Abstract
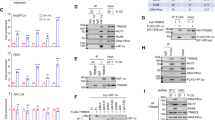
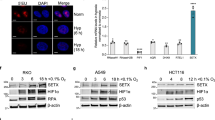
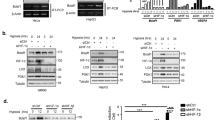
Tumor cells adaptation to severe oxygen deprivation (hypoxia) plays a major role in tumor progression. The transcription factor HIF-1 (hypoxia-inducible factor 1), whose α-subunit is stabilized under hypoxic conditions is a key component of this process. Recent studies showed that two members of the phosphoinositide 3-kinase-related kinases (PIKKs) family, ATM (ataxia telangiectasia mutated) and DNA-PK (DNA-dependent protein kinase), regulate the hypoxic-dependent accumulation of HIF-1. These proteins initiate cellular stress responses when DNA damage occurs. In addition, it has been demonstrated that extreme hypoxia induces a replicative stress resulting in regions of single-stranded DNA at stalled replication forks and the activation of ATR (ataxia telangiectasia and Rad3 related protein), another member of the PIKKs family. Here, we show that even less severe hypoxia (0.1% O2) also induces activation of ATR through replicative stress. Importantly, in using either transiently silenced ATR cells, cells expressing an inactive form of ATR or cells exposed to an ATR inhibitor (CGK733), we demonstrate that hypoxic ATR activation positively regulates the key transcription factor HIF-1 independently of the checkpoint kinase Chk1. We show that ATR kinase activity regulates HIF-1α at the translational level and we find that the elements necessary for the regulation of HIF-1α translation are located within the coding region of HIF-1α mRNA. Finally, by using three independent cellular models, we clearly show that the loss of ATR expression and/or kinase activity results in the decrease of HIF-1 DNA binding under hypoxia and consequently affects protein expression levels of two HIF-1 target genes, GLUT-1 and CAIX. Taken together, our data show a new function for ATR in cellular adaptation to hypoxia through regulation of HIF-1α translation. Our work offers new prospect for cancer therapy using ATR inhibitors with the potential to decrease cellular adaptation in hypoxic tumors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Yee Koh M, Spivak-Kroizman TR, Powis G . HIF-1 regulation: not so easy come, easy go. Trends Biochem Sci 2008; 33: 526–534.
Majmundar AJ, Wong WJ, Simon MC . Hypoxia-inducible factors and the response to hypoxic stress. Mol Cell 2010; 40: 294–309.
Monti E, Gariboldi MB . HIF-1 as a target for cancer chemotherapy, chemosensitization and chemoprevention. Curr Mol Pharmacol 2011; 4: 62–77.
Brahimi-Horn MC, Pouyssegur J . HIF at a glance. J Cell Sci 2009; 122: 1055–1057.
Semenza GL . Evaluation of HIF-1 inhibitors as anticancer agents. Drug Discov Today 2007; 12: 853–859.
Brahimi-Horn MC, Pouyssegur J . Harnessing the hypoxia-inducible factor in cancer and ischemic disease. Biochem Pharmacol 2007; 73: 450–457.
Onnis B, Rapisarda A, Melillo G . Development of HIF-1 inhibitors for cancer therapy. J Cell Mol Med 2009; 13: 2780–2786.
Bouquet F, Ousset M, Biard D, Fallone F, Dauvillier S, Frit P et al. A DNA-dependent stress response involving DNA-PK occurs in hypoxic cells and contributes to cellular adaptation to hypoxia. J Cell Sci 2011; 124: 1943–1951.
Cam H, Easton JB, High A, Houghton PJ . mTORC1 signaling under hypoxic conditions is controlled by ATM-dependent phosphorylation of HIF-1alpha. Mol Cell 2010; 40: 509–520.
Abraham RT . mTOR as a positive regulator of tumor cell responses to hypoxia. Curr Top Microbiol Immunol 2004; 279: 299–319.
Gibson SL, Bindra RS, Glazer PM . Hypoxia-induced phosphorylation of Chk2 in an ataxia telangiectasia mutated-dependent manner. Cancer Res 2005; 65: 10734–10741.
Bencokova Z, Kaufmann MR, Pires IM, Lecane PS, Giaccia AJ, Hammond EM . ATM activation and signaling under hypoxic conditions. Mol Cell Biol 2009; 29: 526–537.
Czornak K, Chughtai S, Chrzanowska KH . Mystery of DNA repair: the role of the MRN complex and ATM kinase in DNA damage repair. J Appl Genet 2008; 49: 383–396.
Lopez-Contreras AJ, Fernandez-Capetillo O . The ATR barrier to replication-born DNA damage. DNA Repair (Amst) 2010; 9: 1249–1255.
Zou L, Elledge SJ . Sensing DNA damage through ATRIP recognition of RPA-ssDNA complexes. Science 2003; 300: 1542–1548.
Adams KE, Medhurst AL, Dart DA, Lakin ND . Recruitment of ATR to sites of ionising radiation-induced DNA damage requires ATM and components of the MRN protein complex. Oncogene 2006; 25: 3894–3904.
Cuadrado M, Martinez-Pastor B, Murga M, Toledo LI, Gutierrez-Martinez P, Lopez E et al. ATM regulates ATR chromatin loading in response to DNA double-strand breaks. J Exp Med 2006; 203: 297–303.
Jazayeri A, Falck J, Lukas C, Bartek J, Smith GC, Lukas J et al. ATM- and cell cycle-dependent regulation of ATR in response to DNA double-strand breaks. Nat Cell Biol 2006; 8: 37–45.
Myers JS, Cortez D . Rapid activation of ATR by ionizing radiation requires ATM and Mre11. J Biol Chem 2006; 281: 9346–9350.
Hammond EM, Dorie MJ, Giaccia AJ . ATR/ATM targets are phosphorylated by ATR in response to hypoxia and ATM in response to reoxygenation. J Biol Chem 2003; 278: 12207–12213.
Hammond EM, Green SL, Giaccia AJ . Comparison of hypoxia-induced replication arrest with hydroxyurea and aphidicolin-induced arrest. Mutat Res 2003; 532: 205–213.
Hammond EM, Dorie MJ, Giaccia AJ . Inhibition of ATR leads to increased sensitivity to hypoxia/reoxygenation. Cancer Res 2004; 64: 6556–6562.
Paulsen RD, Cimprich KA . The ATR pathway: fine-tuning the fork. DNA Repair (Amst) 2007; 6: 953–966.
Crescenzi E, Palumbo G, de Boer J, Brady HJ . Ataxia telangiectasia mutated and p21CIP1 modulate cell survival of drug-induced senescent tumor cells: implications for chemotherapy. Clin Cancer Res 2008; 14: 1877–1887.
Pereg Y, Shkedy D, de Graaf P, Meulmeester E, Edelson-Averbukh M, Salek M et al. Phosphorylation of Hdmx mediates its Hdm2- and ATM-dependent degradation in response to DNA damage. Proc Natl Acad Sci USA 2005; 102: 5056–5061.
Aberle H, Bauer A, Stappert J, Kispert A, Kemler R . beta-catenin is a target for the ubiquitin-proteasome pathway. EMBO J 1997; 16: 3797–3804.
Lovejoy CA, Cortez D . Common mechanisms of PIKK regulation. DNA Repair (Amst) 2009; 8: 1004–1008.
Veuger SJ, Curtin NJ, Richardson CJ, Smith GC, Durkacz BW . Radiosensitization and DNA repair inhibition by the combined use of novel inhibitors of DNA-dependent protein kinase and poly(ADP-ribose) polymerase-1. Cancer Res 2003; 63: 6008–6015.
Willmore E, de Caux S, Sunter NJ, Tilby MJ, Jackson GH, Austin CA et al. A novel DNA-dependent protein kinase inhibitor, NU7026, potentiates the cytotoxicity of topoisomerase II poisons used in the treatment of leukemia. Blood 2004; 103: 4659–4665.
Wan C, Kulkarni A, Wang YH . ATR preferentially interacts with common fragile site FRA3B and the binding requires its kinase activity in response to aphidicolin treatment. Mutat Res 2010; 686: 39–46.
Laughner E, Taghavi P, Chiles K, Mahon PC, Semenza GL . HER2 (neu) signaling increases the rate of hypoxia-inducible factor 1alpha (HIF-1alpha) synthesis: novel mechanism for HIF-1-mediated vascular endothelial growth factor expression. Mol Cell Biol 2001; 21: 3995–4004.
Galban S, Kuwano Y, Pullmann R, Martindale JL, Kim HH, Lal A et al. RNA-binding proteins HuR and PTB promote the translation of hypoxia-inducible factor 1alpha. Mol Cell Biol 2008; 28: 93–107.
Lou JJ, Chua YL, Chew EH, Gao J, Bushell M, Hagen T . Inhibition of hypoxia-inducible factor-1alpha (HIF-1alpha) protein synthesis by DNA damage inducing agents. PloS one 2010; 5: e10522.
Zhong H, Chiles K, Feldser D, Laughner E, Hanrahan C, Georgescu MM et al. Modulation of hypoxia-inducible factor 1alpha expression by the epidermal growth factor/phosphatidylinositol 3-kinase/PTEN/AKT/FRAP pathway in human prostate cancer cells: implications for tumor angiogenesis and therapeutics. Cancer Res 2000; 60: 1541–1545.
Hudson CC, Liu M, Chiang GG, Otterness DM, Loomis DC, Kaper F et al. Regulation of hypoxia-inducible factor 1alpha expression and function by the mammalian target of rapamycin. Mol Cell Biol 2002; 22: 7004–7014.
Gingras AC, Gygi SP, Raught B, Polakiewicz RD, Abraham RT, Hoekstra MF et al. Regulation of 4E-BP1 phosphorylation: a novel two-step mechanism. Genes Dev 1999; 13: 1422–1437.
Chen C, Pore N, Behrooz A, Ismail-Beigi F, Maity A . Regulation of glut1 mRNA by hypoxia-inducible factor-1. Interaction between H-ras and hypoxia. J Biol Chem 2001; 276: 9519–9525.
Lang KJ, Kappel A, Goodall GJ . Hypoxia-inducible factor-1alpha mRNA contains an internal ribosome entry site that allows efficient translation during normoxia and hypoxia. Mol Biol Cell 2002; 13: 1792–1801.
Lei Z, Li B, Yang Z, Fang H, Zhang GM, Feng ZH et al. Regulation of HIF-1alpha and VEGF by miR-20b tunes tumor cells to adapt to the alteration of oxygen concentration. PloS One 2009; 4: e7629.
Airley RE, Loncaster J, Raleigh JA, Harris AL, Davidson SE, Hunter RD et al. GLUT-1 and CAIX as intrinsic markers of hypoxia in carcinoma of the cervix: relationship to pimonidazole binding. Int J Cancer 2003; 104: 85–91.
Semenza GL . Targeting HIF-1 for cancer therapy. Nat Rev Cancer 2003; 3: 721–732.
Ousset M, Bouquet F, Fallone F, Biard D, Dray C, Valet P et al. Loss of ATM positively regulates the expression of hypoxia inducible factor 1 (HIF-1) through oxidative stress: Role in the physiopathology of the disease. Cell Cycle 2010; 9: 2814–2822.
Kang MJ, Jung SM, Kim MJ, Bae JH, Kim HB, Kim JY et al. DNA-dependent protein kinase is involved in heat shock protein-mediated accumulation of hypoxia-inducible factor-1alpha in hypoxic preconditioned HepG2 cells. FEBS J 2008; 275: 5969–5981.
Economopoulou M, Langer HF, Celeste A, Orlova VV, Choi EY, Ma M et al. Histone H2AX is integral to hypoxia-driven neovascularization. Nat Med 2009; 15: 553–558.
Martin L, Rainey M, Santocanale C, Gardner LB . Hypoxic activation of ATR and the suppression of the initiation of DNA replication through cdc6 degradation. Oncogene 2011; 31: 4076–4084.
Rapisarda A, Melillo G . UVC inhibits HIF-1alpha protein translation by a DNA damage- and topoisomerase I-independent pathway. Oncogene 2007; 26: 6875–6884.
Semenza GL . Hypoxia-inducible factors in physiology and medicine. Cell 2012; 148: 399–408.
Zundel W, Schindler C, Haas-Kogan D, Koong A, Kaper F, Chen E et al. Loss of PTEN facilitates HIF-1-mediated gene expression. Genes Dev 2000; 14: 391–396.
Masuda K, Abdelmohsen K, Gorospe M . RNA-binding proteins implicated in the hypoxic response. J Cell Mol Med 2009; 13: 2759–2769.
Dolt KS, Mishra MK, Karar J, Baig MA, Ahmed Z, Pasha MA . cDNA cloning, gene organization and variant specific expression of HIF-1 alpha in high altitude yak (Bos grunniens). Gene 2007; 386: 73–80.
Wang C, Song B, Song W, Liu J, Sun A, Wu D et al. Underexpressed microRNA-199b-5p targets hypoxia-inducible factor-1alpha in hepatocellular carcinoma and predicts prognosis of hepatocellular carcinoma patients. J Gastroenterol Hepatol 2011; 26: 1630–1637.
Moretti F, Thermann R, Hentze MW . Mechanism of translational regulation by miR-2 from sites in the 5′ untranslated region or the open reading frame. RNA 2010; 16: 2493–2502.
van Uden P, Kenneth NS, Webster R, Muller HA, Mudie S, Rocha S . Evolutionary conserved regulation of HIF-1beta by NF-kappaB. Plos Genet 2011; 7: e1001285.
Reaper PM, Griffiths MR, Long JM, Charrier JD, Maccormick S, Charlton PA et al. Selective killing of ATM- or p53-deficient cancer cells through inhibition of ATR. Nat Chem Biol 2011; 7: 428–430.
Nghiem P, Park PK, Kim Y, Vaziri C, Schreiber SL . ATR inhibition selectively sensitizes G1 checkpoint-deficient cells to lethal premature chromatin condensation. Proc Natl Acad Sci USA 2001; 98: 9092–9097.
Monferran S, Paupert J, Dauvillier S, Salles B, Muller C . The membrane form of the DNA repair protein Ku interacts at the cell surface with metalloproteinase 9. EMBO J 2004; 23: 3758–3768.
Acknowledgements
We thank Catherine Botanch, Emilie Mirey and Christine Bordier for their help, Dr Nathalie Mazure and Dr Delphine Larrieu for critical comments on the manuscript and Aymeric Poizot for English rereading. We acknowledge Dr P Calsou, Dr Y Martineau and Dr S Pyronnet for technical advices and reagents. This work was supported by the ‘Ligue Nationale Contre le Cancer’ (Equipe Labélisée, BS), the Cancéropole GSO (BS and CM), the Association contre le Cancer, ARC (2008-1102, CM) and the Radioprotection Committee of EDF (CM).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Fallone, F., Britton, S., Nieto, L. et al. ATR controls cellular adaptation to hypoxia through positive regulation of hypoxia-inducible factor 1 (HIF-1) expression. Oncogene 32, 4387–4396 (2013). https://doi.org/10.1038/onc.2012.462
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2012.462
Keywords
This article is cited by
-
Chloroquine prevents hypoxic accumulation of HIF-1α by inhibiting ATR kinase: implication in chloroquine-mediated chemosensitization of colon carcinoma cells under hypoxia
Pharmacological Reports (2023)
-
Inside the hypoxic tumour: reprogramming of the DDR and radioresistance
Cell Death Discovery (2020)
-
Genomic analysis of snub-nosed monkeys (Rhinopithecus) identifies genes and processes related to high-altitude adaptation
Nature Genetics (2016)