Abstract
Objectives
To examine whether areal bone mineral density (aBMD) differs between metabolically healthy (MHO) and unhealthy (MUO) overweight/obese children and to examine the role of moderate-to-vigorous physical activity (MVPA) and cardiorespiratory fitness (CRF) in this association.
Methods
A cross-sectional study was developed in 188 overweight/obese children (10.4 ± 1.2 years) from the ActiveBrains and EFIGRO studies. Participants were classified as MHO or MUO based on Jolliffe and Janssen’s metabolic syndrome cut-off points for triglycerides, glucose, high-density cholesterol and blood pressure. MVPA and CRF were assessed by accelerometry and the 20-m shuttle run test, respectively. Body composition was measured by dual-energy X-ray absorptiometry.
Results
In model 1 (adjusted for sex, years from peak high velocity, stature and lean mass), MHO children had significantly higher aBMD in total body less head (Cohen’s d effect size, ES = 0.34), trunk (ES = 0.43) and pelvis (ES = 0.33) than MUO children. These differences were attenuated once MVPA was added to model 1 (model 2), and most of them disappeared once CRF was added to the model 1 (model 3).
Conclusions
This novel research shows that MHO children have greater aBMD than their MUO peers. Furthermore, both MVPA and more importantly CRF seem to partially explain these findings.
Similar content being viewed by others
Introduction
About 200 million people suffer osteoporosis worldwide1 and approximately 3.4 million people died because of overweight and obesity,2 and its prevalence is expected to increase. As in osteoporosis,3 obesity in childhood tracks into adulthood,4 and therefore it is of utmost importance to focus on this population. Obesity and osteoporosis are different disorders seldom found in the same individual, but scientific evidence supports a controversial link between fat and bone. It has traditionally been considered that excess weight due to obesity has a positive effect on bone mineral density (BMD), which may decrease osteoporotic fracture risk;5 nevertheless, more recent findings suggest that BMD is compromised.6 From a mechanistic point of view, obese youngsters may have greater BMD than their normoweight peers due to their greater lean mass and the higher impact associated with it, consequence of the extra body weight they have to carry on their everyday life activities.7 However, evidence also suggests a negative association between abdominal adiposity and BMD in obese youngsters.8 This may be explained by the role of the abdominal adipose tissue in the release of adipokines into the bloodstream, which negatively affects bone remodelling.8
Also, childhood obesity is commonly associated with increased risk of cardiovascular and metabolic factors.9 Nevertheless, some obese individuals do not show any of these metabolic disorders and they are named as metabolically healthy but obese (MHO).10 MHO individuals despite having a body mass index (BMI) ≥ 30 kg/m2 present a normal/healthy metabolic and inflammation profile11 and may represent for 30% of the obese population.10 Previous researches have shown that MHO adolescents and adults have better cardiorespiratory fitness (CRF)12,13 and they are more active14 than their metabolically unhealthy obese (MUO) peers, which was confirmed in a recent meta-analysis.15 Moreover, adolescents with good CRF have more bone mass than those with poor CRF16 and those more active also present greater levels of bone mass.17 However, whether MHO children and adolescents have greater or lower bone mass than their MUO peers and whether moderate-to-vigorous physical activity (MVPA) or CRF contribute to these differences has yet to be elucidated. In this regard, MVPA refers to a lifestyle behaviour while CRF refers to a physical state derived from a genetic component. Therefore, testing their independent contribution is necessary.
So far, the only study published examining the association between MHO and MUO with BMD (in adult population) showed that the MUO phenotype was associated with higher total BMD than the MHO, but no association was found with lumbar spine BMD.11 Thereby, there could be a novel link between BMD and MHO/MUO phenotype. To the best of our knowledge, there is no scientific evidence about this possible link in young population. Thus the aim of the present study was twofold: (1) to examine whether areal BMD (aBMD) differs between MHO and MUO children and (2) to examine the role of MVPA and CRF in this association.
Methods
The present cross-sectional study was developed in a subsample of 188 overweight and obese children (age 10.4 ± 1.2 years) with valid data in the variables of interest. Both the ActiveBrains and EFIGRO studies were developed from 2014 to 2017, under the same protocols and with data collection obtained in parallel. Details of design and methods have been published elsewhere.18,19
Briefly, ActiveBrains (ClinicalTrials.gov ID: NCT02295072) is a randomised controlled trial designed to examine the effects of an exercise programme on brain, cognitive and academic performance, as well as on selected physical and mental health outcomes in overweight/obese children.18 A total of 110 overweight/obese children aged 8–11 years were recruited from Granada (south of Spain) according to the following inclusion criteria:18 (1) to be overweight or obese based on World Obesity Federation (formerly named International Obesity Task Force) cut-off points, (2) to be aged 8–11 years, (3) not to have any physical disabilities or neurological disorder that affects their physical performance, and (4) in the case of girls, not to have started the menstruation at the time of the assessments. EFIGRO (ClinicalTrials.gov ID: NCT02258126) is a randomised controlled trial designed to evaluate the effect of a multidisciplinary intervention programme on hepatic fat fraction, cardiometabolic risk factors, self-esteem and well-being in overweight or obese children.19 A total of 116 overweight/obese children aged 9–11 years were recruited from Vitoria-Gasteiz (north of Spain) according to the following inclusion criteria:19 (1) primary overweight or obesity status defined according to the International Obesity Task Force, (2) aged between 9 and 11 years, (3) to have at least one parent or caregiver willing to participate in the programme sessions, (4) not to have medical conditions or medications that would affect study results or limit physical activity, and (5) in the case of girls, not to have started the menstruation at the time of the assessments.
The research was conducted according to the Declaration of Helsinki and the protocols were approved by the Review Committee for Research Involving Human Subjects at the University of Granada and the Ethic Committee of Clinical Investigation of Euskadi. Informed consent was obtained from all participants.
Outcome measures
Weight (kg) and stature (cm) were measured with an electronic scale (SECA 861 and 760, Hamburg, Germany) and a stadiometer (SECA 225 and 220, Hamburg, Germany), respectively. BMI was calculated as weight (kg)/stature (m2) and classified into BMI categories (overweight and obesity), according to Cole and Lobstein20 cut-offs. Years from peak height velocity (PHV) was used as a maturational landmark and was predicted through age and stature using validated algorithms in boys and girls.21
Physical activity was objectively measured by tri-axial accelerometers (GT3X+, ActiGraph, Pensacola, FL, USA) for 7 consecutive days. The participants wore the accelerometers on the non-dominant wrist during 24 h a day and removed it only while bathing or swimming. Accelerometer raw data were processed using the GGIR package for R (GGIR Package, v.1.5.−24, https://cran.r-project.org/web/packages/GGIR/). In brief, raw data were aggregated as the Euclidean Norm Minus One g (ENMO) over 5 s epochs with all negative values rounded to 0. The ENMO metric was used to determine the time spent in MVPA by applying previously proposed cut-points validated against metabolic equivalents (measured with indirect calorimetry) for MVPA.22
CRF was assessed by the 20-m shuttle run test from the ALPHA (Assessing Levels of Physical fitness and Health in Adolescents) test battery, which has been shown to be valid, reliable, feasible, and safe for the assessment of health-related physical fitness in children.23 The last completed lap was recorded and used as an indicator of CRF levels.
Fat mass (%), lean mass (kg) and aBMD (g/cm2) were measured by dual-energy X-ray absorptiometry (DXA) using the Hologic QDR 4500W (EFIGRO study) and the Discovery Wi (ActiveBrains study) (both from the manufacturer Hologic Series Discovery QDR, Bedford, MA, USA). The total body scan was used to obtain aBMD of the total body less head (TBLH), trunk, lumbar spine, arms, legs, and pelvis. All DXA scans and analyses were performed using the GE encore software (version 4.0.2) and were completed following the same protocol by the same researcher within each study, following recommendations from the International Society of Clinical Densitometry.24 Data from DXA scans were normalised separately within each study and then data were combined.
Briefly, serum triglycerides (mmol/L), fasting glucose (GL, mmol/L) and high-density lipoprotein (HDL) (mmol/L) cholesterol were measured from fasting blood samples using standard protocols. Venepuncture was performed to obtain blood samples and those were immediately centrifuged, aliquoted within 1 h after collection, and stored at −70 °C or below. Systolic and diastolic blood pressure was obtained with an automatic oscillometric device (Omron M6, The Netherlands).
We followed the metabolic syndrome definition proposed by Ortega et al.9 and the cut-off points reported by Jolliffe and Janssen25 linked to the International Diabetes Federation adult criteria and Adult Treatment Panel III.25 Participants who did not have any of the following cardiometabolic risk factors altered were classified as MHO: high triglycerides levels (>1.44 and >1.60 mmol/L in boys and girls, respectively), high fasting glucose levels (>5.60 mmol/L), low HDL cholesterol (<1.13 and <1.25 mmol/L in boys and girls, respectively), and elevated systolic (>121 mmHg) or diastolic (>76 and >80 mmHg in boys and girls, respectively) blood pressure. Accordingly, MUO were those who had one or more of these cardiometabolic risk factors altered. Waist circumference was excluded as a risk factor following previous literature12,14 since overweight and obese individuals usually present a waist circumference above age- and sex-specific cut-off points for the metabolic syndrome.9,13
Statistical analysis
All statistical analyses were performed using SPSS version 20.0 for Windows (IBM Corp, New York, USA), and the significance level was set at p < 0.05. Data are presented for overweight and obese children. Descriptive analyses were performed by using independent samples T test to detect differences between MHO and MUO within each study (ActiveBrains and EFIGRO).
Analysis of covariance was used to examine differences in normalised (Blom’s transformation) aBMD between MHO and MUO phenotypes. Sex, years from PHV, stature and TBLH lean mass (normalised) were used as covariates (model 1). The contribution of MVPA or CRF was tested in additional models (models 2, and 3, respectively). The effect size (ES, Cohen’s d) on aBMD between MHO and MUO with one cardiometabolic risk factor or with two or more cardiometabolic risk factors using the same models of covariates described above is shown. The interpretation of ES is: 0.2 small, 0.5 medium, and 0.8 large.26
The selection of the covariates was based on the association with the outcome and exposure variables. Sensitivity analyses including sedentary time, vitamin D and dietary calcium intake were done (exploratory analysis). These variables did not have influence on the outcome variables, so they were not included as part of the models. Finally, complementary information running the statistical tests only for obese participants is shown as Supplementary Material.
Results
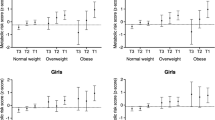
The descriptive characteristics of the MHO and MUO children within each study are shown in Table 1. In the ActiveBrains study, 50.6% of the participants did not present any metabolic risk factors, 28.4% presented 1 and 21% presented at least 2. In the EFIGRO study, 54.2% of the participants did not present any metabolic risk factor, 34.6% presented 1 and 11.2% presented 2. Overall, MUO children weighed more and had more BMI, fat mass, waist circumference and triglycerides and lower HDL cholesterol, levels of MVPA and CRF than MHO children (all p < 0.05). In addition, MUO from the EFIGRO study were more mature and older and had higher glucose levels and lean mass than MHO (all p < 0.05). Finally, aBMD did not differ between MHO and MUO children within each study.
Adjusted normalised values in aBMD between MHO and MUO overweight/obese children
Table 2 shows a comparison of the adjusted normalised values in aBMD between MHO and MUO. In model 1, MHO children had higher aBMD in TBLH (ES = 0.34; p = 0.026), trunk (ES = 0.43; p = 0.005) and pelvis (ES = 0.33; p = 0.029), compared to MUO children. Most differences remained significant after MVPA was added as a new covariate (model 2). Interestingly, most significant differences shown in model 1 disappeared once CRF was accounted for (model 3), except for trunk aBMD (ES = 0.37; p = 0.016).
In an attempt to find out whether differences in aBMD were related with the number of cardiometabolic risk factors altered, MUO children were split into two different groups (1 vs ≥2 cardiometabolic risk factors) and compared against MHO children. Then the ES in aBMD between MHO, MUO with 1 cardiometabolic risk factor and MUO with ≥2 cardiometabolic risk factors were obtained (Fig. 1). Taking MHO as the reference group, generally all ES negatively increased from MUO with 1 cardiometabolic risk factor to MUO with ≥2 cardiometabolic risk factors (model 1). This trend remained similar but with smaller ES when MVPA (model 2) or CRF (model 3) was added into the model. The ES change (model 2 − model 1) explained by the contribution of MVPA ranged from 0.00 to 0.01 in participants with 1 cardiometabolic risk factor and from 0.04 to 0.11 in participants with ≥2 cardiometabolic risk factors. Finally, the ES change (model 3 − model 1) explained by the contribution of CRF ranged from 0.06 to 0.14 in participants with 1 cardiometabolic risk factor and from 0.05 to 0.18 in participants with ≥2 cardiometabolic risk factors.
Sensitivity analysis only with obese children
Descriptive characteristics only for obese participants are shown in Supplementary Table 1. Overall, MUO children had higher levels of triglycerides and lower HDL cholesterol (all p < 0.05) in both studies. In addition, MHO children from the ActiveBrains study spent more time in MVPA than their MUO peers (p < 0.05). Moreover, MHO children from the EFIGRO were less mature than MUO children (p < 0.05). Adjusted normalised values in aBMD (Supplementary Table 2) showed that MHO children had higher trunk aBMD (ES = 0.34, p = 0.025) in model 1, compared to MUO children. The addition of MVPA as a covariate (model 2) did not alter these differences. Finally, the significant differences in trunk BMD disappeared once CRF was accounted for (model 3).
Discussion
To the best of our knowledge, this is the first study reporting differences in aBMD in children with MHO and MUO metabolic profiles. Our findings indicate that (1) MHO children have greater aBMD in TBLH, trunk and pelvis than MUO children and (2) MVPA and, more importantly, CRF seems to partially explain these differences between groups. The ES of the differences observed between MHO and MUO ranged from 0.11 and 0.43, suggesting a small-to-medium ES. Large ESs would have been unexpected owing to the multifactorial aetiology of bone and fat status.
We found only one study examining the association between metabolic profiles and aBMD, and it was carried out in adults.11 The authors concluded that there could be a link between MHO/MUO phenotypes and aBMD, since MUO adults had better bone status at hip site than MHO.11 Albeit in adult population, their findings contradict those observed in our study with children, in which MHO children had better BMD. Both studies are difficult to compare as not only the target population is different but also different confounding variables are taken into account, which may importantly influence the findings. The study by Mirzababaei et al.11 used age, sex and BMI as covariates, while a more complete set of confounding variables was used in the present study, such as sex, years from PHV, stature and TBLH lean mass (model 1). In this regard, the use of BMI as a confounder of bone mass is debatable. BMI is calculated using weight and stature where weight is consisting primarily of fat and lean mass. It is well known that lean mass is the strongest determinant of bone parameters in young population,27 as the development of the muscles produces a higher tension on the bones following the mechanostat theory.28 However, the association between fat and bone mass is debated.6 A study has shown that the association between fat and bone mass disappears once the effect of lean mass is controlled in adolescents.7
In addition, objectively measured MVPA was additionally taken into account in our study (model 2) whose role is clear in the development and maintenance of bone mass29 and is positively associated with better cardiometabolic risk factor profiles in children and adolescents.30 In agreement with our findings, a previous study concluded that MVPA was higher in MHO adolescents compared to MUO adolescents, both in the group with overweight/obese and only obese.14 However, Camhi et al.31 did not find significant differences in MVPA between MHO and MUO adolescents with obesity. These discrepancies may be due to the use of different methods to measure physical activity (objective vs subjective). Even so, owing to the time spent in MVPA is considered a protective factor of cardiometabolic risk factors during youth,14 using MVPA as a covariate in the association between bone mass and metabolic profiles is recommended. This is supported by our data showing greater ES changes explained by the contribution of MVPA in participants with ≥2 cardiometabolic risk factors than in those with 1 cardiometabolic risk factor.
Importantly, we also took into account the effect of CRF (model 3) because of its association with both metabolic profiles12,13,32 and bone parameters.16,33 In our study, the role of CRF is evident from the change in the ES already observed in participants with just one cardiometabolic risk factor. Recently published reviews and updates concluded that a better CRF was considered a characteristic of the MHO individuals and it is considered a paramount in the prognosis of MHO adults and children.13,15 However, a study published about youth showed no significant differences in CRF between metabolic profiles.14 Of note, differences in bone parameters have been reported according to fitness levels in adolescents with those with better CRF having greater bone mineral content in the whole body and extremities than those with poorer CRF.16 Therefore, CRF could be considered a predictor of aBMD in youth27 and its use as a confounder is justified.
With this in mind, the association between the MHO and MUO phenotypes with CRF, and as a consequence with bone parameters, may be due to the known association between CRF with some of the cardiometabolic risk factors, i.e., HDL and triglycerides,34 peculiar of the aforementioned metabolic profiles. In our study, CRF was positively associated with HDL and negatively associated with triglycerides (r = 0.27 and −0.20, respectively, data not shown), which agrees with the only study published with MHO/MUO in European adolescents.32 In addition, changes in CRF were negatively correlated with changes in triglycerides and HDL/total cholesterol ratio in adolescents, even after controlling for age, gender, fat-free mass and pubertal status.35 Also, young adults with high CRF (≥60th percentile) showed lower triglycerides levels and higher HDL levels compared to young adults with low CRF (<20th percentile).36 In this regard, CRF seems to explain the link between metabolic profiles (MHO and MUO) and aBMD in our study with children.
Furthermore, our findings may be explained through the bone-regulating role of hormones and nutrients. A novel study showed that MHO adults had higher serum concentrations of ionised magnesium, dietary magnesium intakes and serum osteocalcin while lower serum parathyroid hormone (PTH) compared to MUO adults.37 Magnesium, together with the thyroid and parathyroid glands support bone health by stimulating the production of calcitonin, known to act as a bone-preserving hormone and regulating PTH.38 It is well known that PTH regulates serum calcium through its effects on bone, kidney and the intestine.38 In the bone, PTH enhances the release of calcium from the large reservoir and leads to bone resorption.38
Some limitations of this study deserve to be commented. First, the results cannot establish a cause–effect relationship due to its cross-sectional design. Second, Supplementary Material data have been obtained using a relatively small sample of obese children (MHO = 54 and MUO = 69) and therefore some of the differences between MHO and MUO are non-significant but borderline. Third, despite the fact that physical activity was objectively measured by tri-axial accelerometers, the obtained data may be affected by various factors and generally produce greater errors than those observed for physical fitness.39 Fourth, data from observational studies generally show a stronger association between fitness and health parameters than for physical activity and health parameters.39 This might also explain why MVPA did not affect the association between bone parameters and metabolic profile to the same extent as CRF.
In conclusion, this novel research shows that MHO children have greater aBMD than their unhealthy peers (MUO). Furthermore, both MVPA and, more importantly, CRF, seem to partially explain these differences. Taking into account that children are in a very sensitive period to changes, further longitudinal studies such as clinical trials and cohort studies are needed. These types of studies would allow a better understanding of the development of bone health in overweight and obese metabolic profiles over time.
References
Nikander, R. et al. Targeted exercise against osteoporosis: A systematic review and meta-analysis for optimising bone strength throughout life. BMC Med. 8, 47 (2010).
Ng, M. et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980-2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet 384, 766–781 (2014).
Baroncelli, G. I., Bertelloni, S., Sodini, F. & Saggese, G. Osteoporosis in children and adolescents. Pediatr. Drugs 7, 295–323 (2005).
Reilly, J. J. & Kelly, J. Long-term impact of overweight and obesity in childhood and adolescence on morbidity and premature mortality in adulthood: systematic review. Int. J. Obes. 35, 891–898 (2011).
Felson, D. T., Zhang, Y., Hannan, M. T. & Anderson, J. J. Effects of weight and body mass index on bone mineral density in men and women: the Framingham study. J. Bone Min. Res. 8, 567–573 (1993).
Shapses, S. A. & Sukumar, D. Bone metabolism in obesity and weight loss. Annu. Rev. Nutr. 32, 287–309 (2012).
Gracia-Marco, L. et al. Adiposity and bone health in Spanish adolescents. The HELENA study. Osteoporos. Int. 23, 937–947 (2012).
Júnior, I. F. F. et al. The relationship between visceral fat thickness and bone mineral density in sedentary obese children and adolescents. BMC Pediatr. 13, 37 (2013).
Ortega, F. B., Lavie, C. J. & Blair, S. N. Obesity and cardiovascular disease. Circ. Res. 118, 1752–1770 (2016).
Primeau, V. et al. Characterizing the profile of obese patients who are metabolically healthy. Int. J. Obes. 35, 971–981 (2011).
Mirzababaei, A., Mirzaei, K., Khorrami-Nezhad, L., Maghbooli, Z. & Keshavarz, S. A. Metabolically healthy/unhealthy components may modify bone mineral density in obese people. Arch. Osteoporos. 12, 95 (2017).
Ortega, F. B. et al. The intriguing metabolically healthy but obese phenotype: cardiovascular prognosis and role of fitness. Eur. Heart J. 34, 389–397 (2013).
Ortega, F. B., Cadenas-Sanchez, C., Sui, X., Blair, S. N. & Lavie, C. J. Role of fitness in the metabolically healthy but obese phenotype: a review and update. Prog. Cardiovasc Dis. 58, 76–86 (2015).
Cadenas-Sanchez, C. et al. Prevalence of metabolically healthy but overweight/obese phenotype and its association with sedentary time, physical activity, and fitness. J. Adolesc. Health 61, 107–114 (2017).
Ortega, F. B. et al. Role of physical activity and fitness in the characterization and prognosis of the metabolically healthy obesity phenotype: a systematic review and meta-analysis. Prog. Cardiovasc. Dis. 61, 190 (2018).
Gracia-Marco, L. et al. Effect of fitness and physical activity on bone mass in adolescents: the HELENA Study. Eur. J. Appl. Physiol. 111, 2671–2680 (2011).
Gracia-Marco, L. et al. Levels of physical activity that predict optimal bone mass in adolescents: the HELENA study. Am. J. Prev. Med. 40, 599–607 (2011).
Cadenas-Sanchez, C. et al. An exercise-based randomized controlled trial on brain, cognition, physical health and mental health in overweight/obese children (ActiveBrains project): rationale, design and methods. Contemp. Clin. Trials 47, 315–324 (2016).
Medrano, M. et al. The effect of a multidisciplinary intervention program on hepatic adiposity in overweight-obese children: protocol of the EFIGRO study. Contemp. Clin. Trials 45, 346–355 (2015).
Cole, T. J. & Lobstein, T. Extended international (IOTF) body mass index cut-offs for thinness, overweight and obesity. Pediatr. Obes. 7, 284–294 (2012).
Moore, S. A. et al. Enhancing a somatic maturity prediction model. Med. Sci. Sports Exerc. 47, 1755–1764 (2015).
Hildebrand et al. group comparability of raw accelerometer output from wrist- and hip-worn monitors. Med. Sci. Sports Exerc. 46, 1816–1824 (2014).
Castro-Piñero, J. et al. Criterion-related validity of field-based fitness tests in youth: a systematic review. Br. J. Sports Med. 44, 934–943 (2010).
Crabtree, N. J. et al. Dual-energy X-ray absorptiometry interpretation and reporting in children and adolescents: the revised 2013 ISCD Pediatric Official Positions. J. Clin. Densitom. 17, 225–242 (2014).
Jolliffe, C. J. & Janssen, I. Development of age-specific adolescent metabolic syndrome criteria that are linked to the Adult Treatment Panel III and International Diabetes Federation criteria. J. Am. Coll. Cardiol. 49, 891–898 (2007).
Cohen, J. Quantitative methods in psychology: a power primer. Psychol. Bull. 112, 155–159 (1992).
Vlachopoulos, D. et al. Determinants of bone outcomes in adolescent athletes at baseline: the PRO-BONE study. Med. Sci. Sports Exerc. 49, 1389–1396 (2017).
Schoenau, E. From mechanostat theory to development of the “Functional Muscle-Bone-Unit”. J. Musculoskelet. Neuronal Interact. 5, 232–238 (2005).
Baxter-Jones, A. D., Kontulainen, S. A., Faulkner, R. A. & Bailey, D. A. A longitudinal study of the relationship of physical activity to bone mineral accrual from adolescence to young adulthood. Bone 43, 1101–1107 (2008).
Prince, R. L., Kuk, J. L., Ambler, K. A., Dhaliwal, J. & Ball, G. D. Predictors of metabolically healthy obesity in children. Diabetes Care 37, 1462–1468 (2014).
Camhi, S. M., Waring, M. E., Sisson, S. B., Hayman, L. L. & Must, A. Physical activity and screen time in metabolically healthy obese phenotypes in adolescents and adults. J. Obes. 2013, 984613 (2013).
Artero, E. G. et al. Muscular and cardiorespiratory fitness are independently associated with metabolic risk in adolescents: the HELENA study. Pediatr. Diabetes 12, 704–712 (2011).
Ubago-Guisado, E., Martinez-Rodriguez, A., Gallardo, L. & Sanchez-Sanchez, J. Bone mass in girls according to their BMI, VO2 max, hours and years of practice. Eur. J. Sport Sci. 16, 1176–1186 (2016).
Ruiz, J. R. et al. Predictive validity of health-related fitness in youth: a systematic review. Br. J. Sports Med. 43, 909–923 (2009).
Janz, K. F., Dawson, J. D. & Mahoney, L. T. Increases in physical fitness during childhood improve cardiovascular health during adolescence: the Muscatine Study. Int. J. Sports Med. 23(Suppl 1), S15–S21 (2002).
Carnethon, M. R. et al. Cardiorespiratory fitness in young adulthood and the development of cardiovascular disease risk factors. JAMA 290, 3092–3100 (2003).
Sukumar, D. et al. Can bone-regulating hormones and nutrients help characterize the metabolically healthy obese phenotype. Nutr. Health 24, 153 (2018).
Shaker, J. L. & Deftos, L. in Endotext (eds De Groot, L. J. et al.) (MDText.com, Inc., South Dartmouth, MA, 2000).
Blair, S. N., Cheng, Y. & Holder, J. S. Is physical activity or physical fitness more important in defining health benefits? Med. Sci. Sports Exerc. 33, S379–S399 (2001).
Acknowledgements
The authors thank the staff and participants of the ActiveBrains and EFIGRO studies for their important contributions. This study has been partially funded by the University of Granada, UGR Research and Knowledge Transfer Fund (PPIT) 2016, Excellence Actions Programme: Units of Scientific Excellence; Scientific Unit of Excellence on Exercise and Health (UCEES), and by the Regional Government of Andalusia, Regional Ministry of Economy, Knowledge, Enterprises and University and European Regional Development Fund (ERDF), ref. SOMM17/6107/UGR. The ActiveBrains study was funded by the Spanish Ministry of Economy and Competitiveness (Reference DEP2013-47540, DEP2016-79512-R, and DEP2017-91544-EXP). The EFIGRO study was funded by the Spanish Ministry of Health, “Fondo de Investigación Sanitaria del Instituto de Salud Carlos III” (PI13/01335), “Fondos Estructurales de la Unión Europea (FEDER), Una manera de hacer Europa,” and by the University of the Basque Country (GIU14/21). L.G.-M. is supported by “La Caixa” Foundation within the Junior Leader fellowship programme (ID 100010434); M.M. is supported by the Spanish Ministry of Education, Culture and Sport (FPU14/03329) and EST17/00210; L.A. is supported by the Education Department of the Government of the Basque Country (PRE_2016_1_0057, PRE_2017_2_0224 and PRE_2018_2_0057); J.M.-G. and J.H.M. are supported by the Spanish Ministry of Education, Culture and Sport (FPU14/06837 and FPU15/02645, respectively).
Author information
Authors and Affiliations
Contributions
M.M., C.D.-S., L.A., J.H.M., J.M.-G. and M.M.-M. obtained the data. E.U.-G., M.M. and J.M.-G. analysed the data. E.U.-G. drafted the manuscript under the supervision of I.L. and F.B.O. (principal investigators). L.G.-M., I.T., V.E.-M. and M.O. contributed and edited the manuscript. E.U.-G. had full access to the data in the study and final responsibility for the decision to submit for publication. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Ubago-Guisado, E., Gracia-Marco, L., Medrano, M. et al. Differences in areal bone mineral density between metabolically healthy and unhealthy overweight/obese children: the role of physical activity and cardiorespiratory fitness. Pediatr Res 87, 1219–1225 (2020). https://doi.org/10.1038/s41390-019-0708-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-019-0708-x
This article is cited by
-
Fat as a Friend or Foe of the Bone
Current Osteoporosis Reports (2024)
-
Fat-free/lean body mass in children with insulin resistance or metabolic syndrome: a systematic review and meta-analysis
BMC Pediatrics (2022)
-
Distinct whole-blood transcriptome profile of children with metabolic healthy overweight/obesity compared to metabolic unhealthy overweight/obesity
Pediatric Research (2021)