Abstract
Drugs of abuse regulate the activity of the mesolimbic dopamine (DA) system, and drug-induced changes in ventral tegmental area (VTA) cellular activity and gene regulation are linked to behavioral outputs associated with addiction. Previous work from our lab determined that VTA serum- and glucocorticoid-inducible kinase 1 (SGK1) transcription and catalytic activity were increased by repeated cocaine administration; however, it was unknown if these biochemical changes contributed to cocaine-elicited behaviors. Using transgenic and viral-mediated manipulations, we investigated the role of VTA SGK1 catalytic activity in regulating cocaine conditioned place preference and self-administration. We showed intra-VTA infusion of a catalytically inactive SGK1 mutant (K127Q) significantly decreased cocaine conditioned place preference (CPP). Further, we found that K127Q expression in VTA DA neurons significantly decreased cocaine CPP, while this same manipulation in VTA GABA neurons had no effect. However, blunted VTA DA SGK1 catalytic activity did not alter cocaine self-administration. Altogether, these studies identify the specific VTA cells critical for SGK1-mediated effects on cocaine CPP but not self-administration.
Similar content being viewed by others
Introduction
Serum- and glucocorticoid-inducible kinase 1 (SGK1) was initially identified as an immediate early gene transcriptionally induced by serum and glucocorticoids [1]. This early work was critical in defining SGK1 as a serine/threonine kinase and member of the AGC family of kinases, which achieve maximal catalytic activity though phosphorylation of the activation segment and hydrophobic motif [2,3,4,5] and share a highly conserved ATP-binding motif [2]. For SGK1, these phosphorylation events occur at Ser422 and Thr256, respectively [5, 6], and Lys127 is critical for ATP binding, as mutations at this site produce a catalytically inactive protein [5, 7,8,9]. SGK1 catalytic activity regulates a variety of ion channels, transporters, and transcription factors in the periphery [10]; however, far less is known about the functional role of SGK1 in the central nervous system.
SGK1 mRNA and protein is expressed throughout the brain [11] and transcription can be induced by learning, stress, seizures, and drugs of abuse [12,13,14,15,16,17,18,19]. However, nearly all information characterizing sgk1 mRNA regulation was collected as part of large-scale screening investigations, with few studies expanding upon transcript findings with protein-level assays. Given that changes in transcription may not directly parallel modifications in protein expression or activity, it is critical to investigate SGK1 protein regulation. Importantly, SGK1 activity modulates learning- and stress-related behaviors. In the hippocampus, training in the Morris water maze task increased SGK1 Thr256 and Ser422 phosphorylation [20], phospho-sites required for maximal kinase activity [21]. Moreover, mice overexpressing SGK1 mutants at these sites (T256A or S422A) exhibited impaired context-dependent learning [19, 20, 22, 23], indicating that hippocampal SGK1 activity is critical for spatial learning. In addition, stress regulates SGK1 protein in the prefrontal cortex (PFC). In a model of learned helplessness, total SGK1 protein was decreased in rats showing greater escape failures, and PFC overexpression of S422A SGK1 increased the number of escape failures [18]. In contrast, acute forced swim stress increased PFC SGK1 protein, and blocking SGK1 function via protein interference (TAT-SGK1) prevented increased performance in a T-maze task, suggesting SGK1 activity promotes stress-induced enhancement of working memory [24]. While these studies highlight opposing SGK1 regulation by different forms of stress, together with hippocampal findings, they implicate SGK1 catalytic activity in the central nervous system as an important mediator of learning and stress behaviors.
Though SGK1 function in response to learning and stress has been examined, relatively little is known about SGK1 catalytic activity in the context of drugs of abuse. Using RNA-sequencing, we previously identified SGK1 as one of only five genes transcriptionally upregulated in the ventral tegmental area (VTA) by both repeated cocaine and morphine treatment [13]. VTA SGK1 catalytic activity was also increased following repeated, but not acute, administration of cocaine [13]; however, the functional relevance of VTA SGK1 catalytic activity remains unknown. Thus, in this study we sought to determine whether blunted SGK1 catalytic activity in VTA attenuated cocaine-elicited behaviors. Moreover, given that the VTA is a heterogenous brain region [25, 26], we utilized cell type-specific approaches to determine the neuronal population/s critical for driving SGK1 effects on behavior.
Materials and methods
Animals
Herpes simplex virus (HSV) overexpression experiments used adult (8–9 weeks) male c57BL/6 J mice (Jackson Laboratory) allowed to habituate to the animal facility >7 days prior to the start of experiments. Cell type-specific experiments utilized adult (8–15 weeks) male and female heterozygous dopamine transporter (DAT)-Cre recombinase (Cre) mice (Jackson Laboratory, 006660) or vesicular GABA transporter (VGAT)-Cre mice (Jackson Laboratory, 028862), bred inhouse. DA-SGK1-KO mice (Supp Fig. 6) were generated via crossing homozygous floxed SGK1 (FlxSGK1) mice [27, 28] with DAT-Cre mice.
All mice were housed at 22–25 °C on a 12 h light–dark cycle with food and water ad libitum. Behavioral testing took place during the light phase, unless otherwise noted. All experiments were approved by Michigan State University Institutional Animal Care and Use Committee and were carried out in accordance with the guidelines set in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health.
Viral constructs
HSV vectors encoding green fluorescent protein (GFP), HA-tagged SGK1 (wild-type SGK1), catalytically inactive K127Q mutant, and N-terminal phosphorylation mutants (S78A, S78D) have been previously used and validated [13, 29]. HSVs (HSV-CMV-GFP, HSV-HA-SGK1-WT-GFP, HSV-HA-SGK1-K127Q-GFP, HSV-HA-SGK1-S78A-GFP, HSV-HA-SGK1-S78D-GFP) were packaged by Dr. Rachael Neve at the Gene Technology Core (Massachusetts General Hospital). AAVs were packaged by the University of North Carolina GTC Vector Core (AAV2-EF1α-DIO-HA-SGK1-K127Q, AAV2-EF1α-DIO-EYFP-WPRE-pA). Expression of HA-tagged SGK1 mutants in the VTA was validated using standard western blot and IHC methods (see supplemental methods).
Viral-mediated gene transfer and stereotaxic surgery
Stereotaxic surgeries used established procedures [13, 30]. Adult mice (8–10 weeks) were anesthetized using 100 mg/kg ketamine and 10 mg/kg xylazine. All mice received bilateral intra-VTA infusions (HSV: 0.5 μl, AAV: 0.4 μl) at established coordinates (−3.2 mm A/P,+1.0 mm M/L,−4.6 mm D/V, 7° angle). Behavioral experiments began either 1 day (HSV) or >21 days (AAV) following surgery.
For AAV experiments, control mice consisted of Cre-negative mice that received AAV-DIO-SGK1-K127Q and Cre-positive mice that received AAV-DIO-EYFP; control data were combined as there were no differences between groups.
Viral targeting and immunohistochemistry (IHC) to verify viral expression
VTA expression of SGK1 was confirmed using standard IHC methods [31]. Viral-mediated expression of SGK1 was verified via IHC for HA-tagged SGK1 and TH was used to assess whether expression was localized to dopamine neurons. VTA targeting was confirmed by a blinded observer following behavioral testing. Slices between −3.64 and −3.08 mm from bregma across the anterior/posterior axis were visualized for bilateral viral expression within the VTA. Mice were excluded from analysis if viral expression was unilateral or outside of the VTA (Supp Fig. 7). Across all experiments, a total of 82 of 448 mice were excluded due to missed injection or lack of fluorescent signal.
Drugs
Cocaine (Sigma) was dissolved in 0.9% sterile saline for all experiments. Dose varied depending on experiment and sex. This included 7.5 mg/kg cocaine for HSV CPP, 12.5 mg/kg cocaine for male AAV CPP and 10 mg/kg cocaine for female AAV CPP; a lower drug dose was selected for females to produce comparable preference scores to males [32]. For locomotor activity, 7.5 mg/kg and 5 mg/kg were used for males and females, respectively. For self-administration experiments, 0.5 mg/kg/infusion of cocaine was used for acquisition and progressive ratio assays. Cocaine doses of 1.0, 0.5, 0.25, 0.063, 0.016, and 0.001 mg/kg/infusion were used during dose–response testing. For biochemical studies, 20 mg/kg cocaine was used.
Cocaine conditioned place preference (CPP)
Cocaine CPP was performed using established procedures [13]. Briefly, mice were placed in the center of a three-chambered CPP box (San Diego Instruments) and allowed to freely explore for a 20 min pretest. Mice that spent ≥20% of total time more in one side chamber than the other during the pretest session were excluded from analysis due to the presence of an initial side bias. Across all experiments, 31 of 351 total mice assessed in CPP were excluded due to pretest side bias. Next, during unbiased conditioning sessions, mice were restricted to one chamber in the morning (saline, 30 min) and the opposite chamber in the afternoon (cocaine, 30 min) for two conditioning days, followed by a 20 min posttest. CPP score was calculated as the time (seconds) spent in the drug-paired minus saline-paired chamber. Total beam breaks (from y-photodetectors only) during each conditioning session were used to calculate locomotor activity, and 2-day averages were used to analyze locomotor activity during the conditioning sessions.
Cocaine locomotor activity
Locomotor activity in response to repeated cocaine injections was performed as previously described with minor modifications [13]. Locomotor activity was measured in a three-chambered CPP box (San Diego Instruments) and calculated as total beam breaks (4 × 16 (x-y) photobeam array). One day prior to the start of the experiment, mice were habituated to the boxes for 60 min. Next, in daily testing sessions (7 days) mice were first i.p. injected with saline and locomotor behavior was recorded for 30 min, this was immediately followed by cocaine injection and an additional 30 min recording of locomotor behavior.
Cocaine self-administration
Cocaine self-administration protocols were adapted from previous methods [33, 34]. Due to the consistently more robust cocaine CPP and locomotor effects in DAT-K127Q male mice and the difficulty in performing self-administration in female mice, only male mice were used for self-administration studies. Mice underwent self-administration experiments in operant boxes (MedAssociates). Selecting the active nose poke resulted in the delivery of a reward and a concurrent light cue (nose poke illumination and house light). Action on the inactive nose poke had no consequences. Mice were first trained to respond for sucrose pellets (food training). Briefly, mice were food restricted to 90% of their free feeding weight and sucrose self-administration was conducted on a fixed ratio of 1 (FR1) schedule in 1 hr sessions for 7 days. Mice that did not acquire were removed from the experiment (a total of 9/42 mice were removed due to not meeting the acquisition criteria). Following food training, mice were anesthetized (100 mg/kg ketamine, 10 mg/kg xylazine) and implanted with a chronic indwelling jugular catheter. Following surgery, mice were allowed to recovery for 3 days. Catheter patency was assessed at the start and end of each experiment using 20 μl Brevital (5 mg/mL in 0.9% sterile saline), and catheters were considered functional if the animal lost muscle tone in <5 seconds from the time of infusion. Cocaine self-administration was conducted (19.6 μl infusion, 0.5 mg/kg/infusion) on a FR1 schedule in 2 h session for 7–10 days. Following acquisition of cocaine self-administration, separate cohorts of mice underwent self-administration (19.6 μl infusions, 0.5 mg/kg/infusion) on a progressive ratio (PR) schedule in a 2 hr session [35] or a dose–response analysis where mice self-administered a descending set of doses (19.6 μl infusion, 1.0, 0.5, 0.25, 0.063, 0.016, and 0.001 mg/kg/infusion) on an FR1 schedule in 2 h sessions (method adapted from [34, 36]). Each dose was run for two consecutive days, and the average of responses at each dose were used for analysis.
Two-bottle sucrose choice (TBC) task
To assess preference, mice were singly housed with access to two 50 mL conical tubes fitted with sipper tops following established procedures [17, 30]. Briefly, bottle volume was measured at the same time each morning and bottle placement was swapped daily to account for potential side bias. Mice that displayed a > 30% side/bottle preference during testing were excluded from analysis. Following water/bottle habituation (1–2 days), sucrose preference (1% sucrose or water) was assessed (4 days). Fluid intake (mL) was measured daily and results are reported as percent preference (sucrose consumed/total fluid consumed x 100, calculated for each day), average preference (4-day average of daily percent sucrose preference), and average fluid intake (4-day averages of fluid intake).
Statistics
Full statistical analyses are reported in Supplemental Table 1. GraphPad Prism was used for all statistical analyses, and all values are represented as mean ± SEM. The means of two groups were compared using an unpaired t-test (two-tailed), and the means of three groups were compared using a one-way analysis of variance (ANOVA), followed by a Dunnett post hoc test when appropriate. A two- or three-way ANOVA was used when comparing two or three variables, and an ANOVA with repeating measures (RM) was used when analyses were repeated on multiple days. ANOVAs were followed by Tukey or Sidak post hoc tests when appropriate. Males and females were always analyzed separately for behavior. Significance was defined as p < 0.05.
Additional methodological details are presented in Supplemental Methods.
Results
Blunted VTA SGK1 catalytic activity decreases cocaine CPP
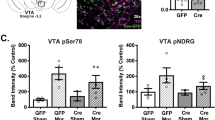
Repeated, but not acute, cocaine administration increases VTA SGK1 catalytic activity [13]; however, the functional relevance of SGK1 activity remains unknown. Therefore, we generated an SGK1 mutant with an amino acid substitution at Lys127 to create a catalytically inactive mutant (K127Q) [9, 29]. To validate this viral approach, we injected a herpes simplex virus (HSV) overexpressing K127Q (HSV-SGK1-K127Q) or mutant constructs that alter the SGK1 N-terminal phosphorylation site (HSV-SGK1-S78D, HSV-SGK1-S78A) into the VTA (Fig. 1A). We then performed western blot analysis on microdissected VTA tissue to assess changes in SGK1 Ser78 phosphorylation and SGK1 catalytic activity (pNDRG) compared to HSV-GFP infused controls. We found that HSV-K127Q significantly increased pS78-SGK1 signal (Fig. 1B; F(3,38) = 15.41, p < 0.0001; GFP vs K127Q: p < 0.0001) consistent with SGK1 protein overexpression, as the K127Q mutation is not expected to impact Ser78 phosphorylation. In contrast, HSV-S78A and HSV-S78D pS78-SGK1 levels do not differ from HSV-GFP controls (Fig. 1B; GFP vs S78A: p = 0.49; GFP vs S78D: p = 0.69), validating specificity of the pSer78-SGK1 antibody to recognize the phosphorylated Ser78 site. SGK1 catalytic activity was assessed through phosphorylation of its specific substrate, NDRG [37]. As expected, HSV-S78A and HSV-S78D both significantly increased pNDRG levels compared to HSV-GFP controls (Fig. 1C; F(3,38) = 8.22, p = 0.0002; GFP vs S78A: p = 0.0006; GFP vs S78D: p = 0.01), similar to previously observed effects with VTA SGK1 overexpression [13]. In contrast, pNDRG levels did not differ between HSV-K127Q and HSV-GFP controls despite the overexpression of SGK1 protein (p = 0.99), validating that K127Q is catalytically inactive. We also assessed whether VTA expression of K127Q could alter cocaine-induced increases in SGK1 catalytic activity. A “binge” cocaine protocol (5–20 mg/kg injections over 3 days [38, 39]) was used as it has previously been shown to increase VTA SGK1 catalytic activity [13] and allowed repeated cocaine administration within the abbreviated HSV expression timeframe. While there was a trend for higher pNDRG levels in the GFP-cocaine group (140.4 ± 19.3) compared to the K127Q group (107.5 ± 12.1) and GFP-saline controls (100.0 ± 10.2), we did not detect a significant difference in pNDRG signal (Supp Fig. 1; F(2,30) = 2.09, p = 0.14). Together, these data support that K127Q is catalytically inactive and may be able to blunt cocaine-induced responses in the VTA, and therefore be a useful tool for assessing the effect of constrained VTA SGK1 catalytic activity on cocaine-elicited behaviors.
A Representative image of VTA HSV-HA-SGK1-K127Q viral expression. B VTA overexpression of a catalytically inactive SGK1 mutant (HSV-HA-SGK1-K127Q) significantly increased phospho-SGK1 (Ser78) compared to GFP controls (GFP = 14, K127Q = 12, one-way ANOVA followed by a Dunnett post hoc test), consistent with increased SGK1 expression, while overexpression of Ser78 mutants (HSV-HA-SGK1-S78A = 8 and HSV-HA-SGK1-S78D = 8) showed no difference from controls, highlighting antibody specificity. C In contrast, K127Q expression did not increase phosphorylation of the SGK1 substrate NDRG above control levels (GFP = 14, K127Q= = 12) while expression of S78A and S78D mutants both significantly increased pNDRG compared to GFP controls (S78A = 8, S78D = 8, one-way ANOVA followed by a Dunnett post hoc test). Representative blots are shown above each graph. D VTA K127Q expression decreased cocaine CPP compared to GFP controls while WT SGK1 had no effect (7.5 mg/kg cocaine, GFP = 22, K127Q = 26, WT = 11, two-way ANOVA followed by a Tukey post hoc test). E While there was a main effect of drug, locomotor activity during the CPP conditioning sessions was not changed by either expression of the K127Q mutant or WT SGK1 (7.5 mg/kg cocaine, GFP = 22, K127Q = 26, WT = 11, two-way ANOVA followed by a Tukey post hoc test). *p < 0.05, **p < 0.01. All data are represented as mean ± SEM.
HSV expressing the catalytically inactive SGK1 mutant (K127Q), wild-type SGK1 (WT), or control GFP was bilaterally injected into the VTA of adult male mice, and cocaine CPP was assessed (Fig. 1A). Following conditioning, we found significant main effects of conditioning (F(1,112) = 15.77, p = 0.0001), virus (F(2,112) = 4.34, p = 0.02), and interaction of conditioning x virus (F(2,112) =3.2, p = 0.02). Time spent in the drug paired chamber was significantly decreased in mice with VTA K127Q expression compared to GFP controls (Fig. 1B; p = 0.02). WT SGK1 mice did not differ from GFP controls (Fig. 1B; p = 0.99), suggesting that VTA overexpression of SGK1 protein alone does not impact CPP. Cocaine injections caused an expected increase in average locomotor activity during the conditioning sessions (Fig. 1C; main effect of drug: F(1,112) = 12.48, p = 0.0006), but no main effect of virus was observed (F(2,112) = 0.53, p = 0.59). These findings indicate that blunted VTA SGK1 catalytic activity is sufficient to impair cocaine CPP.
K127Q expression in VTA DA, but not GABA, neurons alters cocaine CPP and psychomotor activity
Given that SGK1 catalytic activity is capable of regulating cocaine CPP, we next sought to determine if these effects were cell type-specific. The VTA is a heterogenous brain region made up of ~60% DA and ~30% GABA neurons [25, 26]. We generated a novel Cre-dependent K127Q construct and performed VTA injections in adult male and female DAT-Cre or VGAT-Cre mice to blunt SGK1 catalytic activity specifically in VTA DA or GABA neurons, respectively (Fig. 2A, B). In DAT-Cre mice, following conditioning male mice showed significant main effects of conditioning (F(1,46) = 14.08, p = 0.0005), virus (F(1,46) = 8.75, p = 0.005), and an interaction of conditioning x virus (F(1,46) = 7.49, p = 0.009). Female mice displayed a significant main effect of conditioning (F(1,54) = 35.70, p < 0.0001) and an interaction of conditioning x virus (F(1,54) = 7.37, p = 0.009) but only a trend for a main effect of virus (F(1,54) = 2.87, p = 0.096). When K127Q was overexpressed in VTA DA neurons, both male and female mice spent significantly less time in the drug-paired chamber compared to controls following cocaine conditioning (Fig. 2C; male: p = 0.001; female: p = 0.02). In VGAT-Cre mice, both male and female mice showed a significant main effect of conditioning (male: F(1,88) = 27.82, p < 0.0001; female: F(1,76) = 12.40, p = 0.0007) but not virus (male: F(1,88) = 0.074, p = 0.79; female: F(1,76) = 2.08, p = 0.15). In contrast to DAT-Cre mice, both male and female mice with K127Q expression in VTA GABA neurons did not differ from controls (Fig. 2E; male: p = 0.99; female: p = 0.25). Together, these data indicate that reduced catalytic activity in VTA DA, but not GABA, neurons likely drove behavioral results observed with HSV tools (Fig. 1D, E).
A Representative image of AAV-DIO-HA-SGK1-K127Q viral expression in VTA DAT-Cre mice. B Representative image of AAV-DIO-HA-SGK1-K127Q viral expression in VTA VGAT-Cre mice. C Expression of a catalytically inactive SGK1 mutant (K127Q) in VTA DA neurons significantly reduced cocaine CPP in male and female mice (male: 12.5 mg/kg cocaine, n = 10–15, two-way ANOVA followed by a Tukey post hoc test; female: 10 mg/kg cocaine, n = 13–16, two-way ANOVA followed by a Tukey post hoc test). D During CPP conditioning, male and female mice overexpressing VTA DA-specific K127Q displayed a significant increase in cocaine-elicited activity (male: 12.5 mg/kg cocaine, n = 10–15, two-way ANOVA followed by a Tukey post hoc test; female: 10 mg/kg cocaine, n = 13–16, two-way ANOVA followed by a Tukey post hoc test). E Expression of VTA GABA-specific K127Q did not alter cocaine conditioned place preference (male: 12.5 mg/kg cocaine, n = 21–25, two-way ANOVA followed by a Tukey post hoc test; female: 10 mg/kg cocaine, n = 16–24, two-way ANOVA followed by a Tukey post hoc test). F Locomotor activity was not changed by VTA GABA neuron expression of the K127Q mutant in male or female mice (male: 12.5 mg/kg cocaine, n = 21–25, two-way ANOVA followed by a Tukey post hoc test; female: 10 mg/kg cocaine, n = 16-24, two-way ANOVA followed by a Tukey post hoc test). *p < 0.05, **p < 0.01. All data are represented as mean ± SEM.
In addition to decreased cocaine CPP, mice with VTA DA-K127Q expression showed increased psychomotor responses to cocaine. Specifically, male mice displayed main effects of drug (F(1,46) = 19.72, p < 0.0001), virus (F(1,46) = 16.08, p = 0.0002), and an interaction of drug x virus (F(1,46) = 4.36, p = 0.04). Female mice showed main effects of drug (F(1,54) = 37.62, p < 0.0001) and virus (F(1,54) = 4.60, p = 0.04) in average locomotor activity recorded during the two conditioning sessions. DA-specific expression of K127Q elicited enhanced psychomotor effects in both male and female mice (Fig. 2D; male: p = 0.0005, female: p = 0.04). In GABA-specific studies, however, while cocaine produced the expected increase in locomotor activity in males and females (Fig. 2F; males: F(1,88) = 39.85, p < 0.0001; females: F(1,76) = 32.39, p < 0.0001) there was no main effect of virus (males: F(1,88) = 1.24, p = 0.27; females: F(1,76) = 0.09, p = 0.76).
To further investigate the enhanced psychomotor activity driven by cell type-specific K127Q expression, a cohort of mice received repeated cocaine injections to determine if the viral effect on locomotor activity adapted over multiple drug administrations. Male VTA DA-specific K127Q expression robustly increased cocaine-induced locomotor activity (Fig. 3A, Supp Fig. 2; main effects of drug: (F(1,23) = 52.32, p < 0.0001), virus: (F(1,23) = 19.08, p = 0.0002), and drug x virus: (F(1,23) = 13.37, p = 0.001)) and females with VTA DA-specific K127Q expression showed a main effect of drug (Fig. 3A, F(1,21) = 6.02, p = 0.02) and an interaction of day x drug (F(1,21) = 9.53, p = 0.006); however, there was only a non-significant trend for a virus main effect on psychomotor activity (F(1,21) = 4.11, p = 0.06). Importantly, mice with K127Q expression in VTA GABA neurons again showed an expected main effect of cocaine treatment (Fig. 3B; males: F(1,10) = 8.08, p = 0.02; females: F(1,10) = 6.99, p = 0.03), but viral expression did not significantly affect locomotor activity (males: F(1,10) = 0.58, p = 0.46; females: F(1,10) = 0.52, p = 0.49). These data replicated the psychomotor effect originally observed during CPP conditioning of male, but not female, DA manipulated mice (Fig. 2D). As these locomotor assays were performed at lower cocaine doses than CPP conditioning, we next sought to determine if CPP conditioned at a lower dose was similarly altered by VTA DA SGK1 manipulation.
A VTA DA K127Q expression (AAV-DIO-HA-SGK1-K127Q) altered psychomotor activity in male, but not female, mice (male: 12.5 mg/kg cocaine, three-way ANOVA with repeated measures; female: 10 mg/kg cocaine, n = 10–13, three-way ANOVA with repeated measures). B A main effect of drug was observed in male and female mice with VTA GABA K127Q expression (AAV-DIO-HA-SGK1-K127Q), but no effect of virus was detected (male: 12.5 mg/kg cocaine, n = 6, three-way ANOVA with repeated measures: p = 0.0175; female: 10 mg/kg cocaine, n = 6, three-way ANOVA with repeated measures). *p < 0.05, **p < 0.01. All data are represented as mean ± SEM.
When conditioned at a low dose of cocaine, a main effect of conditioning was observed (males: F(1,36) = 7.30, p = 0.01; females: F(1,32) = 9.94, p = 0.004); however, differences in cocaine CPP scores were not observed in mice with VTA DA neuron-specific K127Q expression (Supp Fig. 3A; main effect of virus in males: F(1,36) = 1.12, p = 0.29 and females: F(1,32) = 0.03, p = 0.86), suggesting that effects on cocaine CPP may be dose-dependent. Interestingly, while no main effect of viral expression on CPP was observed at this dose (male: F(1,36) = 1.12, p = 0.29; female: F(1,32) = 0.03, p = 0.86), male K127Q mice still exhibited a robust increase in psychomotor activity during conditioning sessions, with main effects of drug (F(1,36) = 4.80, p = 0.04) and virus (F(1,36) = 11.05, p = 0.002) detected. Specifically, K127Q-expressing mice had significantly increased activity in response to cocaine compared to controls (p = 0.02). In contrast, females showed a main effect of drug (Supp Fig. 3B; F(1,36) = 6.23, p = 0.02) but not virus (F(1,36) = 0.05, p = 0.82). We also tested a higher dose of cocaine in female mice with VTA DA-K127Q expression, since the locomotor effect appeared more robust in male mice (Fig. 3). We observed significant main effects of conditioning (Supp Fig. 4A; F(1,14) = 14.95, p = 0.002) and drug (Supp Fig. 4B; F(1,14) = 104.5, p < 0.0001) in CPP and locomotor activity assays, respectively, but no effect of virus (Supp Fig. 4A; F(1,14) = 0.19, p = 0.67, Supplementary Fig. 4B; F(1,14) = 0.0007, p = 0.98).
K127Q expression in VTA neurons does not alter sucrose preference
Expanding on results demonstrating impaired cocaine CPP driven by blunted VTA SGK1 catalytic activity, we next sought to determine if a responses to a non-drug reward were also affected. Thus, we assessed sucrose preference in a two-bottle choice task following viral manipulations. Non-cell type-specific expression of HSV-K127Q did not significantly alter sucrose preference (Fig. 4A; main effect of virus: F(1,11) = 3.45, p = 0.09). Similarly, both male and female mice with VTA DA-specific K127Q overexpression did not show differences in sucrose preference from controls (Fig. 4B; main effect of virus; males: F(1,25) = 1.43, p = 0.24; females: F(1,30) = 1.55, p = 0.22). Neither viral manipulation altered total fluid consumption compared to controls (Supp Fig. 5A, B; HSV: t = 0.82, df = 11, p = 0.43; male DAT-Cre: t = 0.93, df = 25, p = 0.36; female DAT-Cre: t = 0.93, df = 25, p = 0.62). These data suggest that blunted VTA SGK1 catalytic activity may specifically impair the rewarding effects of drugs.
A VTA expression of catalytically inactive SGK1 (VTA-K127Q; HSV-HA-SGK1-K127Q) did not alter daily or average sucrose preference in male mice compared to controls (GFP) (n = 6–7, two-way ANOVA with repeated measures). B Daily or average sucrose preference of male or female mice was not changed by K127Q expression in VTA DA neurons (DA-K127Q; AAV-DIO-HA-SGK1-K127Q) (males: n = 12–15, two-way ANOVA with repeated measures; females n = 14–18, two-way ANOVA with repeated measures). *p < 0.05, **p < 0.01. All data are represented as mean ± SEM.
A When comparing all mice run through the initial food training phase, there was no difference between groups (AAV-DIO-HA-SGK1-K127Q or controls) on the number of mice to acquire the operant behavior (n = 20–22, Log-rank (Mantel–Cox) test). B Of mice that successfully acquired cocaine self-administration, VTA DA K127Q-expressing mice did not alter the number of rewards earned during food training (n = 10–11, two-way ANOVA with repeated measures). C Acquisition of sucrose pellet self-administration was not altered by VTA DA K127Q expression compared to controls (n = 10–11, unpaired t-test). D Expression of K127Q in VTA DA neurons did not change cocaine intake compared to controls (0.5 mg/kg/infusion cocaine, n = 10–11, two-way ANOVA with repeated measures). E Breakpoint in a progressive ratio test was not different between mice with VTA DA K127Q expression and controls (0.5 mg/kg/infusion cocaine, n = 5–6, unpaired t-test). F Responding for cocaine during a dose–response assay showed a main effect of drug dose, but K127Q expression did not change responding compared to controls (n = 7–8, two-way ANOVA with repeated measures). *p < 0.05, **p < 0.01. All data are represented as mean ± SEM.
Cocaine self-administration is not altered by VTA DA K127Q expression
To better define changes in cocaine response driven by blunted SGK1 activity in VTA DA neurons, cocaine self-administration was used to assess additional cocaine-elicited behaviors. Male mice with VTA DA neuron-specific expression of the K127Q mutant were first trained to respond for food, where sucrose pellets were earned on a fixed ratio of 1 (FR1) schedule, and mice that did not meet criteria for acquisition of food training were excluded from analysis and further experimentation. Both control mice and those expressing the K127Q mutant acquired the operant behavior, with no observed differences between groups in the percentage of mice to meet acquisition criteria (Fig. 5A; Chi square = 0.21, p = 0.65). In addition, VTA DA K127Q expression did not significantly alter the number of days to meet acquisition (Fig. 5C; t = 0.99, df = 19, p = 0.34) or the number of rewards earned during sucrose self-administration sessions (Fig. 5B; main effect of virus: F(1,19) = 3.34, p = 0.08), consistent with sucrose preference findings and indicating that the processing of natural reinforcement is intact in DA SGK1 mutant-expressing mice.
To assess potential changes to cocaine behavior, mice then underwent cocaine self-administration on an FR1 schedule. Both control and VTA DA SGK1 mutant mice acquired cocaine self-administration and the number of cocaine infusions earned was not different between groups (Fig. 5D; main effect of virus: F(1,19) = 1.53, p = 0.23), indicating that blunted VTA DA SGK1 activity did not modulate cocaine intake. Next, to determine if motivation to earn a cocaine reward was altered by K127Q expression, mice were put through progressive ratio (PR) testing. The PR breakpoint was not different between control and K127Q-expressing mice (Fig. 5E; t = 0.17, df = 9, p = 0.87), suggesting that blunted catalytic activity in VTA DA neurons did not alter motivation to obtain cocaine infusions. Finally, given deficits in cocaine CPP at a moderate but not low dose of cocaine, a dose–response assay was completed to determine if effects on self-administration were potentially obscured by the dose administered during acquisition and PR testing [34, 36]. There was a significant main effect of dose on the number of cocaine infusions earned (Fig. 5F; F(2.649, 34.44) = 23.07, p < 0.0001); however, no differences were observed between DA SGK1 K127Q-expressing mice and controls (main effect of virus: F(1,13) = 1.51, p = 0.24). Thus, while cocaine-elicited behaviors were altered in a CPP paradigm, reduced VTA DA SGK1 catalytic activity did not affect cocaine self-administration behaviors.
VTA K127Q expression in DA SGK1 gene knockout mice does not alter cocaine CPP
While viral-mediated overexpression of a catalytically inactive SGK1 mutant (K127Q) has been a critical tool for understanding the role of SGK1 activity in cocaine behaviors, this method overexpresses the mutant on top of endogenous SGK1 protein expressed by the neuron. In order to assess the potential contribution of endogenous SGK1 signaling on the observed cocaine CPP effects, DA-SGK1-KO male mice were utilized to assess the effects of VTA DA-specific expression of the K127Q mutant in a null background. Following conditioning, a main effect of conditioning was observed (F(1,30) = 8.33, p = 0.007) but not SGK1 manipulation (F(2,30) = 0.01, p = 0.99). Mice with DA neuron-specific SGK1 deletion (DA-SGK1-KO group) exhibited preference similar to that of controls (Supp Fig. 6A, p = 0.99), replicating previous findings [28]. When the K127Q mutant was expressed in VTA DA-SGK1-KO mice (KO-K127Q group), preference did not differ from controls (Supp Fig. 6A, p = 0.99), diverging from impaired CPP effects seen with DA-K127Q expression with endogenous SGK1 (Fig. 2C). During the CPP conditioning sessions, there was a main effect of drug (Supp Fig. 6B; F(1,30) = 10.11, p = 0.003) but not SGK1 manipulation (F(2,30) = 0.03, p = 0.97) on locomotor activity. Accordingly, K127Q expression in VTA DA-SGK1-KO mice failed to enhance psychomotor activity (p > 0.99) in contrast to DA-K127Q mice with endogenous SGK1 protein (Fig. 2D). These data suggest that the effect of K127Q expression on cocaine CPP and locomotor activity is mediated in part by expression of endogenous SGK1 protein and that K127Q may have dominant-negative actions.
Discussion
Blunting VTA SGK1 catalytic activity impairs cocaine CPP in a cell type-specific manner
SGK1 is widely expressed in the brain and its transcription can be induced by virtually all classes of drug of abuse [13,14,15,16, 40]; however, few studies have built on transcriptional findings to characterize protein-level regulation and function. Previous work from our lab identified an upregulation in VTA SGK1 mRNA and catalytic activity following repeated cocaine administration [13]. Though SGK1 catalytic activity has been identified as a modulator of stress- and learning-related behaviors [18,19,20, 22,23,24], its effect on cocaine-elicited behavior remained unknown. Thus, this work supports the relevance of SGK1 catalytic activity in modulating cocaine response. We found that blunting VTA SGK1 catalytic activity (K127Q) impaired cocaine CPP, while wild-type SGK1 overexpression alone had no effect on CPP. This is consistent with data showing VTA expression of a constitutively active SGK1 mutant (S422D) does not further increase cocaine CPP compared to GFP controls [13]. Together, these data suggest that while constraining VTA SGK1 catalytic activity is sufficient to disrupt cocaine CPP, increasing expression or activity are not sufficient to further increase CPP.
Chronic cocaine increases VTA SGK1 catalytic activity; however, the neuronal cell type/s of interest remained unidentified. Due to SGK1 antibody limitations, immunohistochemistry could not be used to identify the population in which SGK1 regulation occurs. Instead, we took a viral-mediated approach, overexpressing a Cre-dependent catalytically inactive SGK1 mutant (K127Q) in either VTA DA or GABA neurons. Blunted activity in VTA DA, but not GABA, neurons impaired cocaine CPP, replicating whole VTA findings and identifying VTA DA neurons as the likely neuronal population of interest. Interestingly, both male and female mice with VTA DA-specific expression showed a robust increase in psychomotor activity during CPP conditioning. This effect on cocaine-elicited locomotor activity was replicated in a locomotor activity assay. While it is possible this increase in cocaine-elicited locomotor activity contributed to the impaired cocaine CPP in DAT-K127Q mice, we do not think elevated locomotor activity is necessary for the impairment, as HSV-K127Q mice had a similar decrease in cocaine CPP without a change in cocaine-elicited locomotor activity. Given projection-specific effects of VTA DA neurons on behavior [41, 42], future studies may seek to interrogate whether decreasing SGK1 catalytic activity in specific subpopulations of VTA DA neurons drives effects on cocaine CPP vs. locomotor activity.
We next sought to determine if responses to non-drug rewards were altered by VTA K127Q expression, using sucrose preference as an example. Neither VTA- nor VTA DA-specific K127Q expression altered sucrose preference in a two-bottle choice task. These data are consistent with our operant sucrose behavior, as blunted VTA DA SGK1 activity did not alter the number of sucrose rewards earned during the food training phase. While these measures do not offer a perfect comparison to our cocaine CPP data given that performance in these assays is dependent on both reinforcement and reward, the combined data suggest that non-drug rewards may not be sufficient to drive SGK1 signaling pathways, or that SGK1’s role may be drug-specific.
Blunting SGK1 catalytic activity in VTA DA neurons does not impair cocaine self-administration
While male mice with VTA DA neuron-specific K127Q expression showed deficits in cocaine CPP, effects were not significant in cocaine self-administration paradigms. Blunted VTA DA SGK1 activity did not alter cocaine intake or motivation to earn cocaine rewards. In addition, while K127Q-expressing mice exhibited decreased cocaine CPP at a moderate but not a low conditioning dose, no differences between groups were seen in dose-dependent responding during cocaine self-administration. Several factors could contribute to these differences. First, mice were taught to respond for sucrose pellets prior to cocaine self-administration, and it is possible that any modest effects on drug acquisition or intake were obscured by this prior training. Second, it is possible that there are more subtle effects on motivation (PR breakpoint) or dose response that would be revealed with a larger sample size. Third, CPP and self-administration paradigms differ greatly in both route of administration and cocaine dose. On top of self-administered vs. investigator administered methods of cocaine delivery, self-administrating commonly leads to greater drug intake for a greater number of sessions compared to CPP. These factors may contribute to divergent VTA SGK1 regulation. Evidence of this idea is supported by mRNA studies. We previously identified a significant increase in VTA sgk1 transcription following 7 days of cocaine i.p. injections with 24 h of withdrawal [13]. However, RNA-sequencing of VTA tissue from mice that self-administered cocaine showed a significant reduction in VTA sgk1 mRNA following 24 h of withdrawal [33]. Comparing these studies with our current findings suggests that sgk1 may be differently regulated in these two administration paradigms. In light of this, SGK1 activity may be downregulated at the time we measured PR and ran the dose-response analysis. If this is true, viral-mediated constraint of catalytic activity would understandably have little effect when SGK1 activity was already low. However, as sgk1 mRNA does not always track directly with protein regulation [13], protein-level analysis is required to confirm this potential interpretation.
SGK1 K127Q may achieve effects via interfering with endogenous SGK1 activity
While viral-mediated overexpression of SGK1 mutants has successfully been used to define a role for SGK1 in regulating drug-related behaviors and learning and memory [13, 18,19,20, 22, 23, 29, 43], it remains unknown if these effects occur through dominant-negative effects or direct action of the mutant SGK1, as endogenous SGK1 is still expressed in these models. DA-SGK1-KO mice do not exhibit decreased cocaine CPP [28], likely due to developmental compensation of SGK1 function via either opportunistic compensation of other AGC kinases that share SGK1 substrates such as AKT [44] or via increased expression of other SGK isoforms such as SGK3 [24]. Our results here may support a viral-mediated disruption of endogenous SGK1 protein function, as K127Q mutant expression in DA-SGK1-KO mice does not replicate deficits in cocaine CPP and psychomotor activity observed when the mutant is expressed in DA neurons with endogenous SGK1 protein expression. This potential dominant-negative action of K127Q supports the idea that acutely decreasing endogenous SGK1 signaling may be beneficial for modulating cocaine CPP. Future studies overexpressing WT SGK1 both alone and in combination with the K127Q mutant in a DA-SGK1-KO model that removes the confound of developmental compensation (i.e., viral-mediated CRISPR-Cas9 approach) are essential for further defining this mechanism. Additionally, improved IHC reagents that allow quantification of SGK1 catalytic activity in individual cells will allow determination of whether K127Q prevents cocaine-induced increases in SGK1 catalytic activity in dopamine neurons, which we were unable to confirm using western blot, likely due to increased SGK1 activity in non-neuronal cells induced by surgical procedures. While K127Q has been shown to prevent stimulated increases in SGK1 catalytic activity in cell culture models [9], it remains unclear whether K127Q physically interacts with endogenous SGK1 to alter function, as dimers have not been reported, or may act as a “sponge” interacting with substrates and preventing access of endogenous SGK1. Given our results that K127Q can influence behavior, these are important mechanistic questions deserving of future studies. Additionally, pharmacological inhibition of endogenous SGK1 is a method worthy of further experimentation. Unfortunately, while SGK1 inhibitors have been developed [45,46,47,48], there are significant limitations to their use. Particularly for commercially available GSK650394, the most widely used compound, these issues include specificity for SGK1 over other SGK isoforms and poor membrane permeability [45, 49]. Once these limitations can be overcome, future work should utilize pharmacological inhibitors in the study of VTA SGK1 function.
In summary, this work demonstrates for the first time that VTA SGK1 is capable of modulating cocaine-elicited behaviors, as blunted catalytic activity was sufficient to disrupt cocaine CPP and alter psychomotor activity. Further, it appears that SGK1 activity within VTA DA neurons is critical for effects on cocaine CPP, though this manipulation did not affect cocaine self-administration. Altogether, this work expands on the limited knowledge of SGK1 function in the central nervous system, critical to understanding the effect of SGK1 catalytic activity on cocaine-elicited behavior.
Funding and disclosure
The authors have no financial interests or conflicts of interest to disclose. This research was supported by NIDA (R01 DA039895, M.S.M.-R.), NIMH (R01 MH111604, AJR) and MAD was supported by an institutional training grant (T32 NS044928, NINDS). These funding agencies did not provide direct input on the design, conduct, analysis, or publication of these studies.
References
Webster M, Goya L, Ge Y, Maiyar A, Firestone G. Characterization of sgk, a novel member of the serine/threonine protein kinase gene family which is transcriptionally induced by glucocorticoids and serum. Mol Cell Biol. 1993;13:2031–40.
Pearce LR, Komander D, Alessi DR. The nuts and bolts of AGC protein kinases. Nat Rev Mol Cell Biol 2010;11:9–22.
Kobayashi T, Cohen P. Activation of serum- and glucocorticoid-regulated protein kinase by agonists that activate phosphatidylinositide 3-kinase is mediated by 3-phosphoinositide-dependent protein kinase-1 (PDK1) and PDK2. Biochem J 1999;339:319–28.
Kobayashi T, Deak M, Morrice N, Cohen P. Characterization of the structure and regulation of two novel isoforms of serum- and glucocorticoid-induced protein kinase. Biochem J 1999;344:189–97.
Park J, Leong MLL, Buse P, Maiyar AC, Firestone GL, Hemmings BA. Serum and glucocorticoid-inducible kinase (SGK) is a target of the PI 3-kinase-stimulated signaling pathway. EMBO J. 1999;18:10.
Garcia-Martinez JM, Alessi DR. mTOR complex 2 (mTORC2) controls hydrophobic motif phosphorylation and activation of serum- and glucocorticoid-induced protein kinase 1 (SGK1). Biochem J. 2008;416:375–85.
Snyder PM, Olson DR, Thomas BC. Serum and glucocorticoid-regulated kinase modulates Nedd4-2-mediated inhibition of the epithelial Na+ channel. J Biol Chem. 2002;277:5–8.
Strutz-Seebohm N, Seebohm G, Shumilina E, Mack AF, Wagner H-J, Lampert A, et al. Glucocorticoid adrenal steroids and glucocorticoid-inducible kinase isoforms in the regulation of GluR6 expression. J Physiol. 2005;565:391–401.
Brunet A, Park J, Tran H, Hu LS, Hemmings BA, Greenberg ME. Protein kinase SGK mediates survival signals by phosphorylating the forkhead transcription factor FKHRL1 (FOXO3a). Mol Cell Biol 2001;21:952–65.
Lang F, Bohmer C, Palmada M, Seebohm G, Strutz-Seebohm N, Vallon V. (Patho)physiological significance of the serum- and glucocorticoid-inducible kinase isoforms. Physiol Rev. 2006;86:1151–78.
Lang F, Strutz-Seebohm N, Seebohm G, Lang UE. Significance of SGK1 in the regulation of neuronal function. J Physiol 2010;588:3349–54.
Wang L, Zhou C, Zhu Q, Luo J, Xu Y, Huang Y, et al. Up-regulation of serum- and glucocorticoid-induced protein kinase 1 in the brain tissue of human and experimental epilepsy. Neurochem Int 2010;57:899–905.
Heller EA, Kaska S, Fallon B, Ferguson D, Kennedy PJ, Neve RL, et al. Morphine and cocaine increase serum- and glucocorticoid-inducible kinase 1 activity in the ventral tegmental area. J Neurochem 2015;132:243–53.
McClung CA, Nestler EJ, Zachariou V. Regulation of gene expression by chronic morphine and morphine withdrawal in the locus ceruleus and ventral tegmental area. J Neurosci 2005;25:6005–15.
Gao P, Limpens JH, Spijker S, Vanderschuren LJ, Voorn P. Stable immediate early gene expression patterns in medial prefrontal cortex and striatum after long-term cocaine self-administration. Addict Biol 2017;22:354–68.
Nichols CD, Sanders-Bush E. A single dose of lysergic acid diethylamide influences gene expression patterns within the mammalian brain. Neuropsychopharmacology 2002;26:634–42.
Cooper SE, Kechner M, Caraballo-Perez D, Kaska S, Robison AJ, Mazei-Robison MS. Comparison of chronic physical and emotional social defeat stress effects on mesocorticolimbic circuit activation and voluntary consumption of morphine. Sci Rep. 2017;7:8445.
Licznerski P, Duric V, Banasr M, Alavian KN, Ota KT, Kang HJ, et al. Decreased SGK1 expression and function contributes to behavioral deficits induced by traumatic stress. PLoS Biol 2015;13:e1002282.
Tsai KJ, Chen SK, Ma YL, Hsu WL, Lee EH. sgk, a primary glucocorticoid-induced gene, facilitates memory consolidation of spatial learning in rats. Proc Natl Acad Sci USA. 2002;99:3990–5.
Lee CT, Tyan SW, Ma YL, Tsai MC, Yang YC, Lee EH. Serum- and glucocorticoid-inducible kinase (SGK) is a target of the MAPK/ERK signaling pathway that mediates memory formation in rats. Eur J Neurosci 2006;23:1311–20.
Frodin M, Antal TL, Dummler BA, Jensen CJ, Deak M, Gammeltoft S, et al. A phosphoserine/threonine-binding pocket in AGC kinases and PDK1 mediates activation by hydrophobic motif phosphorylation. EMBO J. 2002;21:5396–407.
Lee EHY, Hsu WL, Ma YL, Lee PJ, Chao CC. Enrichment enhances the expression of sgk, a glucocorticoid-induced gene, and facilitates spatial learning through glutamate AMPA receptor mediation. Eur J Neurosci. 2003;18:2842–52.
Tyan SW, Tsai MC, Lin CL, Ma YL, Lee EH. Serum- and glucocorticoid-inducible kinase 1 enhances zif268 expression through the mediation of SRF and CREB1 associated with spatial memory formation. J Neurochem 2008;105:820–32.
Yuen EY, Liu W, Karatsoreos IN, Ren Y, Feng J, McEwen BS, et al. Mechanisms for acute stress-induced enhancement of glutamatergic transmission and working memory. Mol Psychiatry. 2011;16:156–70.
Nair-Roberts RG, Chatelain-Badie SD, Benson E, White-Cooper H, Bolam JP, Ungless MA. Stereological estimates of dopaminergic, GABAergic and glutamatergic neurons in the ventral tegmental area, substantia nigra and retrorubral field in the rat. Neuroscience 2008;152:1024–31.
Swanson LW. The projections of the ventral tegmental area and adjacent regions: a combined fluorescent retrograde tracer and immunofluorescence study in the rat. Brain Res Bull 1982;9:321–53.
Fejes-Toth G, Frindt G, Naray-Fejes-Toth A, Palmer LG. Epithelial Na+ channel activation and processing in mice lacking SGK1. Am J Physiol Ren Physiol 2008;294:F1298–305.
Doyle MA, Stark AR, Fejes-Toth G, Naray-Fejes-Toth A, Mazei-Robison MS. Behavioral effects of SGK1 knockout in VTA and dopamine neurons. Sci Rep. 2020;10:14751.
Steffke EE, Kirca D, Mazei-Robison MS, Robison AJ. Serum- and glucocorticoid-inducible kinase 1 activity reduces dendritic spines in dorsal hippocampus. Neurosci Lett 2020;725:134909.
Kaska S, Brunk R, Bali V, Kechner M, Mazei-Robison MS. Deletion of Rictor in catecholaminergic neurons alters locomotor activity and ingestive behavior. Neuropharmacology 2017;117:158–70.
Simmons SC, Wheeler K, Mazei-Robison MS. Determination of circuit-specific morphological adaptations in ventral tegmental area dopamine neurons by chronic morphine. Mol Brain 2019;12:10.
Russo SJ, Jenab S, Fabian SJ, Festa ED, Kemen LM, Quinones-Jenab V. Sex differences in the conditioned rewarding effects of cocaine. Brain Res. 2003;970:214–20.
Walker DM, Cates HM, Loh YE, Purushothaman I, Ramakrishnan A, Cahill KM, et al. Cocaine Self-administration Alters Transcriptome-wide Responses in the Brain’s Reward Circuitry. Biol Psychiatry. 2018;84:867–80.
Ozburn AR, Larson EB, Self DW, McClung CA. Cocaine self-administration behaviors in ClockDelta19 mice. Psychopharmacol (Berl). 2012;223:169–77.
Richardson NR, Roberts DCS. Progressive ratio schedules in drug self-administration studies in rats: a method to evaluate reinforcing efficacy. J Neurosci Methods. 1996;66:1–11.
Arena DT, Covington HE, 3rd, DeBold JF, Miczek KA. Persistent increase of I.V. cocaine self-administration in a subgroup of C57BL/6J male mice after social defeat stress. Psychopharmacology (Berl). 2019;236:2027–37.
Murray JT, Campbell DG, Morrice N, Auld GC, Shpiro N, Marquez R, et al. Exploitation of KESTREL to identify NDRG family members as physiological substrates for SGK1 and GSK3. Biochem J. 2004;384:477–88.
Cahill ME, Walker DM, Gancarz AM, Wang ZJ, Lardner CK, Bagot RC, et al. The dendritic spine morphogenic effects of repeated cocaine use occur through the regulation of serum response factor signaling. Mol Psychiatry 2018;23:1474–86.
Russo SJ, Wilkinson MB, Mazei-Robison MS, Dietz DM, Maze I, Krishnan V, et al. Nuclear factor kappa B signaling regulates neuronal morphology and cocaine reward. J Neurosci 2009;29:3529–37.
Piechota M, Korostynski M, Solecki W, Gieryk A, Slezak M, Bilecki W, et al. The dissection of transcriptional modules regulated by various drugs of abuse in the mouse striatum. Genome Biol 2010;11:R48.
Collins AL, Saunders BT. Heterogeneity in striatal dopamine circuits: form and function in dynamic reward seeking. J Neurosci Res 2020;98:1046–69.
Morales M, Margolis EB. Ventral tegmental area: cellular heterogeneity, connectivity and behaviour. Nat Rev Neurosci 2017;18:73–85.
Lee CT, Ma YL, Lee EH. Serum- and glucocorticoid-inducible kinase1 enhances contextual fear memory formation through down-regulation of the expression of Hes5. J Neurochem 2007;100:1531–42.
Sommer EM, Dry H, Cross D, Guichard S, Davies BR, Alessi DR. Elevated SGK1 predicts resistance of breast cancer cells to Akt inhibitors. Biochem J. 2013;452:499–508.
Sherk AB, Frigo DE, Schnackenberg CG, Bray JD, Laping NJ, Trizna W, et al. Development of a small-molecule serum- and glucocorticoid-regulated kinase-1 antagonist and its evaluation as a prostate cancer therapeutic. Cancer Res 2008;68:7475–83.
D’Antona L, Amato R, Talarico C, Ortuso F, Menniti M, Dattilo V, et al. SI113, a specific inhibitor of the Sgk1 kinase activity that counteracts cancer cell proliferation. Cell Physiol Biochem 2015;35:2006–18.
Halland N, Schmidt F, Weiss T, Saas J, Li Z, Czech J. et al. Discovery of N-[4-(1H-Pyrazolo[3,4-b]pyrazin-6-yl)-phenyl]-sulfonamides as highly active and selective SGK1 inhibitors. ACS Med Chem Lett. 2015;6:73–8.
Ackermann TF, Boini KM, Beier N, Scholz W, Fuchß T, Lang F. EMD638683, a novel SGK inhibitor with antihypertensive potency. Cell Physiol Biochem 2011;28:137–46.
Di Cristofano A. SGK1: the dark side of PI3K signaling. Curr Top Developmental Biol. 2017;123:49–71.
Acknowledgements
We would like to thank Ken Moon for his significant assistance with mouse genotyping and breeding and Drs. Sarah Simmons and Sophia Kaska for their contribution in training on the molecular and behavioral techniques used to obtain data for this manuscript. We would also like to thank Cristina Rivera Quiles and Samantha Caico for assistance with viral targeting and validation.
Author information
Authors and Affiliations
Contributions
MAD: conceptualization, investigation, formal analysis, visualization, writing–original draft; VB: investigation, validation, writing–review and editing; ALE: investigation; ARS: investigation, validation; BF: validation; RLN: resources; AJR; writing–review and editing; MSM-R: conceptualization, methodology, writing–review and editing, supervision, project administration, funding acquisition.
Corresponding author
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Doyle, M.A., Bali, V., Eagle, A.L. et al. Serum- and glucocorticoid-inducible kinase 1 activity in ventral tegmental area dopamine neurons regulates cocaine conditioned place preference but not cocaine self-administration. Neuropsychopharmacol. 46, 1574–1583 (2021). https://doi.org/10.1038/s41386-021-01032-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41386-021-01032-3