Abstract
Background
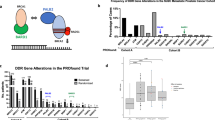
The loss of PTEN function presents in up to 50% of late-stage prostate cancers, and is therefore a potential target for therapeutics. PTEN-deficient cells depend on de novo pyrimidine synthesis, a feature that can present a vulnerability.
Methods
We utilized in vitro growth assays and in vivo xenograft models to test the effect of de novo pyrimidine synthesis inhibition on prostate cell lines.
Results
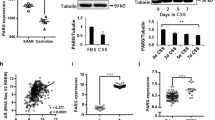
Here, we demonstrate that PTEN-deficient prostate cancer cell lines are susceptible to inhibition of de novo pyrimidine synthesis by leflunomide. Tumor growth inhibition was observed in vitro and in vivo following leflunomide treatment, and is likely due to an overwhelming accumulation of DNA damage.
Conclusions
Our work highlights that synthetic lethality arises upon the combination of PTEN loss and leflunomide treatment in prostate cancer, and may present a therapeutic opportunity for this patient population.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Li J, Yen C, Liaw D, Podsypanina K, Bose S, Wang SI, et al. PTEN, a putative protein tyrosine phosphatase gene mutated in human brain, breast, and prostate cancer. Science. 1997;275:1943–7.
Keniry M, Parsons R. The role of PTEN signaling perturbations in cancer and in targeted therapy. Oncogene. 2008;27:5477–85.
Tian XX, Pang JC, To SS, Ng HK. Restoration of wild-type PTEN expression leads to apoptosis, induces differentiation, and reduces telomerase activity in human glioma cells. J Neuropathol Exp Neurol. 1999;58:472–9.
Stambolic V, Suzuki A, de la Pompa JL, Brothers GM, Mirtsos C, Sasaki T, et al. Negative regulation of PKB/Akt-dependent cell survival by the tumor suppressor PTEN. Cell. 1998;95:29–39.
Cantley LC. The phosphoinositide 3-kinase pathway. Science 2002;296:1655–7.
Jamaspishvili T, Berman DM, Ross AE, Scher HI, De Marzo AM, Squire JA, et al. Clinical implications of PTEN loss in prostate cancer. Nat Rev Urol. 2018;15:222–34.
Krohn A, Diedler T, Burkhardt L, Mayer PS, De Silva C, Meyer-Kornblum M, et al. Genomic deletion of PTEN is associated with tumor progression and early PSA recurrence in ERG fusion-positive and fusion-negative prostate cancer. Am J Pathol. 2012;181:401–12.
Leinonen KA, Saramaki OR, Furusato B, Kimura T, Takahashi H, Egawa S, et al. Loss of PTEN is associated with aggressive behavior in ERG-positive prostate cancer. Cancer Epidemiol, Biomark Prev. 2013;22:2333–44.
Lotan TL, Heumann A, Rico SD, Hicks J, Lecksell K, Koop C, et al. PTEN loss detection in prostate cancer: comparison of PTEN immunohistochemistry and PTEN FISH in a large retrospective prostatectomy cohort. Oncotarget. 2017;8:65566–76.
Mense SM, Barrows D, Hodakoski C, Steinbach N, Schoenfeld D, Su W, et al. PTEN inhibits PREX2-catalyzed activation of RAC1 to restrain tumor cell invasion. Sci Signal. 2015;8:ra32.
Wong JT, Kim PT, Peacock JW, Yau TY, Mui AL, Chung SW, et al. Pten (phosphatase and tensin homologue gene) haploinsufficiency promotes insulin hypersensitivity. Diabetologia. 2007;50:395–403.
Hu H, Juvekar A, Lyssiotis CA, Lien EC, Albeck JG, Oh D, et al. Phosphoinositide 3-kinase regulates glycolysis through mobilization of aldolase from the actin cytoskeleton. Cell. 2016;164:433–46.
Mathur D, Stratikopoulos E, Ozturk S, Steinbach N, Pegno S, Schoenfeld S, et al. PTEN regulates glutamine flux to pyrimidine synthesis and sensitivity to dihydroorotate dehydrogenase inhibition. Cancer Discov. 2017;7:380–90.
Ben-Sahra I, Howell JJ, Asara JM, Manning BD. Stimulation of de novo pyrimidine synthesis by growth signaling through mTOR and S6K1. Science. 2013;339:1323–8.
Greene S, Watanabe K, Braatz-Trulson J, Lou L. Inhibition of dihydroorotate dehydrogenase by the immunosuppressive agent leflunomide. Biochemical Pharmacol. 1995;50:861–7.
Munier-Lehmann H, Vidalain PO, Tangy F, Janin YL. On dihydroorotate dehydrogenases and their inhibitors and uses. J Medicinal Chem. 2013;56:3148–67.
Shawver LK, Schwartz DP, Mann E, Chen H, Tsai J, Chu L. et al.Inhibition of platelet-derived growth factor-mediated signal transduction and tumor growth by N-[4-(trifluoromethyl)-phenyl]5-methylisoxazole-4-carboxamide. Clin Cancer Res.1997;3:1167–77.
Hail N Jr, Chen P, Bushman LR. Teriflunomide (leflunomide) promotes cytostatic, antioxidant, and apoptotic effects in transformed prostate epithelial cells: evidence supporting a role for teriflunomide in prostate cancer chemoprevention. Neoplasia. 2010;12:464–75.
Fragoso YD, Brooks JB. Leflunomide and teriflunomide: altering the metabolism of pyrimidines for the treatment of autoimmune diseases. Expert Rev Clin Pharmacol. 2015;8:315–20.
Ko Y-J, Small EJ, Kabbinavar F, Chachoua A, Taneja S, Reese D, et al. A multi-institutional phase II study of SU101, a platelet-derived growth factor receptor inhibitor, for patients with hormone-refractory prostate cancer. Clin Cancer Res. 2001;7:800–5.
Acknowledgements
We thank all the Parsons’s lab members for their critical input during the planning, execution and reporting phases of the study. Grant Support: Grant support for this work was provided by Prostate Cancer Foundation 2016 PCF Challenge Award 16CHAL14 and National Cancer Institute R35 CA220491.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
RP is a shareholder of Therapten, Inc., a company focused on using an isoform of PTEN protein as a treatment for disease. The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Ozturk, S., Mathur, D., Zhou, R.W. et al. Leflunomide triggers synthetic lethality in PTEN-deficient prostate cancer. Prostate Cancer Prostatic Dis 23, 718–723 (2020). https://doi.org/10.1038/s41391-020-0251-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41391-020-0251-1
This article is cited by
-
Pyrimidine nucleotide starvation induces a decrease in the number of effector T cells but not memory T cells
Cellular & Molecular Immunology (2023)
-
Oral Conventional Synthetic Disease-Modifying Antirheumatic Drugs with Antineoplastic Potential: a Review
Dermatology and Therapy (2022)