Abstract
Objective
To determine whether age and neuropeptide Y (NPY) were involved in the skeletal response to extended periods of diet-induced obesity.
Methods
Male wild-type (WT) and NPY null (NPYKO) mice were fed a mild (23% fat) high-fat diet for 10 weeks from 6 or 16 weeks of age. Metabolism and bone density were assessed during feeding. Skeletal changes were assessed by microCT and histomorphometry.
Results
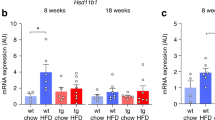
High-fat feeding in 6-week-old WT mice led to significantly increased body weight, adiposity and serum leptin levels, accompanied with markedly suppressed cortical bone accrual. NPYKO mice were less susceptible to fat accrual but, importantly, displayed a complete lack of suppression of bone accrual or cortical bone loss. In contrast, when skeletally mature (16 week old) mice underwent 10 weeks of fat feeding, the metabolic response to HFD was similar to younger mice, however bone mass was not affected in either WT or NPYKO. Thus, growing mice are particularly susceptible to the detrimental effects of HFD on bone mass, through suppression of bone accrual involving NPY signalling.
Conclusion
This study provides new insights into the relationship between the opposing processes of a positive weight/bone relationship and the negative ‘metabolic’ effect of obesity on bone mass. This negative effect is particularly active in growing skeletons, which have heightened sensitivity to changes in obesity. In addition, NPY is identified as a fundamental driver of this negative ‘metabolic’ pathway to bone.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Australian Bureau of Statistics. Australian Health Survey. Australian Bureau of Statistics: Canberra, Australia; 2011–2012.
Ogden CL, Carroll MD, Lawman HG, Fryar CD, Kruszon-Moran D, Kit BK, et al. Trends in obesity prevalence among children and adolescents in the United States, 1988-1994 through 2013-2014. JAMA. 2016;315:2292–9.
Furukawa S, Fujita T, Shimabukuro M, Iwaki M, Yamada Y, Nakajima Y, et al. Increased oxidative stress in obesity and its impact on metabolic syndrome. J Clin Invest. 2004;114:1752–61.
Gregor MF, Hotamisligil GS. Inflammatory mechanisms in obesity. Annu Rev Immunol. 2011;29:415–45.
Bacha F, Gidding SS. Cardiac abnormalities in youth with obesity and type 2 diabetes. Curr Diab Rep. 2016;16:62.
Felson DT, Zhang Y, Hannan MT, Anderson JJ. Effects of weight and body mass index on bone mineral density in men and women: the Framingham study. J Bone Mineral Res. 1993;8:567–73.
Ho-Pham LT, Nguyen UD, Nguyen TV. Association between lean mass, fat mass, and bone mineral density: a meta-analysis. J Clin Endocrinol Metab. 2014;99:30–8.
Compston J. Obesity and fractures in postmenopausal women. Curr Opin Rheumatol. 2015;27:414–9.
Dimitri P, Jacques RM, Paggiosi M, King D, Walsh J, Taylor ZA, et al. Leptin may play a role in bone microstructural alterations in obese children. J Clin Endocrinol Metab. 2015;100:594–602.
Mosca LN, Goldberg TB, da Silva VN, da Silva CC, Kurokawa CS, Bisi Rizzo AC, et al. Excess body fat negatively affects bone mass in adolescents. Nutrition. 2014;30:847–52.
Klein-Nulend J, Bacabac RG, Bakker AD. Mechanical loading and how it affects bone cells: the role of the osteocyte cytoskeleton in maintaining our skeleton. Eur Cell Mater. 2012;24:278–91.
Cao JJ, Gregoire BR, Gao H. High-fat diet decreases cancellous bone mass but has no effect on cortical bone mass in the tibia in mice. Bone. 2009;44:1097–104.
Cao JJ, Sun L, Gao H. Diet-induced obesity alters bone remodeling leading to decreased femoral trabecular bone mass in mice. Ann N Y Acad Sci. 2010;1192:292–7.
Fehrendt H, Linn T, Hartmann S, Szalay G, Heiss C, Schnettler R, et al. Negative influence of a long-term high-fat diet on murine bone architecture. Int J Endocrinol. 2014;2014:318924.
Inzana JA, Kung M, Shu L, Hamada D, Xing LP, Zuscik MJ, et al. Immature mice are more susceptible to the detrimental effects of high fat diet on cancellous bone in the distal femur. Bone. 2013;57:174–83.
Lecka-Czernik B, Stechschulte LA, Czernik PJ, Dowling AR. High bone mass in adult mice with diet-induced obesity results from a combination of initial increase in bone mass followed by attenuation in bone formation; implications for high bone mass and decreased bone quality in obesity. Mol Cell Endocrinol. 2015;410:35–41.
Ionova-Martin SS, Do SH, Barth HD, Szadkowska M, Porter AE, Ager JW 3rd, et al. Reduced size-independent mechanical properties of cortical bone in high-fat diet-induced obesity. Bone. 2010;46:217–25.
Fujita Y, Watanabe K, Maki K. Serum leptin levels negatively correlate with trabecular bone mineral density in high-fat diet-induced obesity mice. J Musculoskelet Neuron Interact. 2012;12:84–94.
Scheller EL, Khoury B, Moller KL, Wee NK, Khandaker S, Kozloff KM, et al. Changes in skeletal integrity and marrow adiposity during high-fat diet and after weight loss. Front Endocrinol. 2016;7:102.
Wee NK, Herzog H, Baldock PA. Diet-induced obesity alters skeletal microarchitecture and the endocrine activity of bone. In: Watson RR, Mahadevan D, editors. Nutrition and diet in therapy of bone diseases. Wageningen Academic; 2016. pp 375–94.
Shi YC, Lau J, Lin Z, Zhang H, Zhai L, Sperk G, et al. Arcuate NPY controls sympathetic output and BAT function via a relay of tyrosine hydroxylase neurons in the PVN. Cell Metab. 2013;17:236–48.
Lee SJ, Verma S, Simonds SE, Kirigiti MA, Kievit P, Lindsley SR, et al. Leptin stimulates neuropeptide Y and cocaine amphetamine-regulated transcript coexpressing neuronal activity in the dorsomedial hypothalamus in diet-induced obese mice. J Neurosci. 2013;33:15306–17.
Baldock PA, Sainsbury A, Allison S, Lin E-JD, Couzens M, Boey D. et al. Hypothalamic control of bone formation: distinct actions of letin and Y2 receptor pathways. J Bone Miner Res. 2005;20:1851–7.
Wong IP, Nguyen AD, Khor EC, Enriquez RF, Eisman JA, Sainsbury A, et al. Neuropeptide Y is a critical modulator of leptin’s regulation of cortical bone. J Bone Miner Res. 2013;28:886–98.
Karl T, Duffy L, Herzog H. Behavioural profile of a new mouse model for NPY deficiency. Eur J Neurosci. 2008;28:173–80.
Baldock PA, Allison S, McDonald MM, Sainsbury A, Enriquez RF, Little DG, et al. Hypothalamic regulation of cortical bone mass: opposing activity of Y2 receptor and leptin pathways. J Bone Miner Res. 2006;21:1600–7.
Zhang L, Lee IC, Enriquez RF, Lau J, Vahatalo LH, Baldock PA, et al. Stress- and diet-induced fat gain is controlled by NPY in catecholaminergic neurons. Mol Metab. 2014;3:581–91.
Baldock PA, Lee NJ, Driessler F, Lin S, Allison S, Stehrer B, et al. Neuropeptide Y knockout mice reveal a central role of NPY in the coordination of bone mass to body weight. PloS ONE. 2009;4:e8415.
Ducy P, Amling M, Takeda S, Priemel M, Schilling AF, Beil FT, et al. Leptin inhibits bone formation through a hypothalamic relay: a central control of bone mass. Cell. 2000;100:197–207.
Takeda S, Elefteriou F, Levasseur R, Liu X, Zhao L, Parker KL, et al. Leptin regulates bone formation via the sympathetic nervous system. Cell. 2002;111:305–17.
Elefteriou F, Takeda S, Ebihara K, Magre J, Patano N, Ae Kim C, et al. Serum leptin level is a regulator of bone mass. Proc Natl Acad Sci USA. 2004;101:3258–63.
Enriori PJ, Sinnayah P, Simonds SE, Garcia Rudaz C, Cowley MA. Leptin action in the dorsomedial hypothalamus increases sympathetic tone to brown adipose tissue in spite of systemic leptin resistance. J Neurosci. 2011;31:12189–97.
Igwe JC, Jiang X, Paic F, Ma L, Adams DJ, Baldock PA, et al. Neuropeptide Y is expressed by osteocytes and can inhibit osteoblastic activity. J Cell Biochem. 2009;108:621–30.
Zhang L, Macia L, Turner N, Enriquez RF, Riepler SJ, Nguyen AD, et al. Peripheral neuropeptide Y Y1 receptors regulate lipid oxidation and fat accretion. Int J Obes (Lond). 2010;34:357–73.
Gautam J, Choudhary D, Khedgikar V, Kushwaha P, Singh RS, Singh D, et al. Micro-architectural changes in cancellous bone differ in female and male C57BL/6 mice with high-fat diet-induced low bone mineral density. Br J Nutr. 2014;111:1811–21.
Kessler J, Koebnick C, Smith N, Adams A. Childhood obesity is associated with increased risk of most lower extremity fractures. Clin Orthop Relat Res. 2013;471:1199–207.
Loh K, Herzog H, Shi YC. Regulation of energy homeostasis by the NPY system. Trends Endocrinol Metab. 2015;26:125–35.
Baldock PA, Sainsbury A, Couzens M, Enriquez RF, Thomas GP, Gardiner EM, et al. Hypothalamic Y2 receptors regulate bone formation. J Clin Invest. 2002;109:915–21.
Wee NKY, Kulkarni RN, Horsnell H, Baldock PA. The brain in bone and fuel metabolism. Bone. 2016;82:56–63.
Khor EC, Wee NK, Baldock PA. Influence of hormonal appetite and energy regulators on bone. Curr Osteoporos Rep. 2013;11:194–202.
Baldock PA, Allison SJ, Lundberg P, Lee NJ, Slack K, Lin EJ, et al. Novel role of Y1 receptors in the coordinated regulation of bone and energy homeostasis. J Biol Chem. 2007;282:19092–102.
Hamrick MW, Della-Fera MA, Choi YH, Pennington C, Hartzell D, Baile CA. Leptin treatment induces loss of bone marrow adipocytes and increases bone formation in leptin-deficient ob/ob mice. J Bone Miner Res. 2005;20:994–1001.
Matic I, Matthews BG, Kizivat T, Igwe JC, Marijanovic I, Ruohonen ST, et al. Bone-specific overexpression of NPY modulates osteogenesis. J Musculoskelet Neuron Interact. 2012;12:209–18.
Compston J. Obesity and bone. Curr Osteoporos Rep. 2013;11:30–5.
Greco EA, Francomano D, Fornari R, Marocco C, Lubrano C, Papa V, et al. Negative association between trunk fat, insulin resistance and skeleton in obese women. World J Diabetes. 2013;4:31–9.
Johansson H, Kanis JA, Odén A, McCloskey E, Chapurlat RD, Christiansen C, et al. A meta-analysis of the association of fracture risk and body mass index in women. J Bone Miner Res. 2014;29:223–33.
Goulding A, Grant AM, Williams SM. Bone and body composition of children and adolescents with repeated forearm fractures. J Bone Miner Res. 2005;20:2090–6.
Macri EV, Gonzales Chaves MM, Rodriguez PN, Mandalunis P, Zeni S, Lifshitz F, et al. High-fat diets affect energy and bone metabolism in growing rats. Eur J Nutr. 2012;51:399–406.
Bonnet N, Somm E, Rosen CJ. Diet and gene interactions influence the skeletal response to polyunsaturated fatty acids. Bone. 2014;68:100–7.
Lau BY, Fajardo VA, McMeekin L, Sacco SM, Ward WE, Roy BD, et al. Influence of high-fat diet from differential dietary sources on bone mineral density, bone strength, and bone fatty acid composition in rats. Appl Physiol Nutr Metab. 2010;35:598–606.
Dong XL, Li CM, Cao SS, Zhou LP, Wong MS. A high-saturated-fat, high-sucrose diet aggravates bone loss in ovariectomized female rats. J Nutr. 2016;146:1172–9.
Tian L, Yu X, Fat, sugar, and bone health: a complex relationship. Nutrients 2017; 9: pii: E506. https://doi.org/10.3390/nu9050506.
Lai M, Chandrasekera PC, Barnard ND. You are what you eat, or are you? The challenges of translating high-fat-fed rodents to human obesity and diabetes. Nutr Diabetes. 2014;4:e135.
Ortinau LC, Linden MA, Dirkes R, Rector RS, Hinton PS. Obesity and type 2 diabetes, not a diet high in fat, sucrose, and cholesterol, negatively impacts bone outcomes in the hyperphagic Otsuka Long Evans Tokushima Fatty rat. Bone. 2017;105:200–11.
Munzberg H, Flier JS, Bjorbaek C. Region-specific leptin resistance within the hypothalamus of diet-induced obese mice. Endocrinology. 2004;145:4880–9.
Acknowledgements
NW was supported by an Australian Postgraduate Award (APA) administered by UNSW Australia. We would also like to thank the Biological Testing Facility at the Garvan Institute for assistance with the animal work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Wee, N.K.Y., Enriquez, R.F., Nguyen, A.D. et al. Diet-induced obesity suppresses cortical bone accrual by a neuropeptide Y-dependent mechanism. Int J Obes 42, 1925–1938 (2018). https://doi.org/10.1038/s41366-018-0028-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41366-018-0028-y
This article is cited by
-
Energy Balance and Bone Health: a Nutrient Availability Perspective
Current Osteoporosis Reports (2023)
-
Endocrine role of bone in the regulation of energy metabolism
Bone Research (2021)
-
Neuropeptide Y Regulation of Energy Partitioning and Bone Mass During Cold Exposure
Calcified Tissue International (2020)