Abstract
We investigated the association between age at menarche, and adiposity and blood pressure in adulthood, as well as the mediation effects of adiposity on the relationship between age at menarche and blood pressure. This was a nationally representative survey conducted in Brazil in 2013 (Brazilian Health Survey). The participants included 33,715 women between 18 and 100 years old. Age at menarche was self-reported and outcomes (body mass index [BMI], waist circumference, and blood pressure) were objectively measured. As covariates, data on chronological age, race, educational status, tobacco smoking, leisure physical activity, TV viewing, sodium consumption, alcohol drinking, menopause status, and antihypertensive medication use were obtained by questionnaires. Negative and linear relationships were observed between age at menarche and adiposity indicators. Early maturers presented a greater prevalence of obesity (waist circumference: 56.3% [54.0–58.7] vs. 52.4% [51.4–53.5], BMI: 32.4% [30.2–34.6] vs. 25.1% [24.2–26.0]) than on time/late maturers. Age at menarche was significantly inversely related to blood pressure and adiposity in adulthood (p < 0.05), although these associations were weaker than the association between obesity and early menarche. Adiposity indicators mediated the relationship between age at menarche, and systolic (partly) and diastolic (fully) blood pressure. In conclusion, early maturation is related to obesity and higher blood pressure in adulthood. Obesity is an important mediator of the influence of early maturation on high blood pressure in women.
Similar content being viewed by others
Introduction
Chronic diseases have emerged as the leading cause of death in recent decades [1]. Among chronic diseases, hypertension is one of the ten main isolated causes of death worldwide [2]. The large global disease burden of hypertension is due directly to its high prevalence and considerable mortality risk and indirectly through its influence on other associated cardiovascular diseases (e.g., chronic kidney disease, ischemic heart disease, and cerebrovascular diseases) [3]. In Brazil, an estimated 265 thousand people died of hypertension in 2015 [3].
The etiology of hypertension is complex. Multilevel factors, such as race, chronological age, stress, physical activity level, and dietary patterns, have influence on the onset and development of hypertension [4,5,6]. However, one of the strongest proximal risk factors of hypertension is overweight/obesity [4, 5]. Thus, increased body adiposity (especially centrally located obesity) represents a higher risk for the development of hypertension.
In addition to the traditional determinants, early biological maturation has been related to obesity and high blood pressure (BP) in adolescents [7]. However, it is not clear whether changes in biological processes in the early years can have impact on cardiovascular and hemodynamic outcomes across the life span [8,9,10,11,12]. As obesity is directly associated with biological maturation, it has been speculated that the relationship between biological maturation and cardiometabolic risk factors can be mediated by obesity [9, 12, 13]. Although speculative, the mediating role of adiposity on the effect of early maturation on BP in adulthood has not been tested. However, this is especially important given that the onset of biological maturation, especially age at menarche, now occurs at a much earlier age than in previous decades [14]. Moreover, understanding the mediating effect of adiposity on the relationship between early maturation and BP in adulthood could help identify women subgroups that are at risk (based on adiposity status) and those that should be targeted for early intervention. Thus, our aim was to investigate the associations between biological maturation (based on age at menarche) and adiposity and BP in adulthood and to examine whether adiposity mediates the associations between age at menarche and BP in adulthood.
Materials and methods
Sample
The Brazilian Health Survey (PNS in Portuguese) is a nationally representative cross-sectional epidemiological survey that was conducted in 2013. The survey employed a complex sampling design. Census tracts were first randomly selected from a national plan, households were then randomly selected from a national registry of addresses, and finally, within each household, one randomly selected adult 18 years or older was invited to participate in the study. Interviews were eventually conducted in 64,348 households, which represented an overall response rate of 78%. Person-level survey weights were applied to account for the probability of selection as well as non-response rates [15]. For the present analyses, only data from female participants were explored. Overall, 34,282 women participated in the PNS, but 567 women were excluded due to missing data. Therefore, our final sample size comprised 33,715 women between 18 and 100 years of age. The Brazilian national council of ethics in research approved all procedures, which were in accordance with the Helsinki Declaration.
Age at menarche
Age at menarche was used as an indicator of sexual maturation, as it marks the final stage of sexual maturation [16]. The assessment was conducted through self-report with participants recalling their age at menarche retrospectively. For the descriptive analysis, age at menarche was categorized as ≤ 11, 12, 13, 14, or ≥ 15 years, and continuous data for actual age at menarche were used in the mediation models.
Obesity indicators
Waist circumference (WC) and body mass index (BMI) were adopted as obesity (adiposity) indicators. For measurements, a portable stadiometer, a portable electronic weighing machine and an anthropometric tape were used. All body adiposity measurements were conducted by trained staff. Obesity indicators were categorized according to standard cut-points: WC > 88 cm and BMI ≥ 30 kg/m2 [17].
Blood pressure
The measurement of BP was carried out by trained staff using calibrated digital equipment (TECH® model MA 100). Initially, participants rested for 10 min and then three measurements were taken. Before these measurements, all the participants were instructed to empty their bladders and not to smoke or drink for at least 30 min before the measurements. The measurements were taken with the individual in a seated position and with the right arm supported by a table. The average of the second and third measurements was used for the present study. The adopted cut-point for high systolic BP was ≥ 140 mm Hg and for high diastolic BP was ≥ 90 mm Hg. More detailed information was previously published [18].
Covariates
Participants reported their own chronological age in years and details of their highest education level were used to create three education categories (no academic qualification, high school, and more than high school). Race was self-reported and the information was dichotomized (white and non-white). Leisure-time physical activity was assessed by asking participants if they had (no/yes) performed any sports or exercise in the preceding 3 months and how many days per week, and hours and minutes per day they usually performed sports or physical exercise. Participants who engaged in ≥ 150 min of physical activity per week (weekly frequency multiplied by daily time) were classified as active [19]. Participants further reported how many days per week they usually smoked tobacco; participants were classified as smokers if they engaged in the behavior ≥ 1 day weekly. With regards to sodium consumption, a five-point Likert scale was used to assess whether participants perceived their diet was characterized by “a little” to “a lot” of salt (diets containing “quite a lot” and “a lot” were considered to be indicative of high salt intake). In addition, participants reported how many days per week they usually consumed alcohol and their responses were categorized as follows: none, moderate (1–3 days per week), and heavy (4 or more days per week). Daily TV viewing was self-reported by participants in response to the question “How many hours a day do you usually spend watching TV?” One of the following responses was permitted: (a) Less than 1 h; (b) More than 1 h but less than 2 h; (c) More than 2 h but less than 3 h; (d) More than 3 h but less than 4 h; (e) More than 4 h but less than 5 h; (f) More than 5 h but less than 6 h; (g) More than 6 h; or (h) I do not watch TV. The data were used to create three categories of daily TV viewing time (less than 2 h; between 2 h and 4 h; and more than 4 h). The participants also reported if they had already entered menopause and if they used any antihypertensive medication. Responses to these questions were treated as dichotomous variables (no or yes). All covariates were treated as controlled variables in the main analyses.
Statistical analysis
Means and 95% confidence intervals were used to describe the sample characteristics. Values with non-overlapping confidence intervals were considered significantly different [20, 21]. Pearson’s correlations and adjusted linear regression analyses were used to assess the relationships between age at menarche and adiposity indicators (BMI and WC) and BP (systolic and diastolic BP). In the regression analysis, model 1 tested the association of age at menarche with BP, adjusted for chronological age, race, menopause status, antihypertensive medication use, education status, smoking status, leisure physical activity, TV viewing, and alcohol consumption. Model 2 tested the association of WC with BP, including all the covariates that were adjusted for in model 1, whereas model 3 tested the association between BMI and BP, including all the covariates that were adjusted for in models 1 and 2. As women usually gain weight after menopause, the correlation and regression analyses were conducted overall and stratified by menopause status (i.e., before menopause and after menopause).
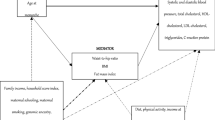
The mediation analysis was performed according to the principles of Baron and Kenny [22]. In the first instance, the mediator variable (WC and BMI) was regressed onto the independent variable (age at menarche). In the second equation, the dependent variable (systolic and diastolic BP) was regressed onto the independent variable. Finally, in the third equation, the dependent variable was regressed onto the independent variable, adjusted for the mediator. Mediating effects were identified if the following criteria were met: (a) in the first equation, the independent variable was a significant predictor of the mediator (if age at menarche was significantly associated with WC/BMI); (b) in the second equation, the independent variable was a significant predictor of the dependent variable (if age at menarche was significantly associated with systolic/diastolic BP); (c) in the third equation, the mediator was a significant predictor of the dependent variable and the association between the dependent and independent variables (from equation two) was either partially or fully removed (if WC/BMI was significantly associated with systolic/diastolic BP and if this association accounted for part or all of the significance of the association between age at menarche and systolic/diastolic BP). All covariates were adjusted for in the mediation models.
Analyses including the indirect and direct effects with 95% confidence intervals were calculated using structural equation modeling building in STATA 14 using the maximum likelihood method, with a 5% significance level. Analyses were conducted during 2017.
Results
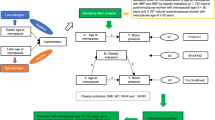
The final sample consisted of 33,715 women with an average age of 43.2 years (95% confidence interval from 42.9 to 43.6). The characteristics of the sample according to age at menarche are presented in Table 1. In general, chronological age was greater among the groups with a greater age at menarche than in those with a younger age at menarche. Moreover, the group of women who were ≤ 11 years old at menarche presented a greater average WC and BMI than their older counterparts. The group of women who were ≥ 15 y of age at menarche presented the greatest prevalence of women who had already entered menopause. Descriptive relationships between age at menarche and WC, BMI, and BP are presented in Fig. 1. Both adiposity indicators decreased with a higher age at menarche, whereas a weak association was observed between age at menarche and BP. Using categorical data, a greater prevalence of obesity was found among women with an earlier age at menarche, especially those who were ≤ 11 years of age at menarche (Fig. 2).
Correlations between independent, mediators, and dependent variables, according to menopause status, are presented in Table 2. All correlations were small but significant, with the exception of the correlation between age at menarche and diastolic BP after menopause. Moreover, the correlations between age at menarche with mediators and outcomes were weaker after menopause. In this sense, age at menarche was inversely associated with obesity indicators as well as BP (p < 0.001), whereas obesity indicators were positively associated with BP. After this analysis, the mediating effect of obesity indicators on the relationship between age at menarche and BP was tested (Fig. 3). In general, WC and BMI partly mediated the relationship between age at menarche and systolic BP (accounting for 41% and 44% of the effects, respectively), given that the mediators (WC and BMI) accounted for part of the effects and that age at menarche remained significant in equation 3. On the other hand, the relationship between age at menarche and diastolic BP was fully mediated by WC and BMI, given that age at menarche was no longer significant in equation 3 (with the addition of the obesity indicator).
Mediation models of waist circumference and body mass index on the relationship between age at menarche and blood pressure. Note Adjusted by chronological age, race, menopause status, hypertension drug use, educational status, tobacco smoking, leisure physical activity, TV viewing, sodium consumption and alcohol consumption
The results of the linear regression analyses stratified by menopause status using collapsed age at menarche ( ≤ 11, 12, 13, 14, ≥ 15 years) are presented in Table 3. The negative associations between age at menarche and systolic (β = − 0.620, P = 0.008) and diastolic (β = − 0.477, P = 0.005) BP were significant only among pre-menopausal women. In addition, WC significantly contributed to predicting systolic (R2 = 0.170, P = 0.046) and diastolic (R2 = 0.160, P = 0.025) BP only among pre-menopausal women.
Moreover, we tested the interaction between race and age at menarche, as well as the interaction between antihypertensive medication use and age at menarche in predicting systolic and diastolic BP. The interaction term of race*age at menarche was not significant for predicting either systolic BP (WC adjusted p = 0.951, BMI adjusted p = 0.903) or diastolic BP (WC adjusted p = 0.879, BMI adjusted p = 0.749). In addition, the interaction term of antihypertensive medication*age at menarche was not significant for predicting either systolic BP (WC adjusted p = 0.773, BMI adjusted p = 0.823) or diastolic BP (WC adjusted p = 0.516, BMI adjusted p = 0.484).
Discussion
This study investigated the association between age at menarche and body adiposity and BP in adulthood and examined the mediating effects of body adiposity on the relationship between age at menarche and BP. The main findings were that early maturation was related to obesity and higher BP in adulthood and that greater WC and BMI partly or fully mediated the relationship between early maturation and BP in adulthood. In general, this finding is consistent with the results of previous studies that suggest that earlier biological maturation is a risk factor for several negative health outcomes in late adolescence and adulthood [8, 23, 24]. Many previous studies support the conclusion that early biological maturation or puberty is related to obesity [8,9,10] and other metabolic risk factors [8, 11, 13]. On a somewhat similar note, previous studies have also suggested that the relationship between maturation and cardiovascular risk factors seems to be mediated by obesity-related variables, especially lipid variables [9, 13, 24, 25].
Our findings suggest the likelihood of a dose–response relationship between age at menarche and obesity indicators (WC and BMI). The age of 12 years appears to be the point at which there is a decline in obesity risk, as obesity indicators among the participants dropped from a high point at 8 years at menarche to a lower point at approximately 12 years of menarche, where they remained stable. This finding seems to be consistent with the literature that classifies menarche at less than 12 years old as early menarche and that found early menarche to be associated with obesity in adulthood [9, 26, 27]. This association could be explained by the fact that early maturing girls already have greater adiposity before puberty, given that adipose tissue, through leptin release, has a role in the initiation of the biological maturation process [28, 29]. Considering the potential tracking of adiposity from adolescence to adulthood [30], early maturing girls could maintain their higher adiposity in adulthood [31]. Another mechanism could be related to the time of the maturation process. There is evidence that early maturing adolescents experience a more accelerated maturation process than late maturing adolescents [32]. Thus, greater interference with biological and behavioral pathways in adolescents with faster maturation can lead to increased adiposity [32, 33].
We also found a significant inverse relationship between age at menarche and BP in adulthood, although this association was weak. This finding corroborates previous findings regarding other cardiovascular risk factors [8, 11] and is especially relevant given that the measure of BP was, on average, 30 years after the age at menarche. The weaker linear association between age at menarche and BP in adulthood could, in part, be explained by the fact that early maturers were younger than women with on time/late menarche. This finding may be a reflection of the fact that BP is an outcome with a higher latency period than adiposity.
The main novelty of our study is the finding that obesity indicators partly mediated the relationship between age at menarche and systolic BP, and fully mediated the relationship between age at menarche and diastolic BP in adulthood. Obesity could more strongly mediate the association between age at menarche and diastolic BP than systolic BP, because BMI is strongly associated with diastolic function [34, 35]. Nevertheless, our findings confirm the assumption and previous speculation that obesity could be a mediator of the association between age at menarche and onset of chronic diseases [9, 12, 13]. Considering that it is already clear that subjects with higher levels of adiposity present with greater BP [4], and that age at menarche is a risk factor for high adiposity in adulthood, it is biologically plausible that early maturation could influence BP in adulthood through greater body adiposity. However, greater adiposity can be associated with higher BP through different paths, such as increased peripheral resistance, increased sympathetic nervous system activity, renin–angiotensin–aldosterone system, and stimulation of negative hormones for BP [36].
Interestingly, previous studies that adjusted for important covariates and confounders of high BP, including lifestyle, socio-demographic factors and chronological age, suggest that, at least for systolic BP, there are other potential unobserved/unmeasured variables beyond adiposity that can mediate the effect of age at menarche[5, 6]. However, our findings suggest that obesity, due to its inverse relationship with age at menarche, is an important mediator of both systolic and diastolic BP in adulthood. In the present study, the association between age at menarche and diastolic BP was weaker; thus, obesity indicators could exert a greater influence. Moreover, among women who already entered menopause, the effects of menarche can be even weaker, as our results suggest, and obesity can present a greater effect on BP. Therefore, obesity in childhood/adolescence [31] or even behavioral factors through life (the relationship between physical activity and biological maturation) [37] are potential mediators to target when designing future and current interventions for BP control and management.
Our results have clear practical implications and applications. Given the adverse relationship between early age at menarche and body adiposity and BP, early interventions that aim to reduce body adiposity, even in childhood, should be promoted [38]. Moreover, keeping in mind that obesity indicators during childhood mediate the relationship between age at menarche and BP, interventions focusing on obese girls and women should be implemented [39, 40]. Furthermore, given that greater BMI maintenance and weight gain are associated with a greater likelihood of high BP [41], interventions aiming to avoid obesity throughout the life span should also be incorporated as part of public health strategies to control hypertension in women.
The main limitations of the current study are related to the cross-sectional nature of the study and the recall method used for the assessment of age at menarche. However, the results provided baseline data that could be explored further in future prospective studies. Moreover, the recall assessment method has good validity with a low error and bias of less than 1 year [42, 43]. A major strength of the study is the analysis of objectively measured obesity indicators and BP in a large sample of more than 34,000 Brazilian women. Another strength is the analytical method that controlled for potential covariates and confounders, such as chronological age, race, educational status, physical activity level, TV viewing, tobacco smoking, alcohol consumption, sodium ingestion and antihypertension medication use [5, 6].
In conclusion, age at menarche was inversely related with adiposity and BP in adulthood. Early maturation could predict obesity in adulthood, and adiposity indicators mediate the relationship between age at menarche and BP in adulthood. Obesity is an important risk factor to target to reduce the negative influence of early maturation on high BP in women.
Informed consent
Informed consent was obtained from all participants included in the study.
Data sharing statement
All data are open published as microdata at http://www.ibge.gov.br/home/.
References
Global burden of disease - GBD. Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980–2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet. 2015;388:117–71.
WHO. The top 10 causes of death. 2012. http://www.who.int/mediacentre/factsheets/fs310/en/. Accessed 1 October 2016
Forouzanfar MH, Liu P, Roth GA, Ng M, Biryukov S, Marczak L et al. Global burden of hypertension and systolic blood pressure of at least 110 to 115 mm Hg, 1990-2015. JAMA. 2017;317:827–38.
Gnatiuc L, Alegre-Díaz J, Halsey J, Herrington WG, López-Cervantes M, Lewington S et al. Adiposity and blood pressure in 110 000 Mexican adults. Hypertens (Dallas, Tex 1979). 2017;69:608–14.
Shrivastava SR, Shrivastava PS, Ramasamy J. The determinants and scope of public health interventions to tackle the global problem of hypertension. Int J Prev Med. 2014;5:807–12.
Masana L, Ros E, Sudano I, Angoulvant D, Ibarretxe Gerediaga D, Murga Eizagaechevarria N et al. Is there a role for lifestyle changes in cardiovascular prevention? What, when and how? Atheroscler Suppl. 2017;26:2–15.
Werneck AO, Silva DRP, Souza MF, Christofaro DGD, Tomeleri CM, Fernandes RA et al. Correlates of blood pressure according to early, on time, and late maturation in adolescents. J Clin Hypertens. 2016;18:424–30.
Widén E, Silventoinen K, Sovio U, Ripatti S, Cousminer DL, Hartikainen AL et al. Pubertal timing and growth influences cardiometabolic risk factors in adult males and females. Diabetes Care. 2012;35:850–6.
Dreyfus J, Jacobs DR Jr. Mueller N, Schreiner PJ, Moran A, Carnethon MR et al. Age at menarche and cardiometabolic risk in adulthood: the Coronary Artery Risk Development in Young Adults Study. J Pediatr. 2015;167:344–52.e1.
Gomes MB, Negrato CA, Calliari LEP. Early age at menarche: a risk factor for overweight or obesity in patients with type 1 diabetes living in urban areas? Diabetes Res Clin Pract. 2015;107:23–30.
Boyne MS, Thame M, Osmond C, Fraser RA, Gabay L, Taylor-Bryan C et al. The effect of earlier puberty on cardiometabolic risk factors in Afro-Caribbean children. J Pediatr Endocrinol Metab. 2014;27:453–60.
Kivimäki M, Lawlor DA, Smith GD, Elovainio M, Jokela M, Keltikangas-Järvinen L et al. Association of age at menarche with cardiovascular risk factors, vascular structure, and function in adulthood: the Cardiovascular Risk in Young Finns study. Am J Clin Nutr. 2008;87:1876–82. http://www.scopus.com/inward/record.url?eid=2-s2.0-45749120662&partnerID=40&md5=3cac7cb0dde232f4aa628f8b1e59e666
Lu J, Zhang J, Du R, Wang T, Xu M, Xu Y et al. Age at menarche is associated with the prevalence of NAFLD later in life. J Diabetes. 2016;9:53–60.
Krieger N, Kiang MV, Kosheleva A, Waterman PD, Chen JT, Beckfield J. Age at menarche: 50-year socioeconomic trends among US-born black and white women. Am J Public Health. 2015;105:388–97.
IBGE. Pesquisa Nacional de Saúde. 2013. https://doi.org/10.1249/MSS.0b013e3181949333.
Malina RM, Bouchard C, Bar-Or O. Growth, maturation, and physical activity. 2nd ed. Champaign: Human Kinetics. 2004.
World Health Organization. Obesity: preventing and managing the global epidemic. Report of a WHO Consultation. WHO Technical Report Series 894. Geneva.2000.
Ministério da Saúde, Fiocruz. Pesquisa Nacional de Saúde - Manual de Antropometria. http://www.pns.icict.fiocruz.br/arquivos/Portaria.pdf. 2003.
World Health Organization. Global recommendations on physical activity for health. Geneva. 2010. http://medcontent.metapress.com/index/A65RM03P4874243N.pdf%5Cn http://scholar.google.com/scholar?hl=en&btnG=Search&q=intitle:Global+Recomendations+on+physical+activity+for+health#0
Gardner MJ, Altman DG. Statistics in medicine confidence intervals rather than P values: estimation rather than hypothesis testing. Br Med J (Clin ResEd). 1986;292:746–50.
du Prel J-B, Hommel G, Röhrig B, Blettner M. Confidence interval or p-value?: part 4 of a series on evaluation of scientific publications. Dtsch Arztebl Int. 2009;106:335–9.
Baron RM, Kenny DA. The moderator-mediator variable distinction in social psychological research: conceptual, strategic, and statistical considerations. J Pers Soc Psychol. 1986;51:1173–82. http://www.ncbi.nlm.nih.gov/pubmed/3806354
Werneck AO, Silva DRP, Collings PJ, Fernandes RA, Ronque ERV, Coelho-e-Silva MJ et al. Birth weight, biological maturation and obesity in adolescents: a mediation analysis. J Dev Orig Health Dis. 2017;8:502–7.
Werneck AO, Silva DR, Collings PJ, Fernandes RA, Ronque ERV, Barbosa DS et al. Biological maturation, central adiposity, and metabolic risk in adolescents: a mediation analysis. Child Obes. 2016;12:377–83.
Hillman JB, Huang B, Pinney SM, Biro FM. Early pubertal development and insulin sensitivity among school-aged girls: mediation via adiposity. J Pediatr Adolesc Gynecol. 2013;26:47–50.
Leitão RB, Rodrigues LP, Neves L, Carvalho GS. Development of adiposity, obesity and age at menarche: an 8-year follow-up study in Portuguese schoolgirls. Int J Adolesc Med Health. 2013;25:55–63.
Kvalheim S, Sandvik L, Winsvold B, Hagen K, Zwart J-A. Early menarche and chronic widespread musculoskeletal complaints-Results from the HUNT study. Eur J Pain. 2016;20:458–64.
Ahmed ML, Ong KK, Dunger DB. Childhood obesity and the timing of puberty. Trends Endocrinol Metab. 2009;20:237–42.
Jasik CB, Lustig RH. Adolescent obesity and puberty: the ‘Perfect Storm’. Ann N Y Acad Sci. 2008;1135:265–79.
Garn SM. Continuities and changes in fatness from infancy through adulthood. Curr Probl Pediatr. 1985;15:5–46.
Must A. Childhood overweight and maturational timing in the development of adult overweight and fatness: The Newton Girls Study and Its Follow-up. Pediatrics. 2005;116:620–7.
Van Lenthe FJ, Kemper HCG, Van Mechelen W. Rapid maturation in adolescence results in greater obesity in adulthood: the Amsterdam Growth and Health Study. Am J Clin Nutr. 1996;64:18–24.
Reynolds EL, Sontag LW. The distribution of subcutaneous fat in childhood and adolescence. Monogr Soc Res Child Dev. 1950;15:iii.
Russo C, Jin Z, Homma S, Rundek T, Elkind MSV, Sacco RL et al. Effect of obesity and overweight on left ventricular diastolic function. J Am Coll Cardiol. 2011;57:1368–74.
Kossaify A, Nicolas N. Impact of overweight and obesity on left ventricular diastolic function and value of tissue Doppler echocardiography. Clin Med Insights Cardiol. 2013;7:CMC.S11156.
Leggio M, Lombardi M, Caldarone E, Severi P, D’Emidio S, Armeni M et al. The relationship between obesity and hypertension: an updated comprehensive overview on vicious twins. Hypertens Res. 2017;40:947–63.
Sherar LB, Cumming SP, Eisenmann JC, Baxter-Jones ADG, Malina RM. Adolescent biological maturity and physical activity: biology meets behavior. Pediatr Exerc Sci. 2010;22:332–49.
Carrel AL, McVean JJ, Clark RR, Peterson SE, Eickhoff JC, Allen DB. School-based exercise improves fitness, body composition, insulin sensitivity, and markers of inflammation in non-obese children. J Pediatr Endocrinol Metab. 2009;22:409–15.
Deusinger SS. Exercise intervention for management of obesity. Pediatr Blood Cancer. 2012;58:135–9.
Holliday KM, Lin DY, Chakladar S, Castañeda SF, Daviglus ML, Evenson KR et al. Targeting physical activity interventions for adults: when should intervention occur? Prev Med (Baltim). 2016;97:13–18.
Zhou L, Li Y, Guo M, Wu Y, Zhao L. Relations of body weight status in early adulthood and weight changes until middle age with hypertension in the Chinese population. Hypertens Res. 2016;39:913–8.
Must A, Phillips SM, Naumova EN, Blum M, Harris S, Dawson-Hughes BRW. Recall of early menstrual history and menarcheal body size: after 30 years, how well do women remember? Am J Epidemiol. 2002;155:672–679.
Koprowski C, Coates RJ, Bernstein L. Ability of young women to recall past body size and age at menarche. Obes Res. 2001;9:478–85.
Acknowledgements
We gratefully acknowledge the contributions of all participants of the present research as well as the IBGE staff for the data collection. Moreover, we acknowledge São Paulo Research Foundation (FAPESP) for the master’s degree scholarship of AOW (FAPESP process: 2017/27234-2).
Funding:
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethics approval:
All procedures performed in the original studies involving human participants were approved by the national council of ethics in research (CONEP: 10853812.7.0000.0008) in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Werneck, A.O., Oyeyemi, A.L., Cyrino, E.S. et al. Association between age at menarche and blood pressure in adulthood: is obesity an important mediator?. Hypertens Res 41, 856–864 (2018). https://doi.org/10.1038/s41440-018-0079-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41440-018-0079-4
This article is cited by
-
Adiposity indices and their higher predictive value for new-onset hypertension in metabolically healthy young women: findings from a population-based prospective cohort study
BMC Cardiovascular Disorders (2024)
-
Effect of air pollution on age at menarche in polish females, born 1993–1998
Scientific Reports (2022)
-
Structural equation model of the effect of biological maturation on metabolic syndrome risk and C-reactive protein: effect of trunk fat and sports participation
Scientific Reports (2021)
-
Early age at menarche and metabolic cardiovascular risk factors: mediation by body composition in adulthood
Scientific Reports (2021)