are shown in (S.Fig. 1) based on the available chemical data from ChemSpider27.
SMILES mediated input for the test compounds
Structures of the test molecules are presented as a simplified molecular-input line entry system (SMILES) which were used for toxicity predictions. These SMILES-based assessments were shown to be computationally more efficient in in silico studies28. All the test compounds used in this study are shown in S.Table 2 in SMILES format.
Predictive toxicology
Mutagenicity prediction
To assess the mutagenicity of chemical compounds, the bacterial reverse mutation assay (AMES test) is frequently applied29. To reduce experimental time and materials, the LAZAR web server was used to predict the mutagenicity of plasticizers. This web server was developed and published by Maunz and co-workers in 201330 from available experimental data sets of a bacterial AMES mutagenicity test. The LAZAR toxicity prediction tool is available from https://nano-lazar.in-silico.ch/predict. It provides toxicological predictions by the analysis of compound structures. This holds for mutagenicity, blood–brain barrier penetration, rodent carcinogenicity, and maximum recommended doses. This web server applies for the prediction of toxicity of known and unknown chemical structures31. It provides a modular program for toxicity prediction30. LAZAR can predict the complex toxicological properties of compounds like mutagenicity, carcinogenicity, acute toxicity, maximum recommended daily dose, and blood–brain barrier penetration of chemical substances by applying data mining algorithms. This provides LAZAR with the advantage of providing toxicological predictions for endpoints that provide sufficient empirical information. The LAZAR approach prevents incomplete or incorrect background information from affecting the model predictions30. This web server is available from https://nano-lazar.in-silico.ch/predict. In the toxicological endpoints of chemical substances, mutagenicity and carcinogenicity are of great concern because of their serious effects on human health. Mutagenicity represents one of the most important end points of toxicity and also indicates stable changes in the DNA sequence of an organism, which may result in heritable changes in the characteristics of organisms32. The mechanism of action of mutagenic chemicals is mainly through DNA damage, which causes mutagenic effects such as chromosomal aberrations, frameshift mutations, and point mutations. Mutagens are not only involved in carcinogenesis and genotoxicity. They are also involved in the inception and pathogenesis of several chronic diseases including neurodegenerative disorders, diabetes, aging processes, arthritis, cardiovascular impairment, chronic inflammation, and hepatic disorders33. The LAZAR toxicity prediction accessible link is given in Table 1. LAZAR allows the prediction of several toxicological endpoints such as mutagenicity, rodent carcinogenicity blood–brain barrier penetration, and maximum recommended daily exposure of chemicals34.
Honey bee toxicity prediction
VEGA extension allows to evaluate the properties of chemical substances. It allows to use all available VEGA QSAR models inside the analytical platform KNIME. The extension contains a node for the execution of VEGA models, taking as input a molecule set in SMILES format, and a further node to load and import structures from a SDF file. The model's results similarly to the text output from the VEGA stand-alone application, so that it is possible to use such results inside the workflow. VEGA offers a broad series of information, and the user should be aware of the elements offered by VEGA to assess a chemical substance. Considering all the evidence provided by VEGA the user may evaluate how credible a prediction is, and the level of uncertainty associated with the prediction. VEGA is a complex platform which basically includes a number of QSAR models, and an independent tool helping the user in the evaluation of the result, through an Applicability Domain Index. In this way VEGA combines QSAR and read across tools. The QSAR prediction models derive from CAESAR, T.E.S.T., SARpy, EPISuite, Toxtree, and other tools. New models are frequently added. Registered users are notified when new models are available. A completely independent algorithm is used to assess the reliability of the model prediction through what we call the Applicability Domain Index. This algorithm works on all the separate QSAR models. The algorithm shows similar compounds, assesses the QSAR results on similar compounds, and analyzes some relevant chemical features in the target compound and its related compounds. An automatic evaluation for read-across is done, and thus this tool can be used for read across, independently from the prediction obtained through the QSAR models. In the ideal situation both pieces of information, from the QSAR model and from the Applicability Domain Index, should be assessed. Hymenopteran insects, such as solitary bees, bumblebees and honey bees are important for their ecological services as pollinators of wild plants as well as economically important crops; they also provide honey and honey wax35. Toxicity prediction such as bee acute toxicity through the computational model KNN/IRFMN—v.1.0.0 was done via the downloadable JAVA based application VEGA version 1.1.4.
Fish acute toxicity prediction
Acute toxicity assessments using fish are important for the risk assessments of environmental hazards of chemical compounds in aquatic systems by national legislation. Alternative methods like QSAR applications could substitute several ecotoxicity tests including experimental fish acute toxicity36. The acute fish toxicity model Sarpy/IRFMN v.1.0.2 accessed through the VEGA version 1.1.4. platform predicted fish acute toxicity in our study.
Rodent oral toxicity prediction
The PROTOX-II webserver provides a prediction of rodent oral toxicity. For its application compound similarity analysis of median lethal doses (LD50) provided information about toxic fragments of given compounds37. ProTox-II is an online tool and was developed for toxicity prediction to design the development of the drug. This server can be accessed through a web link https://tox-new.charite.de/protox_II/. Animal trials are fundamental procedures for the determination of possible toxic effects of drug candidates and cosmetics. In silico prediction methods represent an alternative approach and aim to rationalize the preclinical drug development, thus enabling the reduction of the associated time, costs and animal experiments. In oral toxicity prediction, ProTox-II is working based on the analysis of the similarity of compounds with known median lethal doses (LD50) and incorporates the identification of toxic fragments, therefore, representing a novel approach in toxicity prediction. In addition, the web server includes an indication of possible toxicity targets which is based on an in-house collection of protein–ligand-based pharmacophore models (‘toxicophores’) for targets associated with adverse drug reactions. For the strong performance of ProTox-II (sensitivity, specificity and precision of 76, 95 and 75%, respectively) and superiority over other toxicity prediction tools, indicating their possible applicability for other compound classes38. Advancement in the field of computational research has made it possible for in silico methods to offer significant benefits to both regulatory needs and requirements for risk assessments, and the pharmaceutical industry to assess the safety profile of a chemical. Here, we present ProTox-II that incorporates molecular similarity, pharmacophores, fragment propensities and machine-learning models for the prediction of various toxicity endpoints; such as acute toxicity, hepatotoxicity, cytotoxicity, carcinogenicity, mutagenicity, immunotoxicity, adverse outcome pathways (Tox21) and toxicity targets. The predictive models are built on data from both in vitro assays (e.g. Tox21 assays, Ames bacterial mutation assays, hepG2 cytotoxicity assays, immunotoxicity assays) and in vivo data (e.g. carcinogenicity, hepatotoxicity). The webserver takes a two-dimensional chemical structure as an input and reports the possible toxicity profile of the chemical for 33 models with confidence scores, and an overall toxicity radar chart along with three most similar compounds with known acute toxicity37. ProTox-II incorporates the prediction of the various toxicities of the chemical which can be measured qualitatively as binary (active or inactive) in terms of endpoints such as acute toxicity, hepatotoxicity, carcinogenicity, immunotoxicity, mutagenicity, cytotoxicity, adverse outcome (toxicology in the twenty-first century) pathways, and toxicity targets39. It can further be measured quantitatively as 50% lethal dose (LD50) values. Toxicity classes (I–VI) can also be predicted based on toxic doses40. ProTox-II) https://tox-new.charite.de/protox_II (the web server is computational freely available in silico toxicity prediction tool consisting of 33 models to design the development of a drug). It incorporates the prediction of oral toxicities of chemicals which can be measured at different levels of toxicity mainly qualitatively in terms of endpoints as binary output (active or inactive) along with confidence scores such as acute toxicity, hepatotoxicity, carcinogenicity, immunotoxicity, mutagenicity, cytotoxicity, adverse outcome (Tox21) pathways, and toxicity (LD50) values in mg/kg body weight. Toxicity classes (Class I–VI) can also be predicted based on their toxic doses. Prediction accuracy, average similarity along with LD50, and toxicity class were generated instantly for the prediction of acute toxicity and toxicity targets. The phytochemicals and heterodimeric drug candidates were subjected to in silico pharmacodynamic studies to determine their potential cytotoxicity using the online web server ProTox-II (https://tox-new.charite.de/protox_II/, accessed on 28 November 2021)37. ProTox-II is a virtual lab for the predictive toxicity of small molecules, a process that can reduce the need for subsequent drug (in vivo) testing in animals. The ProTox-II server was used to determine predicted acute oral toxicity of the phytochemical compounds in rodents based upon 2D structure similarities with 38,000 compounds (provided by the software) and their associated LD50 values.
Carcinogenicity prediction
Prediction of chemical carcinogenicity was attempted by using ensemble learning methods via CarcinoPred-EL. This web application accessible link is given in (Table 1). This application has three integrated ensemble models, namely Ensemble RF, SVM and XGBoost to provide carcinogenicity predictions. The ensemble learning method is commonly used to predict the carcinogenicity of chemicals41. Carcinogenicity refers to a highly toxic end point of certain chemicals, and has become an important issue in the drug development process. Three novel ensemble classification models, namely Ensemble SVM, Ensemble RF, and Ensemble XGBoost, were developed to predict carcinogenicity of chemicals using seven types of molecular fingerprints and three machine learning methods based on a dataset containing 1003 diverse compounds with rat carcinogenicity. More precisely in chemical datasets, the problem of imbalanced data is common. Most of the in silico models for the prediction of bioactivities as well as toxicity profiles had to rely on an imbalanced dataset42. In binary classification, the underrepresented class is generally referred to as minority class, and the over represented class is referred to as majority. It is often observed that in case of an asymmetric class distribution, regular classifiers like support vector machine (SVM), as well as neural network (NN) tend to ignore the minority class, and treat them as noise resulting in a class boundary that unduly benefits the majority class43. In CarcinoPred-EL among these three models (SVM,RF and XGBoost), Ensemble XGBoost is found to be the best, giving an average accuracy of 70.1 ± 2.9%, sensitivity of 67.0 ± 5.0%, and specificity of 73.1 ± 4.4% in five-fold cross-validation; an accuracy of 70.0%, sensitivity of 65.2%, and a specificity of 76.5% in external validation. In comparison with some recent methods, the ensemble models outperform some machine learning-based approaches and yield equal accuracy and higher specificity but lower sensitivity than rule-based expert systems. It is also found that the ensemble models could be further improved if more data were available. As an application, the ensemble models are employed to discover potential carcinogens in the DrugBank database. The results indicate that the proposed models are helpful in predicting the carcinogenicity of chemicals41. A web server called CarcinoPred-EL has been built for these models (http://112.126.70.33/toxicity/CarcinoPred-EL/index.html).
Predicting gastrointestinal absorption and brain penetration of plasticizers
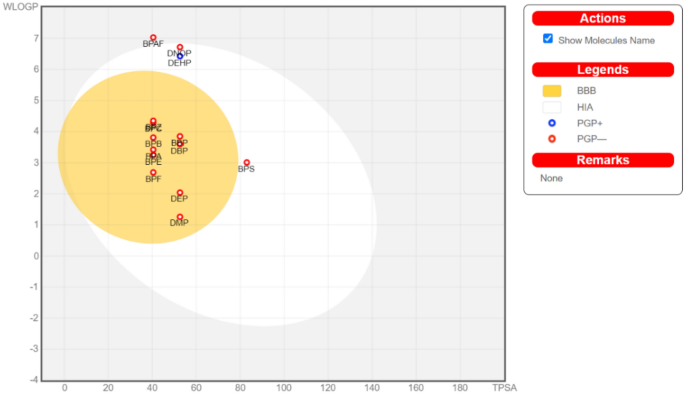
The Brain OrIntestinaLEstimateD permeation method (BOILED‐Egg) was utilized to investigate the gastrointestinal absorption and brain access property of plasticizers. This is a relatively precise prediction model that makes use of lipophilicity and polarity distributions of small molecules. This web application is commonly applied to predict the carcinogenicity of small molecules and lead compounds. We used it here for concomitant predictions of both brain and intestinal infiltration nature of plasticizers because of the accuracy, speed, conceptual simplicity and for the clear graphical output of BOILED-Egg. The Brain OrIntestinaLEstimateD (BOILED)-Egg permeation method is a predictive model that works with accuracy by computing the polarity and lipophilicity of small molecules and generates clear graphical outputs48. It is freely accessible from the Swiss ADME (http://www.swissadme.ch/). This tool is used to estimate various stages of a drug discovery process. Two pharmacokinetic characteristics play a crucial role, i.e., the prediction of gastrointestinal passive absorption and the permeability of the BBB40. According to BOILED-Egg plot analysis, compounds found in the yellow region were considered to have higher blood–brain barrier permeability, whereas compounds found in the white region of the plot were considered to have higher gastrointestinal absorption properties. The BOILED-Egg plot analysis was performed using the SwissADME web server48. The BOILED-Egg permeation method is a predictive model for the estimation of two pharmacokinetic behaviors, i.e., gastrointestinal passive absorption and the permeability of the blood–brain penetration barrier. It works with speed, accuracy, and conceptual ease by computing the polarity and lipophilicity of chemicals and generates clear graphical outputs48.
Results and discussion
The LAZAR tool prediction for mutagenicity of tested plasticizers against the Salmonella typhimurium model indicates that the following bisphenols BPADE, BPAP, BPBP, BPF, BPM, and BPPH provide mutagenic effects. All other tested compounds did not show mutagenic properties (see S.Table 3). According to the published record, structural analogues of BPA, such as BPAF, BZP, BPS including BPF, do not provide mutagenic effects on the Salmonella typhimurium strains TA98 and TA10044, using the Ames assay for their evaluation of mutagenicity.
From the results of acute toxicity predictions on bees using the KNN/IRFMN v.1.0.0. model accessed through the VEGA version 1.1.4., DAP, DBP, DIHXP, and DPP were predicted as strong bee toxicants; DEP, DMP, BPADE, BPAF, BPC, BPF, and TBPA were moderately toxic and other test compounds were mildly toxic to bees (S.Table 4). For DBP there were no chronic toxicity data available from honey bees. However, during the present computational study, DBP was a strong bee toxicant predicted by the VEGA software. Based on predicted results of fish acute toxicity, Sarpy/IRFMN-1.0.2 classifies the toxic compounds in toxic classes 1, 2, 3 etc. For example, if an LC50 value was less than 1 mg/L, such a compound was classified as a class 1 toxicant. BBP, BCP, BDP, DEHP, DBP, DCP, DIDP, DIHPP, DIHXP, DINP, DIOP, DITP, DIUP, DMP, DNHP, DNPP, DPHP, DTDP, DUP, ODP, DEHP, and DNOP were class 1 toxicants in the fish acute toxicity model. BPM, BPP, BPPH, and TBPA were class 2 toxicants to fish. Other tested compounds were classified as class 3 toxicants according to the Sarpy/IRFMN-1.0.2 fish acute toxicity model (S.Table 5). Fish acute toxicity of BBP in shiner perch (Cymatogaster aggregate) may affect its central adrenergic nervous system45.
Carp (Cyprinus carpio) exposed to DBP and DEHP separately and combined exposure of DBP and DEHP are affected and these two plasticizers provide an ecological risk36. The PROTOX-II web server predicted BCP, DEP, DIBP, DIHPP, DIHXP, DNHP, DNPP, DPP, BPADE, BPG, and TBPA as least toxic from all compounds tested in the present study. BBP, DBP, BPAF, BPM, and BPPH were predicted as class 5 toxicants and other tested compounds were predicted as class 4 toxicants during rodent oral toxicity testing (S.Table 6). Carcinogenicity of plasticizers was predicted by using ensemble learning methods of CarcinoPred-EL46. It predicted BBP, BCP, BDP, DBP, DEHP, DCP, DIDP, DIHPP, DIHXP, DINP, DIOP, DITP, DIUP, DNHP, DNPP, DPHP, DTDP, DUP, ODP, BPADE, DEHP, and DNOP as carcinogens in at least one of the CarcinoPred-EL ensemble models. This tool was developed based on animal experimental data. All other tested compounds were non-carcinogenic.
Whereas carcinogenicity of DBP was not studied in humans as yet, BBP was predicted as carcinogenic in humans47 (S.Table 7). However, based on Toxnet data, DBP is a liver carcinogen for rodents. According to the CarcinoPred-EL tool, DBP was predicted as a carcinogen. DEHP may induce cancer in humans and rodents through multiple molecular signaling pathways48. Carcinogenic effects were not conclusively determined for DNO. But, CarcinoPred-EL predicted DNOP as a carcinogen with the RF and XGBoost model which were developed using animal experimental data. The carcinogen predictive tool was not developed from human experimental data. Data were derived from cancer inducing experiments with animal models. Surely, are tumors not only species- but even organ- and tissue-specific. Their modes of development or prevention, and even their diagnosis through appropriate biomarkers is expected to be very different from the distinctions above.
The plasticizer DEHP alone can provide a P-gp substrate (PGP+) (Points located within BOILED-Egg's yolk PGP+ predicted to be effluating from the Central Nervous System (CNS) by the P-glycoprotein) (Fig. 2); for more details see also S.Fig. 2. But all other test compounds were not PGP−. DEHP can get absorbed to the blood stream in the human gastrointestinal tract. Also, DEHP, BPAF, DNOP, and BPS are predicted as HIA (points located in BOILED-Egg's yolk, HIA predicts as to be passively absorbed by the gastrointestinal tract.). These plasticizers are not entering the human blood–brain barrier (BBB) with results provided in Annexure 1 and 2.
Prediction of gastrointestinal absorption and brain penetration of plasticizers, BOILED-EGG accessed through SwissADME (http://www.swissadme.ch/).
Conclusions
Computational tools provide a fast and cost-effective screening of toxicological characteristics of plasticizers. Based on the LAZAR mutagenicity assessment were bisphenol F alone predicted as mutagenic. The BBP and DBP in RF; DEHP in RF and XGBoost; DNOP in RF and XGBoost models were predicted as carcinogenic in the CarcinoPred-EL web application. From the bee predictive model (KNN/IRFMN) BPF, di-n-propyl phthalate, diallyl phthalate, dibutyl phthalate, and diisohexyl phthalate were predicted as strong bee toxicants. Acute toxicity for fish using the model Sarpy/IRFMN predicted 19 plasticizers as strong toxicants with LC50 values of less than 1 mg/L. This study provides predicted data of plasticizers on gastrointestinal absorption and other toxicological endpoints. Web-based in silico tools and VEGA were used in this study for the prediction of toxicological properties of plasticizers in order to reduce and replace toxicological in vivo and in vitro experiments and to deliver early alerts for environmental and public health risks. The web tools used here turned out to be fast and reliable and were open access.
Data availability
Most data generated or analyzed during this study are included in this published article [and its ANNEX information files]. The complete datasets used and/or analyzed during the current study is available from the corresponding author upon reasonable request.
References
Braga, R. C. et al. Pred-skin: A fast and reliable web application to assess skin sensitization effect of chemicals. J. Chem. Inf. Model. 57, 1013–1017 (2017).
Claudio, L. Our food: Packaging and public health (2012).
LaPensee, E. W., Tuttle, T. R., Fox, S. R. & Ben-Jonathan, N. Bisphenol A at low nanomolar doses confers chemoresistance in estrogen receptor-α-positive and -negative breast cancer cells. Environ. Health Perspect. 117, 175–180 (2009).
Thompson, R. C., Moore, C. J., VomSaal, F. S. & Swan, S. H. Plastics, the environment and human health: Current consensus and future trends. Philos. Trans. R. Soc. B Biol. Sci. 364, 2153–2166 (2009).
Rochester, J. R. & Bolden, A. L. Bisphenol S and F: A systematic review and comparison of the hormonal activity of bisphenol A substitutes. Environ. Health Perspect. 123, 643–650 (2015).
Jenkins, S. et al. Oral exposure to bisphenol A increases dimethylbenzanthracene-induced mammary cancer in rats. Environ. Health Perspect. 117, 910–915 (2009).
Benjamin, S., Pradeep, S., Sarath Josh, M., Kumar, S. & Masai, E. A monograph on the remediation of hazardous phthalates. J. Hazard. Mater. 298, 58–72 (2015).
Fiege, H. et al. Phenol derivatives. Ullmann’s Encycl. Ind. Chem. (Wiley, 2000).
Hassan, Z. K. et al. Bisphenol A induces hepatotoxicity through oxidative stress in a rat model. Oxid. Med. Cell. Longev. 2012, 194829 (2012).
Richter, C. A. et al. In vivo effects of bisphenol A in laboratory rodent studies. Reprod. Toxicol. 24, 199–224 (2007).
Moon, M. K. et al. Bisphenol A impairs mitochondrial function in the liver at doses below the no observed adverse effect level. J. Korean Med. Sci. 27, 644–652 (2012).
Xing, L. et al. Embryotoxic and teratogenic effects of the combination of bisphenol A and genistein on in vitro cultured postimplantation rat embryos. Toxicol. Sci. 115, 577–588 (2010).
Vandenberg, L. N., Hauser, R., Marcus, M., Olea, N. & Welshons, W. V. Human exposure to bisphenol A (BPA). Reprod. Toxicol. 24, 139–177 (2007).
Niu, Y., Zhang, J., Wu, Y. & Shao, B. Analysis of bisphenol A and alkylphenols in cereals by automated on-line solid-phase extraction and liquid chromatography tandem mass spectrometry. J. Agric. Food Chem. 60, 6116–6122 (2012).
Pupo, M. et al. Bisphenol A induces gene expression changes and proliferative effects through GPER in breast cancer cells and cancer-associated fibroblasts. Environ. Health Perspect. 120, 1177–1182 (2012).
Durando, M. et al. Prenatal bisphenol A exposure induces preneoplastic lesions in the mammary gland in Wistar rats. Environ. Health Perspect. 115, 80–86 (2007).
Clark, E. Sulfolane and sulfones. Kirk‐Othmer Encycl. Chem. Technol. (2000).
Liao, C. & Kannan, K. Concentrations and profiles of bisphenol A and other bisphenol analogues in foodstuffs from the United States and their implications for human exposure. J. Agric. Food Chem. 61, 4655–4662 (2013).
Liao, C. & Kannan, K. A survey of alkylphenols, bisphenols, and triclosan in personal care products from China and the United States. Arch. Environ. Contam. Toxicol. 67, 50–59 (2014).
Coggon, D. Work with pesticides and organophosphate sheep dips. Occup. Med. (Chic. Ill.) 52, 467–470 (2002).
Howard, G. J. Chemical alternatives assessment: The case of flame retardants. Chemosphere 116, 112–117 (2014).
Bergman, Å. et al. A novel abbreviation standard for organobromine, organochlorine, and organophosphorus flame retardants and some characteristics of the chemicals. Environ. Int. 49, 57–82 (2012).
Seachrist, D. D. et al. A review of the carcinogenic potential of bisphenol A. Reprod. Toxicol. 59, 167–182 (2016).
Gao, Y., An, T., Ji, Y., Li, G. & Zhao, C. Eco-toxicity and human estrogenic exposure risks from OH-initiated photochemical transformation of four phthalates in water: A computational study. Environ. Pollut. 206, 510–517 (2015).
Worth, A., Lapenna, S., Lo Piparo, E., Mostrag-Szlichtyng, A. & Serafimova, R. A framework for assessing in silico toxicity predictions: Case studies with selected pesticides. JRC Rep. EUR 24705 (2011).
Xu, C. et al. In silico prediction of chemical Ames mutagenicity. J. Chem. Inf. Model. 52, 2840–2847 (2012).
Evans, M. J. & Moore, J. S. A collaborative, wiki-based organic chemistry project incorporating free chemistry software on the Web. J. Chem. Educ. 88, 764–768 (2011).
Öztürk, H., Ozkirimli, E. & Özgür, A. A comparative study of SMILES-based compound similarity functions for drug-target interaction prediction. BMC Bioinform. 17, 1–11 (2016).
Modi, S. et al. Integrated in silico approaches for the prediction of Ames test mutagenicity. J. Comput. Aided. Mol. Des. 26, 1017–1033 (2012).
Maunz, A. et al. Lazar: A modular predictive toxicology framework. Front. Pharmacol. 4, 3830 (2013).
Helma, C. Lazy structure-activity relationships (lazar) for the prediction of rodent carcinogenicity and Salmonella mutagenicity. Mol Divers. 10(2), 147–58 (2006).
Słoczyńska, K., Powroźnik, B., Pękala, E & Waszkielewicz, A. M. Antimutagenic compounds and their possible mechanisms of action. J Appl Genet. 55(2), 273–85 (2014).
Bhattacharya, S. Consumer Attitude towards Green Marketing in India. The IUP Journal of Marketing Management 31, 62–70 (2011).
Maunz, A. & Helma, C. Prediction of chemical toxicity with local support vector regression and activity-specific kernels. SAR QSAR Environ. Res. 19, 413–431 (2008).
Como, F. et al. Predicting acute contact toxicity of pesticides in honeybees (Apis mellifera) through a k-nearest neighbor model. Chemosphere 166, 438–444 (2017).
Zhao, X., Gao, Y. & Qi, M. Toxicity of phthalate esters exposure to carp (Cyprinus carpio) and antioxidant response by biomarker. Ecotoxicology 23, 626–632 (2014).
Banerjee, P., Eckert, A. O., Schrey, A. K. & Preissner, R. ProTox-II: A webserver for the prediction of toxicity of chemicals. Nucleic Acids Res. 46, W257–W263 (2018).
Drwal, M. N., Banerjee, P., Dunkel, M., Wettig, M. R. & Preissner, R. ProTox: a web server for the in silico prediction of rodent oral toxicity. Nucleic acids research 42(W1) (2014).
Tian, B., Wu, N., Pan, X., Yan, C., Sharma, V. K. & Qu, R. Ferrate (VI) oxidation of bisphenol E: Kinetics, removal performance, and dihydroxylation mechanism. Water Research p.118025 (2015).
Riyad, P., Purohit, A., Karishma, S. & Ram, H. Atherosclerotic plaque regression and HMG-CoA reductase inhibition potential of curcumin: An integrative omics and in-vivo study. J. Appl. Biol. Biotechnol 10(1), 129–135 (2022).
Zhang, L. et al. CarcinoPred-EL: Novel models for predicting the carcinogenicity of chemicals using molecular fingerprints and ensemble learning methods. Sci. Rep. 7, 1–14 (2017).
Nanni, L., Fantozzi, C. & Lazzarini, N. Coupling different methods for overcoming the class imbalance problem. Neurocomputing 158, 48–61 (2015).
Maltarollo, V. G., Gertrudes, J. C., Oliveira, P. R. & Honorio, K. M. Applying machine learning techniques for ADME-Tox prediction: a review. Expert opinion on drug metabolism & toxicology, 11(2), 259–271 (2015).
Fic, A., SollnerDolenc, M., Filipič, M. & PeterlinMašić, L. Mutagenicity and DNA damage of bisphenol A and its structural analogues in HepG2 cells. Arh. Hig. Rada Toksikol. 64, 189–199 (2013).
Ozretich, R. J., Randall, R. C., Boese, B. L., Schroeder, W. P. & Smith, J. R. Acute toxicity of butylbenzyl phthalate to shiner perch (Cymatogaster aggregata). Arch. Environ. Contam. Toxicol. 12, 655–660 (1983).
Hsieh, J. C. Y. & Sun, M. Evidence on the Carcinogenicity of Butyl Benzyl Phthalate (BBP). in Meeting of the Carcinogen Identification Committee (Citeseer, 2013).
Caldwell, J. C. DEHP: Genotoxicity and potential carcinogenic mechanisms—A review. Mutat. Res. Mutat. Res. 751, 82–157 (2012).
Daina, A. & Zoete, V. A boiled-egg to predict gastrointestinal absorption and brain penetration of small molecules. ChemMedChem 11(11), 1117 (2016).
Acknowledgements
Marvin version 17.21.0, ChemAxon was used for drawing, displaying and to obtain SMILES of chemical structures. CDA acknowledges a Kaohsiung Medical University International Students Grant. HUD acknowledges the support of grants from MOST (MOST 108-2621-M-037-001, MOST 109-2621-M-037-001, and MOST 110-2621-M-037-001 to T.H. Shih). This work was financially supported by grants from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE); from Ministry of Science and Technology, Taiwan (MOST 111-2314-B-037-004-;110-23144-B-037-047-MY3; from National Health Research Institutes, Taiwan (NHRI-EX111-10703PI). The financial support from the Ministry of Science and Technology of Taiwan (Grant Nos. MOST 107-2621-M-019-001, MOST 108-2621-M019-003, MOST 109-2621-M019-002 and MOST 110-2621-M-019-001) and Center of Excellence for Ocean Engineering (Grant No. 109J3801-51, 110J13801-51) to J.-S. Hwang. This study was supported by grants from Taiwan's Ministry of Science and Technology (MOST111-2314-B-037-004- and MOST110-2314-B-037-047-MY3); from Taiwan's National Health Research Institutes (NHRI-EX111-10703PI); and from the Research Center for Precision Environmental Medicine, Kaohsiung Medical University, Kaohsiung, Taiwan under The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan. The funding organizations played no role in study design, data collection and analysis, preparation of the manuscript, or decision to publish.
Author information
Authors and Affiliations
Contributions
C.D.A., A.J.R. and H.U.D. wrote the manuscript and prepared the figures; J.S.H. and H.U.D. supervised the project, made funding available, and reviewed the draft. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Arulanandam, C.D., Hwang, JS., Rathinam, A.J. et al. Evaluating different web applications to assess the toxicity of plasticizers. Sci Rep 12, 19684 (2022). https://doi.org/10.1038/s41598-022-18327-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-18327-0
This article is cited by
-
Toxicity prediction and analysis of flavonoid apigenin as a histone deacetylase inhibitor: an in-silico approach
In Silico Pharmacology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.