Abstract
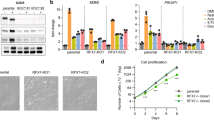
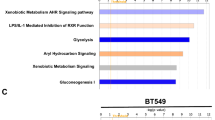
We recently reported that ZBTB7A is a bona fide transcription repressor of key glycolytic genes and its downregulation in human cancer contributes to tumor metabolism. As reduced expression of ZBTB7A is found only in a subset of human cancers, we explored alternative mechanisms of its inactivation by mining human cancer genome databases. We discovered recurrent somatic mutations of ZBTB7A in multiple types of human cancers with a marked enrichment of mutations within the zinc finger domain. Functional characterization of the mutants demonstrated that mutations within the zinc finger region of ZBTB7A invariably resulted in loss of function. As a consequence, the glycolytic genes were markedly upregulated in cancer cells harboring ZBTB7A zinc finger mutation, leading to increased glycolysis and proliferation. Our study uncovers the loss-of-function mutation in ZBTB7A as a novel mechanism causing elevated glycolysis in human cancer, which carries important therapeutic implication.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Maeda T, Hobbs RM, Merghoub T, Guernah I, Zelent A, Cordon-Cardo C et al. Role of the proto-oncogene Pokemon in cellular transformation and ARF repression. Nature 2005; 433: 278–285.
Pessler F, Pendergrast PS, Hernandez N . Purification and characterization of FBI-1, a cellular factor that binds to the human immunodeficiency virus type 1 inducer of short transcripts. Mol Cell Biol 1997; 17: 3786–3798.
Liu CJ, Prazak L, Fajardo M, Yu S, Tyagi N, Di Cesare PE . Leukemia/lymphoma-related factor, a POZ domain-containing transcriptional repressor, interacts with histone deacetylase-1 and inhibits cartilage oligomeric matrix protein gene expression and chondrogenesis. J Biol Chem 2004; 279: 47081–47091.
Kukita A, Kukita T, Ouchida M, Maeda H, Yatsuki H, Kohashi O . Osteoclast-derived zinc finger (OCZF) protein with POZ domain, a possible transcriptional repressor, is involved in osteoclastogenesis. Blood 1999; 94: 1987–1997.
Stogios PJ, Downs GS, Jauhal JJ, Nandra SK, Prive GG . Sequence and structural analysis of BTB domain proteins. Genome Biol 2005; 6: R82.
Lee SU, Maeda T . POK/ZBTB proteins: an emerging family of proteins that regulate lymphoid development and function. Immunol Rev 2012; 247: 107–119.
Kelly KF, Daniel JM . POZ for effect—POZ-ZF transcription factors in cancer and development. Trends Cell Biol 2006; 16: 578–587.
Costoya JA . Functional analysis of the role of POK transcriptional repressors. Brief Funct Genomic Proteomic 2007; 6: 8–18.
Lo Coco F, Ye BH, Lista F, Corradini P, Offit K, Knowles DM et al. Rearrangements of the BCL6 gene in diffuse large cell non-Hodgkin's lymphoma. Blood 1994; 83: 1757–1759.
Chen Z, Brand NJ, Chen A, Chen SJ, Tong JH, Wang ZY et al. Fusion between a novel Krüppel-like zinc finger gene and the retinoic acid receptor-alpha locus due to a variant t(11;17) translocation associated with acute promyelocytic leukaemia. EMBO J 1993; 12: 1161–1167.
Maeda T, Merghoub T, Hobbs RM, Dong L, Maeda M, Zakrzewski J et al. Regulation of B versus T lymphoid lineage fate decision by the proto-oncogene LRF. Science 2007; 316: 860–866.
Maeda T, Ito K, Merghoub T, Poliseno L, Hobbs RM, Wang G et al. LRF is an essential downstream target of GATA1 in erythroid development and regulates BIM-dependent apoptosis. Dev Cell 2009; 17: 527–540.
Sakurai N, Maeda M, Lee SU, Ishikawa Y, Li M, Williams JC et al. The LRF transcription factor regulates mature B cell development and the germinal center response in mice. J Clin Invest 2011; 121: 2583–2598.
Beroukhim R, Mermel CH, Porter D, Wei G, Raychaudhuri S, Donovan J et al. The landscape of somatic copy-number alteration across human cancers. Nature 2010; 463: 899–905.
Zack TI, Schumacher SE, Carter SL, Cherniack AD, Saksena G, Tabak B et al. Pan-cancer patterns of somatic copy number alteration. Nat Genet 2013; 45: 1134–1140.
Wang G, Lunardi A, Zhang J, Chen Z, Ala U, Webster KA et al. Zbtb7a suppresses prostate cancer through repression of a Sox9-dependent pathway for cellular senescence bypass and tumor invasion. Nat Genet 2013; 45: 739–746.
Liu XS, Haines JE, Mehanna EK, Genet MD, Ben-Sahra I, Asara JM et al. ZBTB7A acts as a tumor suppressor through the transcriptional repression of glycolysis. Genes Dev 2014; 28: 1917–1928.
Lawrence MS, Stojanov P, Mermel CH, Robinson JT, Garraway LA, Golub TR et al. Discovery and saturation analysis of cancer genes across 21 tumour types. Nature 2014; 505: 495–501.
International Cancer Genome Consortium. International network of cancer genome projects. Nature 2010; 464: 993–998.
Cancer Genome Atlas Research Network. The Cancer Genome Atlas Pan-Cancer analysis project. Nat Genet 2013; 45: 1113–1120.
Cerami E, Gao J, Dogrusoz U, Gross BE, Sumer SO, Aksoy BA et al. The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov 2012; 2: 401–404.
Forbes SA, Bindal N, Bamford S, Cole C, Kok CY, Beare D et al. COSMIC: mining complete cancer genomes in the Catalogue of Somatic Mutations in Cancer. Nucleic Acids Res 2011; 39: D945–D950.
Carnero A, Hudson JD, Price CM, Beach DH . p16INK4A and p19ARF act in overlapping pathways in cellular immortalization. Nat Cell Biol 2000; 2: 148–155.
Nelson JD, Denisenko O, Bomsztyk K . Protocol for the fast chromatin immunoprecipitation (ChIP) method. Nat Protoc 2006; 1: 179–185.
Acknowledgements
We are grateful to current and former members of the Yuan lab for experimental support, advice and helpful discussions. This work was supported in part by the Morningside Foundation and grants from NIH/NCI (R01CA085679, RO1CA167814 and RO1CA125144).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Liu, XS., Liu, Z., Gerarduzzi, C. et al. Somatic human ZBTB7A zinc finger mutations promote cancer progression. Oncogene 35, 3071–3078 (2016). https://doi.org/10.1038/onc.2015.371
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2015.371
This article is cited by
-
An order independent algorithm for inferring gene regulatory network using quantile value for conditional independence tests
Scientific Reports (2021)
-
AML1/ETO and its function as a regulator of gene transcription via epigenetic mechanisms
Oncogene (2021)
-
ZBTB7A, a potential biomarker for prognosis and immune infiltrates, inhibits progression of endometrial cancer based on bioinformatics analysis and experiments
Cancer Cell International (2020)
-
De novo ZBTB7A variant in a patient with macrocephaly, intellectual disability, and sleep apnea: implications for the phenotypic development in 19p13.3 microdeletions
Journal of Human Genetics (2020)
-
ZBTB7A prevents RUNX1-RUNX1T1-dependent clonal expansion of human hematopoietic stem and progenitor cells
Oncogene (2020)