Abstract
Morus alba is one of the most important cultivated crop in Indian sub-continent contributing towards production of silk fibre that carries economic importance worldwide. Post harvest preservation of M. alba leaves is a challenging factor as decrease in concentration of essential metabolites that needed for silk gland development takes place. Decrease in chlorophyll, protein, sugar concentration and increase in accumulation of free radicals and ROS takes place at post harvest stage of preservation, putting negative impact on larval development indicated by high mortality rate. Silver nitrate and nanosilver solution acts as an effective preservative, enhances the activity of enzymatic and non-enzymatic antioxidants thereby reducing the harmful effect of accumulated free radicals and ROS. The effectiveness of nanosilver solution was found to be on the upper site without any significant difference than silver nitrate, as higher retention of primary metabolites like pigments, proteins, and sugar takes place. The impact of feeding nanosilver preserved leaves on silkworm was found on the positive trend as larval growth rate, cocoon weight, shell weight, effective rate of rearing was observed almost same to the larvae fed with fresh leaves.
Similar content being viewed by others
Introduction
Morus alba an economically important crop, placed by Bentham and Hooker under the family Moraceae. M. alba leaves are used for feeding monophagous insect silkworm, which produces raw silk contributing towards world economy. Development of silk gland and production of quality silk depends upon leaf protein content. Indeed larvae act as an intermediate agent converting leaf protein into silk fibre1. Deviation in leaves nutrient quality puts significant impact over larval growth and cocoon formation2.
Silk industry was an agro-based labour intensive industry which generates high employment among rural peoples3. Practice of silkworm rearing consists of two important activities: (1) cultivation of M. alba, for fresh leaves and (2) rearing of silkworm. Rearing is an indoor practice while cultivation of M. alba leaves is an outdoor practice, requiring an open land. Thus rearing practice remains restricted to those farmers those bears marginal to small scale lands4. Landless farmers generally migrate from one garden to another or move to urban areas in search of work leaving the traditional practice. Some farmers even purchase leaves from others gardens4 but regular purchasing of leaves and its carrying cost increases the overall cost of production. Preservation by retaining the leaf quality on purchasing the leaves once in a while may serve as a solution to this problem.
Wilting, discolouration, senescence, high respiration rate, decay and microbial growth are the main cause which limits postharvest extension of shelf life. Fresh leaves are live and are subjected to environmental and physiological variables leading to changes in preharvest and postharvest quality5,6. Microbial proliferation causes rapid senescence and degradation of macromolecules by rising ROS and free radical percentage7. Decolouration by chlorophyll degradation resulting in yellowing of leaves is the most conspicuous indicative phenomenon of leaf senescence7. It has been reported that many internal and external factors are generally involved in leaf chlorophyll preservation8,9. In presence of light, leaf chloroplast was the major site for ROS generation and during senescence disassembly of photosynthetic apparatus causes disturbance of reducing equivalents of electron chain resulting enhancement in ROS accumulation10. Excessive ROS accumulation causes cellular damage, evident by degradation of pigment, proteins, lipids, carbohydrates and even nucleic acid11. To overcome oxidative injury, plant causes detoxification of excess generated ROS by activating enzymatic and non-enzymatic enzymological activities12. ROS scavenging activity was triggered by the activity of the enzymes like superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPOX), ascorbate peroxidase (APX) which works in a coordinated fashion converting superoxide (O2•−) to hydrogen peroxide (H2O2) and finally to H2O13,14. Glutathione, ascorbic acid, carotenoids are mainly involved in non-enzymatic antioxidant activities. Glutathione helps in the generation of ascorbic acid which detoxify O2•− inside chloroplast15, whereas carotenoids protects photosynthetic apparatus from stress mediated damages16. Phenolics are also classified as non-enzymatic antioxidants which nullifies the toxic effect of free radicals17.
With implementation of stress, auto-activation of defensive enzymatic and non-enzymatic molecules takes place. But at post harvest stage with increase in days of preservation these defensive activities decreases. Presence of elicitor or preservative may prolong the shelf life by up-regulating the defensive pathways. Silver nitrate (SN) was the most applied silver salt as preservative in the field of horticultural crop, as they inhibit microbial proliferation, preventing vascular occlusion18. The impact of SN in prolonging vase life of rose19, tuberose20 has been well documented. It has been reported that there might be the possibility of SN causing toxicity to living organism21, putting a backward thrust towards its application. Nanosilver (NS) may serve as an alternate option as it has lowest toxic effect than any other silver forms22. In comparison to ionic form of silver, nanosilver have highly developed surface area making it more reactive, besides this its physico-chemical property allows to interact with living cell differentially23. Nanosilver at low concentration may act as an effective preservative which not only prevents microbial growth24,25, maintaining xylem integrity but also activates protective enzymological activities26. Implementation of NS as preservatives has been reported to extend the shelf life of Dianthus27, Chrysanthemum28, tulip29, Gerbera30. However most of the preservative aspects of SN and NS have been reported to extend shelf life of economically important flowering twigs, almost no report was obtained that describes their ability to preserve postharvest leaf samples.
Current study was conducted assuming the hypothesis that biosynthesized silver nanoparticles will bear the ability to extend the shelf life of S1 cultivar of M. alba leaves at postharvest stage. In the current study three attempts were made, first to investigate the effect of NS as preservative solution, in extending shelf life of M. alba leaves by retaining valuable metabolite concentration; secondly a comparative aspect of two preservative solutions, NS and SN; and lastly whether there is any adverse effect of feeding preserved leaves on silkworm rearing system.
Results
Synthesis of silver nanoparticles and its characterization
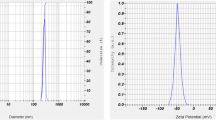
Initial confirmation of nanosilver formation was obtained by observing the colour change of the solution from transparent to blackish brown. Confirmatory validation was done through UV–Visible spectrophotometer which showed plasmon peak at ~441 nm. Synthesized nanoparticles were spherical in shape with size distribution range from 12–39 nm, as revealed by TEM analysis. FT-IR spectra showed almost similar peak positions of biosynthesized nanosilver and plant extract used for biosynthesis. The FT-IR absorption peak at 3422.98, 2929.71, 1625.90, 1382.88, 1053.07 and 921.92 cm−1 corresponds to –OH stress of phenols and alcohols and N–H vibrational stretching, –CH2 and –CH3 vibration, C–N vibration of amide I band, C-N vibrational stress, C–O– and C–OH vibration of protein and carbohydrate, and C–C bond of branch alkanes respectively. Four major 2θ peaks at 38.312°, 44.402°, 64.579°, 77.547° were obtained from X-ray diffraction pattern which corresponds to (hkl) values of (111), (200), (220), (311) of Bragg’s reflection plane. Electron diffraction (SAED) pattern showed bright diffraction spots of hkl value corresponds to (111), (200), (220), and (311) (Fig. 1).
Changes in primary metabolites and proline content
With increase in days of preservation, the concentration of total chlorophyll, total soluble protein, total and reducing sugar decreases and that of free proline content increases (Fig. 2). In distilled water set a greater increase in proline content and greater decrease in content of total soluble protein and other primary metabolites was observed. Leaves preserved in SN and NS solution showed a lower magnitude of increase in free proline content and less depletion in soluble protein and other primary metabolites. Leaves preserved in NS solution exhibited greater retention of primary metabolites than distilled water set (p ≤ 0.05).
Effect of preservation of S1 genotype of M. alba leaves with distilled water, nanosilver and silver nitrate on (A) total chlorophyll, (B) total protein, (C,D) total and reducing sugar, and (E) proline content. Effect of preservative solutions on M. alba leaves was observed at regular interval of 0D, 1D, 4D, 6D and 7D. The results were expressed as Mean ± SDEV, n = 3.
Changes in reactive oxygen species and MDA content
The contents of H2O2, O2•−, and MDA increased rapidly in distilled water set than in leaves preserved in NS and SN solution (Fig. 3). The magnitude of increase of these substances in leaves preserved in NS solution showed significant negative difference in comparison to distilled water set (p ≤ 0.05). Overall increase in ROS and MDA content was higher in leaves preserved in SN solution than NS solution but lesser than distilled water without any significant difference (p ≤ 0.05) except for H2O2, which exhibited large significant (p ≤ 0.05) increase in distilled water set. Increase in ROS and MDA content with increase in days of preservation bears negative correlation with primary metabolite content (p ≤ 0.01) and positive correlation with proline and antioxidant activity (p ≤ 0.01 and 0.05) (Fig. 4).
Effect of preservation of S1 genotype of M. alba leaves with distilled water, nanosilver and silver nitrate on (A) hydrogen peroxide, (B) superoxide, and (C) malondialdehyde content. Effect of preservative solutions on stress content of M. alba leaves was observed at regular interval of 0D, 1D, 4D, 6D and 7D. The results were expressed as Mean ± SDEV, n = 3.
Two tailed Pearson correlation representing interrelationship among the post-preservative quality detecting parameters and rearing parameters. Significance level was indicated by “**” and “*” indicating correlation was significant at the 0.01 and 0.05 level (2-tailed) respectively. The abbreviation used means the following: TC= total chlorophyll, TP=total protein, PRL= proline, TS= total sugar, RS= reducing sugar, H2O2= hydrogen peroxide, SOC= superoxide content, MDA= malondialdehyde, SOS= superoxide scavenging, NO= nitricoxide, MC= metal chelating, CAT= catalase, GSR= glutathione disulfide reductase, GPOX= glutathione peroxidase, GST= glutathione S-transferase, APX= ascorbate peroxidase, SOD= superoxide dismutase, CAR= carotenoids, AA= ascorbic acid, TG= total glutathione content, PHNO= phenol, OP= ortho-phenol, FLV= flavonoid, GI= growth index, SCW= single cocoon weight, SSW= single shell weight, SR= shell ratio, ERR= effective rate of rearing, MR= mortality rate.
Changes in free radical scavenging activities, and metal chelating activity
With increase in days of preservation free radical scavenging activity increased in leaves preserved with NS and SN solution as revealed by decreased in IC50 values, while reverse trend was observed in distilled water set (Fig. 5). In distilled water set high magnitude of decrease in DPPH and SO scavenging activity was observed from 4DS onwards. Changes in DPPH scavenging activity with increase in days of preservation in NS and SN solution differs significantly with the observed changes in distilled water set (p ≤ 0.05). Significant difference was also observed for ABTS scavenging activity between leaves preserved in NS solution as compared to distilled water preservation (p ≤ 0.05). The correlation matrix showed negative significant correlation (p ≤ 0.01 and 0.05) between free radical scavenging activity and primary metabolite content, but the correlation was actually positive because free radical scavenging activity was expressed in terms of IC50. Leaves preserved in distilled water showed greater metal chelating activity than leaves preserved in NS and SN solution without any significant difference.
Effect of preservation of S1 genotype of M. alba leaves with distilled water, nanosilver and silver nitrate on (A) diphenyl-1-picrylhydrazyl, (B) 2,2-azino-bis 3-ethylbenzthiazoline-6-sulphonic acid, (C) superoxide, (D) nitric oxide, and (E) metal chelating activity. Effect of preservative solutions on antioxidant activity of M. alba leaves was observed at regular interval of 0D, 1D, 4D, 6D and 7D. Smaller the IC50 value more was the antioxidant activity. The results were expressed as Mean ± SDEV, n = 3.
Changes in enzymatic antioxidant activities
As days of preservation increases, an escalating trend in enzymatic antioxidant activity was observed (Fig. 6). A rapid increase in GSR, GST, and APX activity was observed in distilled water set up to 4DS, after which gradual decrease in activity takes place, except for GPOX which maintained a linear increasing trend till last day. While SOD activity showed declining trend from day one of preservation. Antioxidant enzymatic activity of leaves preserved in NS and SN solution increases gradually, reaching a maximum at 6DS, after which drop in activity in CAT, and GST was observed for SN. Broadly, antioxidant enzymatic activity in leaves preserved in NS and SN solution increases with days of preservation without any significant difference (p ≤ 0.05) between the two preservatives. Correlation matrix reveals that with increase in concentration of secondary metabolites, increase in enzymatic antioxidant activity takes place (p ≤ 0.01 and 0.05).
Effect of preservation of S1 genotype of M. alba leaves with distilled water, nanosilver and silver nitrate on (A) superoxide dismutase, (B) catalase, (C) ascorbate peroxidase, (D) glutathione S-transferase, (E) glutathione peroxidase, and (F) glutathione disulfide reductase content. Effect of preservative solutions on antioxidant enzymatic activity of M. alba leaves was observed at regular interval of 0D, 1D, 4D, 6D and 7D. Larger the content, greater was the defensive activity, less was the stress accumulation. The results were expressed as Mean ± SDEV, n = 3.
Changes in non-enzymatic antioxidant activities
An increase in content of total glutathione was observed from early days, in leaves preserved in NS, reaching a maximum on 6DS, after which rapid decline takes place (Fig. 7). In distilled water set decrease in content of total glutathione was observed after 4DS. Leaves in NS and SN solution showed significant (p ≤ 0.05) higher ascorbic acid content than distilled water set, where decrease content takes place from 1DS of preservation. Decrease in carotenoids content was observed from 4DS onwards in all the preservative solutions, with maximum magnitude of decrease in distilled water followed by SN. Leaves preserved in NS showed significant (p ≤ 0.05) retention of carotenoids with increase in days of preservation in relation to distilled water preservation. Increase in ascorbic acid activity in preserved leaves plays a significant (p ≤ 0.01) role in retention of chlorophyll, protein, and sugar content as revealed from correlation matrix.
Effect of preservation of S1 genotype of M. alba leaves with distilled water, nanosilver and silver nitrate on (A) carotenoids, (B) ascorbic acid, and (C) total glutathione content. Effect of preservative solutions on non-enzymatic antioxidant activity of M. alba leaves was observed at regular interval of 0D, 1D, 4D, 6D and 7D. The results were expressed as Mean ± SDEV, n = 3.
Changes in polyphenol contents
The concentration of polyphenol increases with increase in days of preservation (Fig. 8). Significant difference (p ≤ 0.05) in phenol content was observed between initial and final day of preservation. In NS and SN set increase in phenol content was observed till last day, with greater increase in NS than SN, while in distilled water set decrease in content was observed on the last day. Almost similar trend of changes was noticed for ortho-dihydric phenol content without any significant difference (p ≤ 0.05) between preservative solutions. In distilled water set a greater increase in flavonoid content takes place till 4DS, after which a greater decrease was noted, while in NS and SN set increasing trend was observed till last day without any significant difference (p ≤ 0.05).
Effect of preservation of S1 genotype of M. alba leaves with distilled water, nanosilver and silver nitrate on (A) total phenol, (B) ortho-dihydric phenol, and (C) flavonoid content. Effect of preservative solutions on secondary metabolite content of M. alba leaves was observed at regular interval of 0D, 1D, 4D, 6D and 7D. The results were expressed as Mean ± SDEV, n = 3.
Changes in rearing data collection
A significant decrease in growth index was observed when fifth instar larvae was fed with leaves preserved in distilled water (Fig. 9). Decrease in GI reaches up to ~71% when fed with 7DS preserved leaves. On feeding leaves preserved with NS and SN decrease in GI was negligible in comparison with fresh leaves, with maximum decrease takes place on feeding 7DS preserved leaves which was ~2% and ~6% respectively. Significant (p ≤ 0.05) difference was observed on comparing GI of larvae fed with leaves preserved in NS and distilled water. It was observed from correlation analysis that greater the retention of primary metabolite (p ≤ 0.01) along with lesser the accumulation of free radicals (p ≤ 0.01) in leaves better was the GI.
Effect of feeding S1 genotype of M. alba leaves, preserved with distilled water, nanosilver and silver nitrate on (A) growth index, (B) single cocoon weight, (C) single shell weight, (D) shell ratio, (E) effective rearing rate, and (F) mortality rate. Silkworm larvae were fed with leaves preserved for 0D, 1D, 4D, 6D and 7D. The results were expressed as Mean ± SDEV, n = 3.
After the larvae stops spinning, a significant decrease in SCW and SSW was observed from the larvae fed with 4DS, 6DS and 7DS distilled water preserved leaves, with maximum decrease on feeding 7DS preserved leaves ~78% and ~88% respectively. Larvae fed with leaves preserved in NS and SN showed significant (p ≤ 0.05) retention of SCW and SSW. More over larvae fed with leaves preserved in NS solution showed higher retention of SCW and SSW than larvae fed with leaves preserved in SN solution. Significant (p ≤ 0.01) positive relationship was observed between the concentration of chlorophyll, protein, sugar present in the leaves and cocoon produced by the larvae. Shell ratio of larvae fed with leaves preserved in NS solution was observed to be more elevated than larvae fed with leaves preserved in SN and distilled water solution.
The ERR of larvae depends on the amount of stress imposed on leaves with increase in days of preservation in different preservative solutions. It was observed that reduction in ERR was less in larvae fed with leaves preserved in NS solution, with maximum reduction of ~7%. Consequently larvae fed with leaves preserved in distilled water and SN solution showed maximum reduction of 92% and 24% respectively.
A gradual increase in mortality rate was observed on feeding larvae with leaves preserved with distilled water, with maximum mortality of ~93% on feeding with 7DS leaves. Consequently larvae fed with leaves preserved in NS and SN solution showed high survival rate, with maximum mortality of ~13% and ~30% respectively on feeding 7DS preserved leaves.
Discussion
The prime challenge faced during leaf preservation was the gradual appearance of senescence with increase in days of preservation. Senescence was primarily characterized by breakdown of photosynthetic pigments resulting in gradual reduction in photosynthetic rate and photosynthesis: respiration ratio31. Beside this, differences which were observed on feeding senescence leaves include low growth index, decrease in cocoon and shell weight, and high larval mortality. Several senescence retardation mechanisms were adopted by the leaves and it’s up and down regulation in presence of silver nitrate and nanosilver was evaluated for determining their efficacy. In presence of silver nitrate and nanosilver up-regulation of enzymatic and non-enzymatic antioxidant activity and decreasing ROS content was observed which eventually resulted in extension of post harvest shelf life.
Our result indicated significant decrease in chlorophyll content in distilled water preserved leaves with increase in days of preservation. Decrease in chlorophyll content directly links with less chlorophyll biosynthesis8 due to the activation of the enzyme chlorophyllase32. We observed significant negative correlation (p ≤ 0.01) between chlorophyll degradation and generation of ROS. With increase in days of preservation, excessive accumulation of O2•− and H2O2, generates oxidative stress. The major site for ROS generation in leaves was chloroplast33. During senescence photosynthetic efficiency decreases, as a consequence CO2 fixation reduces and ROS generation increases using the unutilized energy of the chloroplast34. It was reported that senescence causes chloroplast isolation and generation of O2•− through reduction of O2 via photosynthetic electron transport chain35 and consequently by the action of the enzyme superoxide dismutase, the generated O2•− get converted into H2O236. Generated ROS was capable of causing oxidative damage to proteins, carbohydrates, lipids, and nucleic acids, while excessive ROS accumulation leads to programme cell death37,38. O2•−, H2O2 generation in our study showed direct correlation with the increase in content of MDA. MDA is considered as an indicator of stress39 associated with cell and organelle damage40. Multiple regression analysis of positive and negative correlated parameters associated with chlorophyll (Table 1) content indicated the major involvement of MDA during stress in chlorophyll degradation. Our outcome was supported by the earlier works, indicating increase in lipid peroxidation causes degradation of chlorophyll41,42. Rajinder et al.43 reported that decrease in chlorophyll content during leaf senescence was due to the lipid peroxidation of chloroplast membrane. However leaves preserved in NS solution showed significant (p ≤ 0.05) retention of chlorophyll content than distilled water set. Our result suggest that NS and SN both acts as an effective preservative by preventing ROS generation, lipid peroxidation and retention of chlorophyll content. In comparison to distilled water, SN showed high efficacy in nullifying generation H2O2 (p ≤ 0.05), while NS was proven to be more effective preservative showing its potential in checking O2•−, and H2O2 generation, and lipid peroxidation (p ≤ 0.05). Prevention of ROS generation helps in preventing plastid membrane peroxidation and thus maintaining chlorophyll content, extending the shelf life.
Leaves preserved in NS and SN displays appropriate oxidative stress tolerance strategy by enhancing antioxidant enzymological activity. SOD converts O2•− into H2O213, present study demonstrated gradual increase in SOD in leaves preserved with NS solution, while reversed trend was observed in distilled water preserved leaves. CAT, APX and GPOX are primarily synthesized by the plant for scavenging excess H2O2 produced by the activity of SOD14. Current study showed an increasing trend of CAT, APX and GPOX activity with increase in days of preservation in NS and SN set. While CAT activity increases in leaves preserved in NS and SN up to 6DS, in distilled water preserved leaves CAT activity fails to enhance during preservation, indicating its inability to nullify the toxic effect of H2O2. Similar results were obtained by application of nitrogenous compound on rose and Chrysanthemum cut flower9,31. Moreover CAT activity showed direct correlation with chlorophyll content (p ≤ 0.05), indicating H2O2 detoxification by CAT activity helps in chlorophyll retention with increase in days of preservation. APX and GPOX activity also showed increasing trend till the last day of preservation in NS and SN set. In distilled water set APX activity increases up to 4DS after which rapid decrease in activity was noted, while GPOX activity was much higher than leaves preserved in NS and SN solution till the last day. It has been reported that if CAT fails to nullify H2O2, APX and GPOX activity increases as a compensatory mechanism14. In distilled water set as CAT activity fails to rise during preservation so probably by increasing APX and GPOX activity leaves tried to detoxify generated H2O2. Obtained result suggest that in distilled water preserved leaves APX fails to detoxify H2O2 from 4DS onwards after which only GPOX plays the role as a single protecting agent against H2O2. GSR and GST activity shoot up in distilled water set till 4DS indicating high stress accumulation which the leaves tries to nullify by activating its protective measures. GST was reported to counteract the different abiotic damage caused during stress44 and also protects cells against oxygen toxicity45. On the contrary GSR was a flavo-protein oxidoreductase which plays crucial role in reducing biotic stress by maintaining ascorbate and glutathione pool in reduced state inside the chloroplast46. GSR and GST activity gradually increases in leaves preserved with NS solution till the last day indicating the potential of NS solution in strengthening the biological tissue for withstanding both abiotic and biotic stress.
A high increase in glutathione concentration in leaves preserved with NS solution was observed from 4DS till 6DS after which decrease in concentration takes place while no such high increase takes place for leaves preserved in SN solution but a gradual increasing trend was maintained till last day, reflecting an attempt in minimizing oxidative stress. Leaves in distilled water set fails to increase the concentration of glutathione and thus faces early senescence due to accumulation of oxidative stress. Glutathione was a low molecular weight non-protein thiol that performs multiple functions in biosynthetic pathways, detoxification and antioxidant activities which the other thiols cannot perform47. The non-enzymatic antioxidant activity of glutathione resides in its ability to generate another non-enzymatic antioxidant ascorbic acid, by participating in ascorbate – glutathione cycle48. Ascorbic acid, was reported to exist mostly in reduced state inside chloroplast49 and can directly participate in scavenging O2•− and H2O2 via APX activity15. Our study reports increased concentration of ascorbic acid in leaves preserved in NS and SN solution indicating their ability to detoxify free radicals by involving APX, reported earlier and thereby enhancing postharvest shelf life. In addition to the pool of ascorbate and glutathione, leaves in NS solution also maintained almost steady pool of carotenoids which bears significant difference with decreasing content in distilled water set (p ≤ 0.05). Carotenoids belong to the class of lipophilic antioxidants, synthesized in plastids and thus might play role in preventing ROS accumulation in chloroplast16.
Besides ascorbic acid, glutathione and carotenoids, phenolics (total phenol, orthodihydric phenol and flavonoid) are another group of non-enzymatic antioxidant actively participates in stress shielding mechanism. The prime defensive function of polyphenols resides in lipid free radical scavenging activity48. Polyphenols by complexing metal ions prevents metal ion mediated conversion of O2•− and H2O2 in to highly reactive hydroxyl radical and thereby preventing DNA damage50 and thus allowing continuous synthesis of compounds through enzymatic mechanism that are involved in enhancing shelf life. Our study reflects almost similar trend as describe above, with increase in days of preservation phenol and orthodihydric phenol content increases in leaves present in preservative solution reflecting there potentiality to overcome stress. Result of distilled water preserved leaves also reflects an increasing trend but the extent of increase in comparison to NS and SN was much less. Flavonoid content does not show any significant change with increasing days, however in distilled water set from 4DS onward a decreasing trend was observed explaining inability to overcome generated stress.
Free radical scavenging and metal chelating activity was measured for determining the potential of secondary metabolites in preventing stress caused by free radicals and metal ions, described above. It was observed that with increase in days of preservation ABTS+, DPPH, SO and NO scavenging activity increases in leaves preserved in NS and SN solution, on contradictory in distilled water set their activity decreases. Biogenic silver nanoparticles were reported to show high activity towards scavenging free radicals51,52 and chelating metal ions53. It seems that the scavenging potential of NS, as well as SN protects leaf tissue from the intracellular harmful effect of free radicals. Because of gradual deterioration at tissue level, secreted secondary metabolites oozes out from leaves preserved in distilled water showing high metal chelating activity than leaves preserved in NS and SN solution.
At post harvest stage, significant (p ≤ 0.05) retention of total protein content was recorded in leaves preserved in NS and SN solution. Retention of total and reducing sugar in NS set was also recorded to be significant (p ≤ 0.05). Post harvest shelf life was effected by number of factors and among them water relationship and balance plays a key role54. Water relation was interrupted mainly due to microbial proliferation, blocking the cut end and thereby preventing water channel55,56. NS was reported to enhance post harvest shelf life of flowering twigs at low concentration by decreasing microbial load29,57. It has also been reported that both SN and NS acts as an effective preservatives, but water conduction rate was high when NS was used as preservative58. Thus NS and SN by retaining the concentration of primary metabolite in preserved leaves to optimum level, allowing the synthesis of defensive enzymes needed to overcome the generated stress. We observed increased level of proline in all the preservative sets indicating activation of ROS scavenging mechanism59 and thereby stabilizing cellular proteins and membrane integrity60.
The production of good quality cocoon crop directly depends on quality of M. alba leaves provided during rearing phase61. Silkworm bears a special ability of converting ingested leaf protein into silk protein. Variation on leaf nutritional level puts significant impact on cocoon production both qualitatively and quantitatively62. Protein and carbohydrate are the two most important constituents of M. alba leaves playing crucial role in silkworm rearing. While leaf protein was essential for larval growth, silk gland development, and cocoon characters, carbohydrate serves as major energy source during accumulation and secretion of silk fibre from silk gland63. Our rearing records suggests that GI of larvae fed with leaves preserved in NS and SN solution remains almost same as that of larvae fed with fresh leaves. While significant (p ≤ 0.05) decrease in GI was observed when fed with leaves of distilled water set. Larval growth depends on protein content of the leaves, and retention of GI indicates retention of protein in leaves preserved in preservative solution. Multiple regression analysis of significantly correlated parameters associated with protein content (Table 2) in preserved leaves indicates greater the superoxide scavenging activity less was the ROS mediated protein oxidation and thus more was the native protein content in the leaves. ERR (%) was best observed in NS preserved leaves and thus indicating high viability and low mortality. Low ERR in larvae fed with distilled water preserved leaves describes their inability for cocoon production and thus high mortality rate. Productivity can be described in terms of three most important commercial parameters viz. cocoon weight, shell weight and shell ratio63. Larvae fed with NS preserved leaves showed best cocoon weight, shell weight and shell ration than larvae fed with leaves preserved with SN and distilled water. Such result was displayed might be due to more ingestion, digestion and conversion ratio of larvae fed with NS preserved leaves, demonstrating the ability of nanosilver solution as an effective preservative.
The above experiments and explanations suggest that silver nanoparticles synthesized using M. alba leaf extract acts as an effective preservative in extending the shelf life of M. alba leaves at postharvest stage. Thus the specification of formed NS becomes an important criterion for leaf preservation. Synthesized NS showed SPR band at ~441 nm confirming the presence of nanosilver, as SPR spectra of silver nanoparticles appears in the wavelength range of 400–500 nm64. The synthesized nanoparticles are spherical in shape with more than 50% particles are in monodispersed phase and average particle size of ~14 nm representing high bioactivity, as bioactivity was inversely proportional to the size of nanoparticles65,66,67. Probably because of these bioactive nature biosynthesized silver nanoparticles able to retain the shelf life of M. alba leaves at postharvest stage. FT–IR spectra showed the involvement of proteins, amino acid, short peptides, carbohydrates and secondary metabolites like phenol as capping, stabilizing and bio-reducing agents68. The deviation in peak position between plant extract and nanosilver was the indication of bioreduction and was another confirming module of nanosilver formation69. From XRD peak at 38.312° corresponds to (111) Bragg’s reflection plane found to be most intensified peak, corresponding to this peak 10.071 nm crystalline size was obtained according to Debye-Scherrer’s equation and inter planar spacing from Bragg’s law was obtained to be 2.347 Å. XRD result also matches with standard JCPDS library file no: 04-0783 confirming face centered cubic structure of NS. Crystalline nature was further validated by SAED pattern, confirming orientation of nanocrystals around (111) plane.
Conclusions
At postharvest stage of preservation, M. alba leaves exhibited decrease in chlorophyll, protein and carbohydrate concentration, accumulation of free radicals including ROS causing oxidative damage, lipid peroxidation and thus imposing changes in rearing parameters in the present study. Leaves preserved in NS and SN solution showed two line of defensive strategy; first the activation and upregulation of antioxidant enzymatic (SOD, CAT, APX, GPOX, GSR, GST) and non-enzymatic antioxidant (ascorbic acid, glutathione and carotenoids) activities; and secondly enhancement in production of secondary metabolites involved in free radical scavenging activity. These metabolic alterations allow the leaves at postharvest stage to withstand and extend shelf life. Leaves preserved in NS solution exhibits greater retention of photosynthetic pigments and other primary metabolites, probably due to greater ability to scavenge ROS both by enzymatic and non-enzymatic pathways. Moreover, feeding leaves preserved in NS solution showed better growth and cocoon parameters, indicating the absence of any adverse toxic effects. Thus it may be concluded that phytosynthesized silver nanoparticles at 6 ppm concentration bears the potentiality to extent the shelf life of Morus alba L. leaves. Our finding may regain the interest of landless farmers towards this wide field of application as they can undergo the process of rearing for some days by purchasing the leaves once.
Materials and Methods
Plant sample and experimental system
The experiment was carried out at Department of Botany, University of North Bengal, India (26°42′44.11″ N and 88°29′40.09″ E) during April – September of 2017–2018 and 2018–2019. The most widely grown and used cultivar of Morus alba in this region, namely S1 cultivar was used as experimental material and were collected from Matigara Sericulture Complex, Siliguri, India (26°70′40″ N and 88°35′37″ E) during daytime between 6 and 7 am, for maintaining fresh texture of leaves. Immature, unhealthy and diseased leaves were screened out for maintaining uniform experimental condition. Leaves were preserved up to 7 days in preservative solutions and during preservation perforated zipper bags were used for retaining moisture content. During experimental process three solutions were used as preservatives viz. phyto-synthesized silver nanoparticles (NS) containing 6 ppm colloidal silver, silver nitrate (SN) solution bearing 6 ppm silver ion which serves as positive control and distilled water serving as negative control; all the solutions were maintained at neutral pH (pH 7). Prior to preservation fine oblique section under water was made to the petiole of the leaves for maintaining intact water column. The experimental setup was kept at 25 °C maintaining light intensity of 260–270 lux and 40% humidity. Preserved leaves were collected at an interval of 1, 4, 6 and 7DS for evaluating the potentiality of preservative solutions in extending shelf life through chemical parameters with respect to fresh leaves.
Phyto-synthesis of silver nanoparticles and its characterization
For phyto-synthesis 5 ml distilled water refluxed M. alba leaf extract (10 g leaf in 100 ml distilled water) was added to 45 ml 10−3 M silver nitrate solution (Sigma Aldrich) with continuous stirring in magnetic stirrer for 10 min. The colour of the solution gradually turns yellowish and finally to blackish brown within 20 min span, indicating nano formation70.
Silver nanoparticles formation was recorded in UV – Vis Spectrophotometer (Systronics 2201) as a function of wavelength ranges between 300–800 nm. The size and shape of nanoparticles was studied through TEM micrographic (Philips CM 200) operating at an accelerating voltage of 120 kV. The possible involvement of functional groups in nano formation was predicted through FT – IR analysis at a wavelength range of 500–4000 nm using potassium bromide pellet technique. The crystallinity of nanoparticles was determined through X-ray diffraction study (Burker ASX D8). The sample was Cu Kα radiated at an operational speed of 5° sec−1, keeping current, voltage, and wavelength constant at 35 mA, 40 kV, and 1.5 Å respectively.
Determination of total chlorophyll, total soluble protein, free proline, total soluble sugar, and reducing sugar concentrations
Total chlorophyll content in preserved leaves was determined by Arnon method71. For extraction, 1 g leaf sample was homogenized with 20 ml of 80% (v/v) acetone and was centrifuged at 10,000 × g for 5 min at 25 °C. Obtained residue was re-extracted twice for complete extraction of pigments. The UV-Vis spectrophotometer (Systronics 2201) reading of the supernatants was taken at 663, and 645 nm for estimating chlorophyll content.
Total protein content was estimated following the method of Lowry et al.72. For extraction 0.5 g frozen leaf samples were crushed using sodium phosphate buffer and centrifuged at 5,000 × g at −10 °C for 5 min. To the supernatant 5 ml alkaline copper solution and 0.5 ml Folin-Ciocalteu reagent was added leading to the development of blue colour complex. The absorbance was measured at 660 nm after 30 min of incubation and was standardized using bovine serum albumin for determining the protein content. Free proline content was estimated according to the method described by Bares et al.73. For extraction 0.5 g leaf sample was homogenized in 10 ml 3% sulfosalicylic acid and estimation was done at 520 nm of the upper toluene layer isolated from the reaction mixture containing 1 ml extract, 2 ml acid ninhydrin, 2 ml glacial acetic acid, and 4 ml toluene.
Total soluble sugar and reducing sugar content was determined spectrophotometrically following the methods of Sadasivam and Manickam74. Using mortar and pestle 0.1 g of leaf samples were extracted twice with 80% hot ethanol, 5 ml each time. The ethanol present in the supernatant was heat evaporated and the final volume was made up to 10 ml by distilled water. Total soluble sugar was determined using anthrone reagent at 620 nm by incubating in a boiling water bath for 8 min. Reducing sugar was estimated at 510 nm using dinitrosalicyclic acid and 40% Rochelle salt solution.
Determination of reactive oxygen species and lipid peroxidation accumulation
Accumulation of reactive oxygen species was evaluated by estimating hydrogen peroxide (H2O2) and superoxide (O2•−) content following the method prescribed by Loreto and Velikova75 and Elstner and Heupel76 respectively. H2O2 was estimated by homogenizing 0.5 g leaf sample in 1% (w/v) tri-chloroacetic acid (TCA) and centrifuging at 10,000 × g for 10 min at 4 °C. The obtained supernatant was spectrophotometrically measured at 390 nm after reacting with 10 mM phosphate buffer (pH 7.0) and 1 M potassium iodide. Superoxide content was estimated by homogenizing 1 g leaf samples in 65 mM potassium phosphate buffer (pH 7.8) and centrifuging at 5,000 × g for 10 min. The supernatant was reacted with 65 mM potassium phosphate buffer (pH 7.8) and 10 mM hydroxylamine hydrochloride, and incubated for 15 min at 25 °C. Then 17 mM sulfanilamide and 7 mM α-anaphthylamine were added and absorbance was recorded at 530 nm. Extend of lipid peroxidation was determined by measuring MDA contents following the method of Davenport et al.77. Leaf samples (0.2 g) were homogenized in 2 ml 0.5% (w/v) TCA and centrifuged at 10,000 × g for 10 min at 4 °C. The reaction mixture containing 2 ml supernatant and 2 ml 0.67% thiobarbituric acid (TBA) was incubated for 30 min at 95 °C, followed by ice water treatment to stop the reaction. The absorbance of the supernatant was recorded at 450, 532, 600 nm and MDA content was calculated using the following formula, MDA (µmol g−1) = [6.45 (A532 – A600) − 0.56 A450] × Vt/W, where Vt = 0.0021 and W = 0.2 g.
Determination of free radical scavenging activities, and metal chelating activity
2,2-diphenyl-1-picrylhydrazyl (DPPH), 2,2-azino-bis 3-ethylbenzthiazoline-6-sulphonic acid (ABTS), superoxide (SO), nitric oxide (NO) scavenging activity, and metal chelating (MC) activity were determined following the method of Sidduraju et al.78, Li et al.79, Fu et al.80, Marcocci et al.81, and Dinis et al.82 respectively. Preserved leaf samples (1 g) were homogenized with 10 ml methanol and centrifuged at 10,000 × g for 10 min at 4 °C. The obtained supernatants were used as crude extract for the above cited assays.
DPPH scavenging activity was measured at 517 nm by using 0.2 ml methanolic extract against 2 ml DPPH. ABTS activity was determined by adding 2 ml ABTS to 1 ml extract and measuring the absorbance at 734 nm after 10 min incubation. For measuring superoxide activity absorbance was measured at 560 nm after 30 min illuminated light (4000 flux) incubation of the reaction mixture containing 1 ml nitroblue tetrazolium chloride, 1 ml nicotinamide adenine dinucleotide and 10 μl phenazine methosulphate. To measure nitric oxide scavenging activity 20 mM sodium nitroprusside, 0.5 ml phosphate buffer, and 3 ml Griess reagent was mixed with 0.5 ml extract and absorbance was measured at 540 nm after 30 min incubation. Metal chelating activity was measured at 562 nm by reacting methanolic extract with 2 mM FeCl3 and 5 mM Ferrozine. Scavenging activity was measured as percent inhibition using the following equation: % inhibition = [(A0 − A1)/A0] 100%, Where A0 and A1 are the absorbance of control and sample respectively. Antioxidant activity was expressed as concentration where 50% reduction in free radical takes place referred to as IC50 value.
Determination of enzymatic, and non-enzymatic antioxidant activities
Enzymatic antioxidant activities were measured in terms of superoxide dismutase (SOD), catalase (CAT), glutathione disulfide reductase (GSR), glutathione peroxidase (GPOX), glutathione S-transferase (GST), and ascorbate peroxidase (APX) activity by following the methods given by Esfandiari et al.83, Hasanuzzaman et al.84, Chen and Asada85. SOD (EC: 1.15.1.1) activity was estimated by adding 0.05 ml enzyme extract to reaction mixture containing 200 mM methionine, 2.25 mM nitro-blue tetrazolium and 3 mM EDTA. Reaction was initiated by adding 60 µM riboflavin, incubated under light for 10 min, and absorbance was taken at 560 nm. Enzyme activity was expressed as quantity of enzyme reducing the absorbance of NBT by 50%. CAT (EC: 1.11.1.6) activity was determined by adding enzyme extract to a mixture containing 50 mM potassium phosphate buffer (pH 7.0), and 15 mM H2O2. The reaction was monitored at 240 nm for 1 min and the enzyme activity was expressed as unit (1unit = mmole of H2O2 reduced min−1 mg protein−1) using extinction coefficient of 39.4 M−1 cm−1. GSR (EC: 1.6.4.2) activity was recorded at 340 nm for 1 min by adding 1 mM glutathione disulfide to the reaction mixture containing 0.1 M potassium phosphate buffer (pH 7.8), 1 mM EDTA, 0.2 mM NADPH, and enzyme solution; the enzyme activity was expressed as unit (1unit = µmol (NADPH) min−1 mg protein−1) using extinction coefficient of 6.2 mM−1 cm−1. For measuring GPOX (EC: 1.11.1.9) activity reaction mixture contains 100 mM sodium phosphate buffer (pH 7.5), 1 mM EDTA, 1 mM sodium azide, 0.2 mM NADPH, 2 mM glutathione, 1 unit glutathione reductase, 0.6 mM H2O2, and 20 µl crude enzyme. Enzyme activity (unit) was recorded through oxidation of NADPH at 340 nm for 1 min and was calculated as µmol (NADPH) min−1 mg protein−1 using extinction coefficient of 6.62 mM−1 cm−1. GST (EC: 2.5.1.18) activity was expressed using extinction coefficient of 9.6 mM−1 cm−1 at 340 nm for 1 min from the reaction mixture containing enzyme solution in 100 mM tris-HCl (pH 6.5), 1.5 mM glutathione, and 1 mM 1-chloro-2,4-dinitrobenzene (CDNB). APX (EC: 1.11.1.11) activity was determined at 290 nm for 2 min by adding enzyme extract to 3 ml reaction mixture containing 50 mM potassium phosphate buffer (pH 7.0), 0.5 mM ascorbate, and 0.1 mM H2O2. One unit of APX was defined as amount of enzyme required to consume 1µmol of ascorbate min−1 mg protein−1.
Carotenoids content was determined by homogenizing 1 g samples in 80% acetone and centrifuging at 10,000 × g for 5 min at 25 °C. The absorbance of the supernatant was determined spectrophotometrically at 470, 645, and 663 nm86. For ascorbic acid determination, 0.5 g leaf samples were homogenized in 10% (w/v) trichloroacetic acid and centrifuged at 10,000 × g for 20 min at 25 °C. The obtained supernatant (0.5 ml) was incubated at 37 °C for 3 h after reacting with 2% 2, 4-dinitrophenyl hydrazine in 0.5 N H2SO4, and 10% thiourea. After incubation spectral reading was recorded at 520 nm87. Total glutathione content was determined by reacting 100 µl extract with 0.3 mM NADPH in 20 mM potassium phosphate buffer (pH 7.5) and 6 mM 5′-dithio-bis(2-nitrobenzoic acid). The reaction mixture was incubated at 25 °C for 3 min, and then 10 µl reduced glutathione was added, after the colour was developed absorbance was recorded at 412 nm88.
Determination of polyphenol contents
For total phenol, ortho-dihydric phenol, and flavonoid estimation, 1 g leaf sample was ground with 80% ethanol and centrifuged at 10,000 × g for 20 min. The residue was re-extracted twice using 80% ethanol. The obtained supernatant was evaporated to dryness and the residue was dissolved with distilled water. Total phenol content was determined at 650 nm by adding 50% folin-ciocalteu reagent and 20% sodium carbonate to the extract following the method of Malick and Singh89, using gallic acid as standard. Orthodihydric phenol content was determined at 515 nm by adding 0.05 (N) HCl, Arnow’s reagent and 1 (N) NaOH to the aqueous extract following the method of Mahadevan and Sridhar90 using catechol as standards. Flavonoid content was determined at 510 nm following the method of Atanassova et al.91 by adding 5% NaNO2, 10% AlCl3 and 1 (M) NaOH to the aqueous extract and using quercetin as standard.
Feeding experiment and rearing data collection
The overall rearing process was conducted under laboratory condition following the standard process of Krishnaswami et al.92. For rearing purpose healthy and disease free 5th instar larvae (Nistari) were collected from Matigara Sericulture Complex. The larvae were distributed uniformly and randomly in bamboo made trays, each tray bearing 10 larvae. Larvae were supplemented with S1 genotype M. alba leaves. In one set freshly collected S1 leaves were given for feeding, while in other sets 1, 4, 6, and 7DS preserved leaves in distilled water, silver nitrate and nanosilver solution were given for feeding. During rearing process larval weight and mortality rate was recorded at regular interval. When the larvae started spinning they were left undisturbed. At the end of the trial, larval and cocoon parameters were calculated using following formulas:
Data analysis
Using General Linear Model, two-way analysis of variance (ANOVA) was performed to calculate the effect of preservative solutions on preservation of M. alba leaves and on silk worm rearing (P ≤ 0.05) using SPSS statistical package (IBM SPSS Advanced Statistics 20.0). Bivariate correlations study using Pearson correlation coefficient was done to study interrelationship between different leaf metabolic parameters with rearing parameters (p ≤ 0.01 and 0.05) using SPSS. Multiple regression analysis was conducted using SPSS for determining the stress parameters that are involved in degradation of major primary metabolites. Figures were plotted using OriginPro 2018b software (b9.5.5.409).
References
Prabu, G. P. et al. Effect of food supplementation with silver nanoparticles (AgNps) on feed efficacy of silkworm, Bombyx mori (L.) (Lepidoptera: Bombycidae). Int. J. Res. Biol. Sci. 2(2), 60–67 (2012).
Mahmood, K., Ahmad, M. & Gilani, A. H. Effect of feeding leaves of Morus alba and Morus leavigata on larval growth and silk yield of silkworm, Bombyx mori. Pak. J. Zool. 19, 239–243 (1987).
Kumar, H., Priya, Y. S. & Kumar, M. & Elangovan. Effect of different mulberry varieties and seasons on growth and economic traits of bivoltin silkworm (Bombyx mori). J. Entomol. 10(3), 147–155 (2013).
Sarkar, K., Majumdar, M. & Ghosh, A. Critical analysis on role of women in sericulture industry. Int. J. Soc. Sci. 6(3), 211–222 (2017).
Aghaye Noroozlo, Y., Souri, M. K. & Delshad, M. Effects of Soil Application of Amino Acids, Ammonium, and Nitrate on Nutrient Accumulation and Growth Characteristics of Sweet Basil. Commun. Soil Sci. Plant Ana.l 50(22), 2864–2872 (2019).
Mohammadipour, N. & Souri, M. K. 2019. Beneficial effects of glycine on growth and leaf nutrient concentrations of coriander (Coriandrum sativum) plants. J. Plant Nutr. 42(14), 1637–1644 (2019).
Merzlyak, M. N. & Hendry, G. A. F. Free-radical metabolism, pigment degradation and lipid peroxidation in leaves during senescence. Proc. R. Soc. Edinburgh B. 102, 459–471 (1994).
Souri, M. K. & Hatamian, M. Aminochelates in plant nutrition; a review. J. Plant Nutr. 42(1), 67–78 (2019).
Hatamian, M. & Souri, M. K. Postharvest Quality of Roses under Different Levels of Nitrogenous Compounds in Holding Solution. Open Agric. 4(1), 79–85 (2019).
Juvany, M., Müller, M. & Munné-Bosch, S. Photo-oxidative stress in emerging and senescing leaves: a mirror image. J. Exp. Bot. 64, 3087–3098 (2013).
Das, K. & Roychoudhury, A. Reactive oxygen species (ROS) and response of antioxidants as ROS-scavengers during environmental stress in plants. Front. Environ. Sci. https://doi.org/10.3389/fenvs.2014.00053 (2014).
Sharma, P., Jha, A. B., Dubey, R. S. & Pessarakli, M. Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J. Bot. https://doi.org/10.1155/2012/217037 (2012).
Ighodaro, O. M. & Akinloye, O. A. First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid. Alexandria J. Med. 54, 287–293 (2018).
Apel, K. & Hirt, H. Reactive oxygen species: Metabolism, oxidative stress, and signal transduction. Ann. Rev. Plant Biol. 55, 373–399 (2004).
Noctor, G. & Foyer, C. H. Ascorbate and glutathione keeping active oxygen under control. Annu. Rev. Plant Physiol. Plant. Mol. Biol. 49, 249–279 (1998).
Racchi, M. L. Antioxidant Defenses in Plants with Attention to Prunus and Citrus spp. Antioxidants 2, 340–369 (2013).
Rice-Evans, C. A., Miller, N. J. & Paganga, G. Antioxidant properties of phenolic compounds. Trends Plant. Sci. 2, 152–159 (1997).
Elgimabi, M. E. N. E. Vase Life extension of rose cut flowers (Rosa Hybirida) as influenced by silver nitrate and sucrose pulsing. Amer J. Agri. Biol. Sci. 6(1), 128–133 (2011).
Hussen, S. & Yassin, H. Review on the impact of different vase solutions on the postharvest life of rose flower. Inter. J. Agri. Res. Rev. 1(2), 13–17 (2013).
Anjum, M. A., Naveed, F., Shakeel, F. & Amin, S. Effect of some chemicals on keeping quality and vase- life of tuberose (Polianthes tuberosa L.) cut flowers. J. Res. (Sci) 12(1), 01–07 (2001).
Ratte, H. T. Bioaccumulation and toxicity of silver compounds: a review. Environ. Sci. Tech. 18, 89–108 (1999).
Foldbjerg, R. et al. PVP coated silver nanoparticles and silver ions induce reactive oxygen species, apoptosis and necrosis in THP-1monocytes. Toxicol Lett. 190, 156–162 (2009).
Kȩdziora, A. et al. Similarities and differences between silver ions and silver in nanoforms as antibacterial agents. Int. J. Mol. Sci. https://doi.org/10.3390/ijms19020444 (2018).
Naing, A. H., Win, N. M., Han, J.S., Lim, K.B. & Kim, C. K. Role of nano-silver and the bacterial strain enterobacter cloacae in increasing vase life of cut carnation ‘Omea’. Front. Plant Sci. https://doi.org/10.3389/fpls.2017.01590 (2017).
Sánchez-López, E., et al. Metal-based nanoparticles as antimicrobial agents: an overview. Nanomater. https://doi.org/10.3390/nano10020292 (2020).
Hatami, M. & Ghorbanpour, M. Defence enzyme activities and biochemical variations of Pelargonium zonale in response to nanosilver application and dark storage. Turkish J. Biol. 38, 130–139 (2014).
Solgi, M. The application of new environmentally friendly compounds on postharvest characteristics of cut carnation (Dianthus caryophyllus L.). Brazilian Journal of Botany https://doi.org/10.1007/s40415-018-0464-x (2018).
Carrillo-López, L. M., Morgado-González, A., Morgado-González, A. Biosynthesized silver nanoparticles used in preservative solutions for Chrysanthemum cv. Puma. J. Nanomater. https://doi.org/10.1155/2016/1769250 (2016).
Byczyńska, A. Improvement of postharvest quality of cut tulip ‘White Parrot’ by nano silver. World Scientific News 83, 224–228 (2017).
Kazemi, M. & Ameri, A. Postharvest life of cut gerbera flowers as affected by nano-silver and acetlysalicyclic acid. Asian J. Biochem. 7(2), 106–111 (2012).
Souri, M. K., Goodarzizadeh, S., Ahmadi, M. & Hatamian, M. Characteristics of postharvest quality of chrysanthemum cut flower under pretreatment with nitrogenous compounds. Acta Sci. Pol. Hortorum Cultus 17(3), 83–90 (2018).
Santos, C. V. Regulation of chlorophyll biosynthesis and degradation by salt stress in sunflower leaves. Scientia Horticulturae 103, 93–99 (2004).
Ishida, H., Izumi, M., Wada, S. & Makino, A. Roles of autophagy in chloroplast recycling. Biochim. Biophys. Acta. 1837, 512–521 (2014).
Xie, Q., Michaeli, S., Peled-Zehavi, H. & Galili, G. Chloroplast degradation: one organelle, multiple degradation pathways. Trends Plant Sci. 20, 264–265 (2015).
Allen, J. F. & Hall, D. O. Superoxide reduction as a mechanism of ascorbate-stimulated oxygen uptake by isolated chloroplasts. Biochem. Biophys. Res. Commun. 52, 856–862 (1973).
Asada, K. Production and scavenging of reactive oxygen species in chloroplasts and their functions. Plant Physiol. 141, 391–396 (2006).
Ismail, S. Z., Khandaker, M. M., Mat, N. & Boyce, A. N. Effects of hydrogen peroxide on growth, development and quality of fruits: A Review. J. Agro. 14(4), 331–336 (2015).
Anjum, N. A. et al. Lipids and proteins-major targets of oxidative modifications in abiotic stressed plants. Environ. Sci. Pollut. Res. 22, 4099–4121 (2015).
Hatamian, M., Rezaie Nejad, A., Kafi, M., Souri, M. K. & Shahbazi, K. Growth characteristics of ornamental Judas tree (Cercis siliquastrum L.) seedling under different concentrations of lead and cadmium in irrigation water. Acta Sci. Pol. Hortorum Cultus 18(2), 87–96 (2019).
Wang, H. et al. Involvement of ethylene and hydrogen peroxide in induction of alternative respiratory pathway in salt-treated Arabidopsis calluses. Plant Cell Physiol. 51(10), 1754–1765 (2010).
Orthoefer, F. T. Jr. & Dugan, L. R. The coupled oxidation of chlorophyll a with linoleic acid catalyzed by lipoxidase. J. Sci. Food Agr. 24, 357–365 (1973).
Imamura, M. & Shimizu, S. Metabolism of chlorophyll in higher plants. IV. Relationship between fatty acid oxidation and chlorophyll bleaching in plant extracts. Plant Cell Physiol. 15, 187–190 (1974).
Rajinder, S. D., Pamela, P. D. & Trevor, A. T. Leaf senescence: correlated with increased levels of membrane permeability and lipid peroxidation, and decreased levels of superoxide dismutase and catalase. J. Exp. Bot. 32(126), 93–101 (1980).
Ding, N. et al. Identification and analysis of glutathione S-transferase gene family in sweet potato reveal divergent GST-mediated networks in aboveground and underground tissues in response to abiotic stresses. BMC Plant Biol. https://doi.org/10.1186/s12870-017-1179-z (2017).
Mannervik, B. & Danielson, U. H. Glutathione transferases-structure and catalytic activity. CRC Crit. Rev. Biochem. 23, 283–337 (1988).
Edwards, E. A., Rawsthome, S. & Mullineaux, P. M. Subcellular distribution of multiple forms of glutathione reductase in leaves of pea (Pisum sativum L.). Planta 180, 278–284 (1990).
Noctor, G. et al. Glutathione in plants: an integrated overview. Plant Cell Environ. 35, 454–484 (2012).
Blokhina, O., Virolainen, E. & Fagerstedt, K. V. Antioxidant, oxidative damage and oxygen deprivation stress: a review. Ann. Bot. 91, 179–194 (2003).
Sminnof, N. The function and metabolism of ascorbic acid in plants. Ann. Bot. 78, 661–669 (1996).
Perron, N. R. & Brumaghin, J. L. A review of the antioxidant mechanisms of polyphenol compounds related to iron binding. Cell Biochem. Biophys. 53, 75–100 (2009).
Sundararajan, B., Mahendran, G., Thamaraiselv, R. & Kumari, B. D. R. Biological activities of synthesized silver nanoparticles from Cardiospermum halicacabum L. Bull. Mater. Sci. 39(2), 423–431 (2016).
Phull, A. R. et al. Antioxidant, cytotoxic and antimicrobial activities of green synthesized silver nanoparticles from crude extract of Bergenia ciliate. Future J. Pharm. Sci. 2, 31–36 (2016).
Madhanraj, R., Eyini, M. & Balaji, P. Antioxidant assay of gold and silver nanoparticles from edible basidiomycetes mushroom fungi. Free Radic. Antioxid. 7(2), 137–142 (2017).
Jowkar, M. M., Khalighi, A., Kafi, M. & Hassanzadeh, N. Nano silver application impact as vase solution biocide on postharvest microbial and physiological properties of ‘Cherry Brandy’ rose. J. Food Agri. Environ. 11(1), 1045–1050 (2013).
Van Doorn, W. G. Water relations of cut flowers. Horticultural Reviews 18, 1–85 (1997).
Liu, J. et al. Nano-silver pulse treatments inhibit stem-end bacteria on cut Gerbera cv. Ruikou flowers. Postharvest Biol. Technol. 54, 59–62 (2009).
Mazumdar, H. The impact of silver nanoparticles on plant biomass and chlorophyll content. Int. J. Eng. Sci. 4(7), 12–20 (2014).
Hatami, M., Hatamzadeh, A., Ghasemnezhad, M. & Ghorbanpour, M. The comparison of antimicrobial effects of silver nanoparticles (SNP) and silver nitrate (AgNO3) to extend the vase life of ‘Red Ribbon’ cut rose flowers. Trakia J. Sci. 2, 144–151 (2013).
Hayat, S. et al. Role of proline under changing environments. Plant Signal Behav. 7(11), 1456–1466 (2012).
Matysik, J. B., Bhal, B. & Mohanty, P. Molecular mechanisms of quenching of reactive oxygen species by proline under stress in plants. Curr. Sci. 82, 525–532 (2002).
Bothikar, P. A., Jadhav, S. S. & Shinde, Y. A. Growth and development of silk worm (Bombyx mori L.) on mulberry leaves. J. Indus Poll. Cont. 30(2), 239–241 (2014).
Gawade, B. V. & Medhe, N. K. Feeding effect of some mulberry varieties on larval weight of silkworm, Bombyx mori L. Green Farming 1(5), 545–546 (2010).
Sajgotra, M., Bali, R. K. & Gupta, V. Influence of brushing schedule on leaf biochemical analysis for commercial character expression of bivoltine silkworm hybrids (Bombyx mori L.). J. Pharm. Phytochem. 6(5), 437–442 (2017).
Sastry, M., Mayya, K. S. & Bandyopadhyay, K. pH dependent changes in the optical properties of carboxylic acid derivatized silver colloidal particles. Colloids Surf. A. 127, 221–228 (1997).
Agnihotri, S., Mukherji, S. & Mukherji, S. Size-controlled silver nanoparticles synthesized over the range 5–100 nm using the same protocol and their antibacterial efficacy. RSC Adv. 4, 3974–3983 (2014).
Zielińska, A. et al. Development and optimization of alpha-pinene-loaded solid lipid nanoparticles (SLN) using experimental factorial design and dispersion analysis. Molecules. 24, https://doi.org/10.3390/molecules24152683 (2019).
Campos, J. R.,et al. Solid lipid nanoparticles (SLN): prediction of toxicity, metabolism, fate and physicochemical properties. Nanopharmaceuticals. https://doi.org/10.1016/B978-0-12-817778-5.00001-4
Elamawi, R. M., Al-Harbi, R. E., Awatif, A. & Hendi, A. A. Biosynthesis and characterization of silver nanoparticles using Trichoderma longibrachiatum and their effect on phytopathogenic fungi. Egypt. J. Biol. Pest. Co. https://doi.org/10.1186/s41938-018-0028-1 (2018).
Pirtarighat, S., Ghannadnia, M. & Baghshahi, S. Green synthesis of silver nanoparticles using the plant extract of Salvia spinosa grown in vitro and their antibacterial activity assessment. J. Nanostructure Chem., https://doi.org/10.1007/s40097-018-0291-4
Singh, K., Naidoo, Y., Mocktar, C., & Himansu Baijnath, H. Biosynthesis of silver nanoparticles using Plumbago auriculata leaf and calyx extracts and evaluation of their antimicrobial activities. Adv. Nat. Sci. Nanosci. Nanotechnol., https://doi.org/10.1088/2043-6254/aad1a3 (2018).
Arnon, D. I. Estimation of chlorophyll (DMSO). Plant Physiol. 24, 1–15 (1949).
Lowry, O. H., Rosebrough, N. J., Farr, A. L. & Randall, R. J. Protein measurement with the folin phenol reagent. J. Biol. Chem. 193, 265 (1951).
Bates, L. S., Walderen, R. P. & Teare, I. D. Rapid determination of free proline for water stress studies. Plant Soil 39, 205–207 (1973).
Sadasivam, S. & Manickam, A. Biochemical Methods (Second Ed.) New Age International (P) Ltd. and Tamil Nadu Agricultural University, Coimbatore (1996).
Loreto, F. & Velikova, V. Isoprene produced by leaves protects the photosynthetic apparatus against ozone damage, quenches ozone products, and reduces lipid peroxidation of cellular membranes. Plant Physiol. 127, 1781–1787 (2001).
Elstner, E. F. & Heupel, A. Inhibition of nitrite formation from hydroxylammonium- chloride: a simple assay for superoxide dismutase. Anal Biochem. 70, 616–620 (1976).
Davenport, S. B., Gallego, S. M., Benavides, M. P. & Tomaro, M. L. Behaviour of antioxidant defense system in the adaptive response to salt stress in Helianthus annuus L. cells. Plant Growth Regul. 40, 81–88 (2003).
Sidduraju, P., Mohan, P. & Becker, K. Studies on the antioxidant activity of Indian Laburnum Cassia fistula L: a preliminary assessment of crude extracts from stem bark, leaves, flowers and fruit pulp. Food Chem. 79, 61–67 (2002).
Li, X. C., Wu, X. T. & Huang, L. Correlation between antioxidant activities and phenolic contents of radix Angelicae sinensis (Danggui). Molecules 14, 5349–5361 (2009).
Fu, W. et al. Antioxidant, free radical scavenging, anti- inflammatory and Hepatoprotective potential of the extract from Parathelypteris nipponica (Franch. Et Sav.) Ching. J. Ethnopharmacol. 130, 521–528 (2010).
Marcocci, L., Packer, L., Droy-Lefaix, M. T., Sekaki, A. & Grades-Albert, M. Antioxidant action of Ginkgo biloba extracts EGb 761. Methods Enzymol. 234, 462–475 (1994).
Dinis, T. C. P., Madeira, V. M. & Almeida, L. M. Action of phenolic derivates (acetoaminophen, salicylate and 5-aminosalycilate) as inhibitors of membrane lipid peroxidation and as peroxyl radical scavengers. Arch. Biochem. Biophys. 315(1), 161–169 (1994).
Esfandiari, E., Shekari, F., Shekari, F. and Esfandiari, M. The effect of salt stress on antioxidant enzymes, activity and lipid peroxidation on the wheat seedling. Not. Bot. Hot. Agrobot. Cluj., https://doi.org/10.15835/nbha.35.1.251 (2007).
Hasanuzzaman, M., Hossain, M. A. & Fujita, M. Nitric oxide modulates antioxidant defence and the methylglyoxal detoxification system and reduces salinity-induced damage of wheat seedlings. Plant Biotechnol. Rep. 5, 353–365 (2011).
Chen, G. X. & Asada, K. Ascorbate peroxidase in tea leaves: Occurrence of two isoenzymes and the differences in their enzymatic and molecular properties. Plant cell Physiol. 30, 987–998 (1989).
Lichtenthaler, H. K. Chlorophylls and carotenolds: pigments of photosynthesis. Methods Enzymol. 148, 350–352 (1987).
Omaye, S. T., Turnbull, J. D. & Sauberilich, H. E. Selected methods for the determination of ascorbic acid in animal cells, tissues and fluids. Methods Enzymol. 62, 3–11 (1979).
Griffith, O. W. & Meister, A. Potent and specific inhibition of glutathione synthesis by buthionine sulfoximine (s-n-butylhomocysteine sulfoximine). J. Biol. Chem. 254, 7558–7560 (1979).
Malick, C. P. & Singh, M. B. In: Plants Enzymology and Histoenzymology. Kalyani Publications, New Delhi (1980).
Mahadevan, A. & Sridhar, R. Methods in physiological plant pathology. Sivakami Publication, Chennai (1986).
Atanassova, M., Georgieva, S. & Ivancheva, K. Total phenolic and total flavonoid contents, antioxidant capacity and biological contaminants in medicinal herbs. J. Chem. Technol. Metall. 46(1), 81–88 (2011).
Krishnaswami, S., Narasimhana, M. N., Suryanarayana, S. K. & Kumararaj, S. Sericulture Manual – II: Silk Worm Rearing. Food and Agriculture Organization of the United Nation. 131–786 (1978).
Acknowledgements
The authors would like to thank Directorate of Textiles (Sericulture), Matigara Sericulture Complex for providing necessary M. alba leaves and silkworm larvae during experiment. The author would also like to thank SAIF, IIT Bombay for assisting while conducting different instrument analysis. The first author would like to thank University Grants Commission as the author receives UGC-NET JRF Fellowship.
Author information
Authors and Affiliations
Contributions
Das, D. has conducted the study, collected samples, analyzed the data, software analysis and primarily drafted the manuscript. Mandal, P. conceptualized the work, helped in reviewing and revising the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Das, D., Mandal, P. Use of biogenic silver nanoparticles in enhancing shelf life of Morus alba L. at post harvest stage. Sci Rep 10, 8923 (2020). https://doi.org/10.1038/s41598-020-65953-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-65953-7
This article is cited by
-
Application of Si and Ag Green Nanoparticles, Epibrassinolide, and Methyl Jasmonate Causes Delay in Decay of Malus Domestica Fruits via Improving Postharvest Physiology at Ambient Conditions
Agricultural Research (2024)
-
Transcriptome analysis of mulberry (Morus alba L.) leaves to identify differentially expressed genes associated with post-harvest shelf-life elongation
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.