Abstract
Background
Inhaled nitric oxide (iNO) is widely used for the management of infants with congenital diaphragmatic hernia (CDH); however, evidence of benefit is limited.
Methods
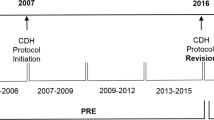
This is a multicenter cohort study using data from the Congenital Diaphragmatic Hernia Study Group between 2015 and 2020. The impact of early iNO use in the first 3 days of life prior to ECLS use on mortality or ECLS use was explored using multivariate logistic regression models and subgroup analyses.
Results
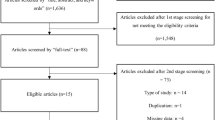
Of the 1777 infants, 863 (48.6%) infants received early iNO treatment. Infants receiving iNO had lower birth weight, larger defect size, more severe pulmonary hypertension, and abnormal ventricular size and function. After controlling for these factors, early iNO use was associated with increased mortality (aOR 2.06, 95% CI 1.05–4.03, P = 0.03) and increased ECLS use (aOR 3.44, 95% CI 2.11–5.60, P < 0.001). Subgroup analyses after stratification by echocardiographic characteristics and defect size revealed no subgroup with a reduction in mortality or ECLS use.
Conclusions
Use of iNO in the first 3 days of life prior to ECLS was not associated with a reduction in mortality or ECLS use in either the regression models or the subgroup analyses. The widespread use of iNO in this vulnerable population requires reconsideration.
Impact
-
Evidence to support widespread use of iNO for infants with congenital diaphragmatic hernia is limited.
-
The use of iNO in the first 3 days of life was associated with significantly increased mortality and ECLS use.
-
Stratification by echocardiographic characteristics and defect size did not reveal a subgroup that benefited from iNO. Even the subset of patients with R-to-L shunts at both ductal and atrial levels, a surrogate for elevated pulmonary arterial pressures in the absence of significantly decreased LV compliance, did not benefit from early iNO use.
-
Early iNO therapy was of no benefit in the management of acute pulmonary hypertension in infants with congenital diaphragmatic hernia, supporting reconsideration of its use in this population.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the CDHSG but membership restrictions apply, therefore the data are not publicly available. Under unique and special circumstances, data could be made available from the authors following reasonable request, for a specific rationale, and with permission of the CDHSG.
Change history
12 April 2023
A Correction to this paper has been published: https://doi.org/10.1038/s41390-023-02581-7
References
Gupta, V. S. & Harting, M. T. Congenital diaphragmatic hernia-associated pulmonary hypertension. Semin. Perinatol. 44, 151–167 (2020).
Levin, D. L. Morphologic analysis of the pulmonary vascular bed in congenital left-sided diaphragmatic hernia. J. Pediatr. 92, 805–809 (1978).
Lath, N. R. et al. Defective pulmonary innervation and autonomic imbalance in congenital diaphragmatic hernia. Am. J. Physiol. Lung Cell. Mol. Physiol. 302, L390–L398 (2012).
Irish, M. S. et al. Contractile properties of intralobar pulmonary arteries and veins in the fetal lamb model of congenital diaphragmatic hernia. J. Pediatr. Surg. 33, 921–928 (1998).
Patel, N. et al. Ventricular dysfunction is a critical determinant of mortality in congenital diaphragmatic hernia. Am. J. Respir. Crit. Care Med. 200, 1522–1530 (2019).
Heiss, K. et al. Reversal of mortality for congenital diaphragmatic hernia with ECMO. Ann. Surg. 209, 225–230 (1989).
Schumacher, R. E., Roloff, D. W., Chapman, R., Snedecor, S. & Bartlett, R. H. Extracorporeal membrane oxygenation in term newborns. ASAIO J. 39, 873–879 (1993).
UK Collaborative ECMO Trial Group. UK collaborative randomised trial of neonatal extracorporeal membrane oxygenation. Lancet 348, 75–82 (1996).
Congenital Diaphragmatic Hernia Study Group. Does extracorporeal membrane oxygenation improve survival in neonates with congenital diaphragmatic hernia? J. Pediatr. Surg. 34, 720–725 (1999).
Kays, D. W. ECMO in CDH: is there a role? Semin. Pediatr. Surg. 26, 166–170 (2017).
Langham, M. R. et al. Twenty years of progress in congenital diaphragmatic hernia at the University of Florida. Am. Surg. 69, 45–52 (2003).
Moyer, V. A. et al. Late versus early surgical correction for congenital diaphragmatic hernia in newborn infants. Cochrane Database Syst. Rev. 2000, CD001695 (2000).
Jancelewicz, T. et al. Survival benefit associated with the use of extracorporeal life support for neonates with congenital diaphragmatic hernia. Ann. Surg. 275, e256–e263 (2022).
Neonatal Inhaled Nitric Oxide Study Group. Inhaled nitric oxide in full-term and nearly full-term infants with hypoxic respiratory failure. N. Engl. J. Med. 336, 597–604 (1997).
Clark, R. H. et al. Low-dose nitric oxide therapy for persistent pulmonary hypertension of the newborn. N. Engl. J. Med. 342, 469–474 (2000).
Barrington, K. J., Finer, N., Pennaforte, T. & Altit, G. Nitric oxide for respiratory failure in infants born at or near term. Cochrane Database Syst. Rev. 2017, CD000399 (2017).
Campbell, B. T. et al. Inhaled nitric oxide use in neonates with congenital diaphragmatic hernia. Pediatrics 134, e420–e426 (2014).
Putnam, L. R. et al. Evaluation of variability in inhaled nitric oxide use and pulmonary hypertension in patients with congenital diaphragmatic hernia. JAMA Pediatr. 170, 1188 (2016).
Kinsella, J. P., Neish, S. R., Ivy, D. D., Shaffer, E. & Abman, S. H. Clinical responses to prolonged treatment of persistent pulmonary hypertension of the newborn with low doses of inhaled nitric oxide. J. Pediatr. 123, 103–108 (1993).
Neonatal Inhaled Nitric Oxide Study Group. Inhaled nitric oxide and hypoxic respiratory failure in infants with congenital diaphragmatic hernia. Pediatrics 99, 838–845 (1997).
Lawrence, K. M. et al. Inhaled nitric oxide is associated with improved oxygenation in a subpopulation of infants with congenital diaphragmatic hernia and pulmonary hypertension. J. Pediatr. 219, 167–172 (2019).
Ferguson, D. M. et al. Early, postnatal pulmonary hypertension severity predicts inpatient outcomes in congenital diaphragmatic hernia. Neonatology 118, 147–154 (2021).
Byrne, F. A. et al. Severe left diaphragmatic hernia limits size of fetal left heart more than does right diaphragmatic hernia. Ultrasound Obstet. Gynecol. 46, 688–694 (2015).
Kailin, J. A. et al. Fetal left‐sided cardiac structural dimensions in left‐sided congenital diaphragmatic hernia – association with severity and impact on postnatal outcomes. Prenat. Diagn. 37, 502–509 (2017).
Massolo, A. C. et al. Fetal cardiac dimensions in congenital diaphragmatic hernia: relationship with gestational age and postnatal outcomes. J. Perinatol. 41, 1651–1659 (2021).
Nair, J. & Lakshminrusimha, S. Update on PPHN: mechanisms and treatment. Semin. Perinatol. 38, 78–91 (2014).
Gien, J. & Kinsella, J. P. Management of pulmonary hypertension in infants with congenital diaphragmatic hernia. J. Perinatol. 36, S28–S31 (2016).
Wehrmann, M. et al. Implications of atrial-level shunting by echocardiography in newborns with congenital diaphragmatic hernia. J. Pediatr. 219, 43–47 (2020).
Tsao, K. & Lally, K. P. The Congenital Diaphragmatic Hernia Study Group: a voluntary international registry. Semin. Pediatr. Surg. 17, 90–97 (2008).
Harting, M. T. & Lally, K. P. The Congenital Diaphragmatic Hernia Study Group registry update. Semin. Fetal Neonatal Med. 19, 370–375 (2014).
CDHSG Data Collection Form v4. https://med.uth.edu/pediatricsurgery/wp-content/uploads/sites/3/2020/08/CDH_Data_Form_V4_2020.docx (2020).
Van Meurs, K. P. et al. Inhaled nitric oxide for premature infants with severe respiratory failure. N. Engl. J. Med. 353, 13–22 (2005).
Mercier, J.-C. et al. Inhaled nitric oxide for prevention of bronchopulmonary dysplasia in premature babies (EUNO): a randomised controlled trial. Lancet 376, 346–354 (2010).
Kinsella, J. P. et al. The left ventricle in congenital diaphragmatic hernia: implications for the management of pulmonary hypertension. J. Pediatr. 197, 17–22 (2018).
Lally, K. P. et al. Standardized reporting for congenital diaphragmatic hernia – an international consensus. J. Pediatr. Surg. 48, 2408–2415 (2013).
Altit, G., Bhombal, S., Van Meurs, K. P. & Tacy, T. A. Ventricular performance is associated with need for extracorporeal membrane oxygenation in newborns with congenital diaphragmatic hernia. J. Pediatr. 191, 28.e1–34.e1 (2017).
Patel, N. et al. Early postnatal ventricular dysfunction is associated with disease severity in patients with congenital diaphragmatic hernia. J. Pediatr. 203, 400.e1–407.e1 (2018).
Moenkemeyer, F. & Patel, N. Right ventricular diastolic function measured by tissue doppler imaging predicts early outcome in congenital diaphragmatic hernia. Pediatr. Crit. Care Med. 15, 49–55 (2014).
Herich, K. et al. iNO therapy in patients with congenital diaphragmatic hernia – discrepancy between widespread use and therapeutic effects. Klin. Pädiatr. 231, 320–325 (2019).
Kumar, V. H. S., Dadiz, R., Koumoundouros, J., Guilford, S. & Lakshminrusimha, S. Response to pulmonary vasodilators in infants with congenital diaphragmatic hernia. Pediatr. Surg. Int. 34, 735–742 (2018).
Mohseni-Bod, H. & Bohn, D. Pulmonary hypertension in congenital diaphragmatic hernia. Semin. Pediatr. Surg. 16, 126–133 (2007).
Shehata, S. M. K., Sharma, H. S., Mooi, W. J. & Tibboel, D. Pulmonary hypertension in human newborns with congenital diaphragmatic hernia is associated with decreased vascular expression of nitric-oxide synthase. Cell Biochem. Biophys. 44, 147–155 (2006).
Solari, V., Piotrowska, A. P. & Puri, P. Expression of heme oxygenase-1 and endothelial nitric oxide synthase in the lung of newborns with congenital diaphragmatic hernia and persistent pulmonary hypertension. J. Pediatr. Surg. 38, 808–813 (2003).
Mous, D. S., Kempen, M. J. B., Wijnen, R. M. H., Tibboel, D. & Rottier, R. J. Changes in vasoactive pathways in congenital diaphragmatic hernia associated pulmonary hypertension explain unresponsiveness to pharmacotherapy. Respir. Res. 18, 187 (2017).
Coppola, C. P. & Gosche, J. R. Oxygen-induced vasodilation is blunted in pulmonary arterioles from fetal rats with nitrofen-induced congenital diaphragmatic hernia. J. Pediatr. Surg. 36, 593–597 (2001).
Galié, N., Manes, A. & Branzi, A. The endothelin system in pulmonary arterial hypertension. Cardiovasc. Res. 61, 227–237 (2004).
Okazaki, T., Sharma, H. S., McCune, S. K. & Tibboel, D. Pulmonary vascular balance in congenital diaphragmatic hernia: enhanced endothelin-1 gene expression as a possible cause of pulmonary vasoconstriction. J. Pediatr. Surg. 33, 81–84 (1998).
Keller, R. L. et al. Congenital diaphragmatic hernia: endothelin-1, pulmonary hypertension, and disease severity. Am. J. Respir. Crit. Care Med. 182, 555–561 (2010).
Acknowledgements
We acknowledge the ongoing contributions of highly committed Congenital Diaphragmatic Hernia Study Group member centers that voluntarily participate in the study of congenital diaphragmatic hernia.
Funding
This study was supported by a grant from the Maternal & Child Health Research Institute at Stanford awarded to C.Y.N. The sponsor had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Author information
Authors and Affiliations
Consortia
Contributions
C.Y.N., V.Y.C., S.B., and K.P.V.M. made substantial contributions to conception and design of the study; acquisition, analysis, and interpretation of data; and drafting and critical revision of the manuscript. E.D. made substantial contributions to conception and design of the study; acquisition, analysis, and interpretation of data; and critical revision of the manuscript for important intellectual content. N.P., A.D., M.T.H., K.P.L., and A.H.E. made substantial contributions to acquisition, analysis, and interpretation of data and critical revision of the manuscript for important intellectual content. All the authors agreed and approved this version to be submitted for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The CDHSG registry is hosted by the University of Texas at Houston and approved by its Institutional Review Board (HSC-MS-03-223) with waiver of informed consent.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: due to a typesetting error, the headings of two separate columns in Table 3 were unfortunately merged into one.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Noh, C.Y., Chock, V.Y., Bhombal, S. et al. Early nitric oxide is not associated with improved outcomes in congenital diaphragmatic hernia. Pediatr Res 93, 1899–1906 (2023). https://doi.org/10.1038/s41390-023-02491-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-023-02491-8
This article is cited by
-
Multicentre, randomised controlled trial of physiological-based cord clamping versus immediate cord clamping in infants with a congenital diaphragmatic hernia (PinC): statistical analysis plan
Trials (2024)
-
Management of infants with congenital diaphragmatic hernia and pulmonary hypertension—one size does not fit all
Pediatric Research (2023)