Abstract
Background
Neurofilament light (NfL) has been identified as a biomarker for neuroaxonal damage in preterm infants, but its relation with bronchopulmonary dysplasia (BPD) has not been established. We hypothesized that BPD is associated with increased NfL levels at an early stage, indicative of early neuroaxonal damage.
Methods
We included preterm infants born <30 weeks of gestation for assessment of NfL levels from cord blood and blood obtained at postnatal days 3, 7, 14, and 28. We used linear regression analysis to compare NfL levels between infants with moderate/severe BPD and infants with no/mild BPD, and linear mixed model analysis to compare the effect of time on NfL levels between groups.
Results
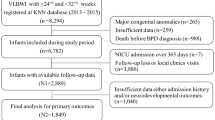
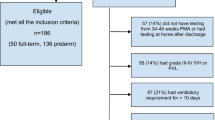
Sixty-seven infants with a gestational age (GA) of 27 ± 1.3 weeks were included for analysis, of whom 19 (28%) developed moderate/severe BPD. Although NfL levels were higher at every time point in infants with BPD, statistical significance was lost after adjustment for GA, small for gestational age (SGA) and intraventricular hemorrhage (IVH). Groups did not differ in NfL change over time.
Conclusions
The positive association between BPD and NfL in the first weeks of life could be explained by GA, SGA and IVH rather than by development of BPD.
Impact
-
Neurofilament light chain (NfL) is a known biomarker for neuroaxonal damage.
-
Biomarkers for brain damage during the first weeks of life in preterm infants developing BPD are lacking.
-
NfL levels obtained during the first weeks of life did not differ between infants with and without BPD in analyses adjusted for GA, SGA, and IVH.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Twilhaar, E. S. et al. Cognitive outcomes of children born extremely or very preterm since the 1990s and associated risk factors: a meta-analysis and meta-regression. JAMA Pediatr. 172, 361–367 (2018).
Natarajan, G. et al. Outcomes of extremely low birth weight infants with bronchopulmonary dysplasia: impact of the physiologic definition. Early Hum. Dev. 88, 509–515 (2012).
Anderson, P. J. & Doyle, L. W. Neurodevelopmental outcome of bronchopulmonary dysplasia. Semin. Perinatol. 30, 227–232 (2006).
DeMauro, S. B. Neurodevelopmental outcomes of infants with bronchopulmonary dysplasia. Pediatr. Pulmonol. 56, 3509–3517 (2021).
Bose, C. et al. Blood protein concentrations in the first two postnatal weeks that predict bronchopulmonary dysplasia among infants born before the 28th week of gestation. Pediatr. Res. 69, 347–353 (2011).
Leviton, A. et al. Two-hit model of brain damage in the very preterm newborn: small for gestational age and postnatal systemic inflammation. Pediatr. Res. 73, 362–370 (2013).
Leviton, A. et al. Early postnatal blood concentrations of inflammation-related proteins and microcephaly two years later in infants born before the 28th post-menstrual week. Early Hum. Dev. 87, 325–330 (2011).
Bose, C. L. et al. Systemic inflammation associated with mechanical ventilation among extremely preterm infants. Cytokine 61, 315–322 (2013).
Perrone, S., Tataranno, L. M., Stazzoni, G., Ramenghi, L. & Buonocore, G. Brain susceptibility to oxidative stress in the perinatal period. J. Matern. Fetal Neonatal Med. 28(Suppl. 1), 2291–2295 (2015).
Vliegenthart, R. J. S., van Kaam, A. H., Aarnoudse-Moens, C. S. H., van Wassenaer, A. G. & Onland, W. Duration of mechanical ventilation and neurodevelopment in preterm infants. Arch. Dis. Child Fetal Neonatal Ed. 104, F631–F635 (2019).
Doyle, L. W., Cheong, J. L., Ehrenkranz, R. A. & Halliday, H. L. Early (<8 days) systemic postnatal corticosteroids for prevention of bronchopulmonary dysplasia in preterm infants. Cochrane Database Syst. Rev. 10, CD001146 (2017).
Burnett, A. C., Cheong, J. L. Y. & Doyle, L. W. Biological and social influences on the neurodevelopmental outcomes of preterm infants. Clin. Perinatol. 45, 485–500 (2018).
Kennedy, E., Poppe, T., Tottman, A. & Harding, J. Neurodevelopmental impairment is associated with altered white matter development in a cohort of school-aged children born very preterm. Neuroimage Clin. 31, 102730 (2021).
Lee, M. K., Xu, Z., Wong, P. C. & Cleveland, D. W. Neurofilaments are obligate heteropolymers in vivo. J. Cell Biol. 122, 1337–1350 (1993).
Nitz, E. et al. Serum neurofilament light chain in pediatric spinal muscular atrophy patients and healthy children. Ann. Clin. Transl. Neurol. 8, 2013–2024 (2021).
Olsson, B. et al. Nfl is a marker of treatment response in children with SMA treated with nusinersen. J. Neurol. 266, 2129–2136 (2019).
Alirezaei, Z. et al. Neurofilament light chain as a biomarker, and correlation with magnetic resonance imaging in diagnosis of CNS-related disorders. Mol. Neurobiol. 57, 469–491 (2020).
Sjobom, U. et al. Analysis of brain injury biomarker neurofilament light and neurodevelopmental outcomes and retinopathy of prematurity among preterm infants. JAMA Netw. Open 4, e214138 (2021).
Depoorter, A. et al. Neurofilament light chain: blood biomarker of neonatal neuronal injury. Front. Neurol. 9, 984 (2018).
Goeral, K. et al. Early life serum neurofilament dynamics predict neurodevelopmental outcome of preterm infants. J. Neurol. 268, 2570–2577 (2021).
Katz, T. A. et al. Severity of bronchopulmonary dysplasia and neurodevelopmental outcome at 2 and 5 years corrected age. J. Pediatr. 243, 40.e2–46.e2 (2021).
Jobe, A. H. & Bancalari, E. Bronchopulmonary dysplasia. Am. J. Respir. Crit. Care Med. 163, 1723–1729 (2001).
Kuhle, J. et al. Comparison of three analytical platforms for quantification of the neurofilament light chain in blood samples: elisa, electrochemiluminescence immunoassay and Simoa. Clin. Chem. Lab. Med. 54, 1655–1661 (2016).
Beerepoot, S. et al. Neurofilament light chain and glial fibrillary acidic protein levels in metachromatic leukodystrophy. Brain 145, 105–118 (2021).
Andreasson, U. et al. A practical guide to immunoassay method validation. Front. Neurol. 6, 179 (2015).
Stoll, B. J. et al. Neonatal outcomes of extremely preterm infants from the NICHD Neonatal Research Network. Pediatrics 126, 443–456 (2010).
Hoftiezer, L. et al. From population reference to national standard: new and improved birthweight charts. Am. J. Obstet. Gynecol. 220, 383 e381–383.e317 (2019).
Pruessner, J. C., Kirschbaum, C., Meinlschmid, G. & Hellhammer, D. H. Two formulas for computation of the area under the curve represent measures of total hormone concentration versus time-dependent change. Psychoneuroendocrinology 28, 916–931 (2003).
Disanto, G. et al. Serum neurofilament light: a biomarker of neuronal damage in multiple sclerosis. Ann. Neurol. 81, 857–870 (2017).
Rohrer, J. D. et al. Serum neurofilament light chain protein is a measure of disease intensity in frontotemporal dementia. Neurology 87, 1329–1336 (2016).
Leigh, S. R. Brain growth, life history, and cognition in primate and human evolution. Am. J. Primatol. 62, 139–164 (2004).
Bouyssi-Kobar, M. et al. Third trimester brain growth in preterm infants compared with in utero healthy fetuses. Pediatrics 138, e20161640 (2016).
Julious, S. A. Sample Sizes for Clinical Trials 317 (Chapman and Hall/CRC, 2010).
Shein-Chung Chow, H. W. & Shao J. Sample Size Calculations in Clinical Research 480 (Chapman and Hall/CRC, 2007).
David Machin, M. J. C., Tan, S. B. & Tan, S. H. Sample Size Tables for Clinical Studies 2nd edn (Wiley-Blackwell Science, 1997).
Zar, J. H. Biostatistical Analysis 2nd edn (Practice-Hall, 1984).
Funding
This research was supported by the Amsterdam Reproduction & Development Research Institute.
Author information
Authors and Affiliations
Contributions
M.R. made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data, drafted and revised the article, and approved the final version to be published. E.M.B., B.I.L.-W., W.O., H.H., and M.J.J.F. made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data, revised it critically for important intellectual content, and approved the final version to be published. M.K., J.O., J.R., A.H.v.K., and C.E.T. made substantial contributions to interpretation of the data, revised the manuscript critically for important intellectual content, and approved the final version to be published.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
Written informed consent for participant and publication of the study results was obtained from all parents. The study was approved by the Medical Ethics Committee of the VU University Medical Center (protocol number 2019.371).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Romijn, M., Baas, E.M., Lissenberg-Witte, B.I. et al. Bronchopulmonary dysplasia is not related to neurofilament light for neuroaxonal damage in preterm infants. Pediatr Res 93, 2014–2018 (2023). https://doi.org/10.1038/s41390-022-02365-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-022-02365-5