Abstract
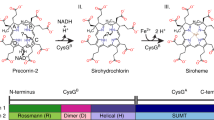
The crystal structure of the bromoperoxidase A2 from Streptomyces aureofaciens (ATCC 10762) has been determined by isomorphous replacement and refined to 2.05 Å resolution with an R-value of 18.4%. The enzyme catalyzes the bromination of organic compounds in the presence of bromide and peroxide. The structure confirms the absence of cofactors such as metal ions or haem groups and shows the general topology of the α/β hydrolase fold. The active centre is at the end of a deep pocket and includes a catalytic triad of Ser 98, Asp 228 and His 257. The active centre is connected by a narrow tunnel to a second pocket on the enzyme surface.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Morris, D.R., & Hager, L.P. Chloroperoxidase. Isolation and properties of the crystalline glycoprotein. J. biol. Chem. 241, 1763–1768 (1966).
Liu, T.N.E. et al. Isolation and characterization of a novel nonheme chloroperoxidase. Biochem. biophys. Res. Commun. 142, 329–333 (1987).
Manthey, J.A., & Hager, L.P. Purification and properties of bromoperoxidase from Penicillus capitatus. J. biol. Chem. 256, 11232–11238 (1981).
Vilter, H. Peroxidase from Phaeophyceae: a vanadium (V)-dependent peroxidase from Ascophyllum nodosum. Phytochemistry, 23, 1387–1380 (1984).
van Pée, K.-H., & Lingens, F. Purification and molecular and catalytic properties of bromoperoxidase from Streptomyces phaeochromogenes. J. gen. Microbiol. 131, 1911–1916 (1985).
van Pée, K.-H., Sury, G., & Lingens, F. purification and properties of a nonheme bromoperoxidase from Streptomyces aureofaciens. Hoppe-Seyler biol. Chem. 368, 5890–5894 (1987).
Dawson, J.H., & Sono, M. Cytochrom P-450 and chloroperoxidase: thiolate ligated heme enzymes, spectroscopic determination of their active site structures and mechanistic implications of thiolate ligation. Chem. Rev. 87, 1255–1276 (1987).
De Boer, E., van Kooyk, Y., Tromp, M.G.M., Plat, H., & Wever, R. Bromoperoxidase from Ascophyllum nodosum: a novel class of enzymes containing vanadium as a prostetic group?. Biochim. biophys. Acta 869, 48–53 (1986).
Wiesner, W., van Pée, K.-H. & Lingens, F. Purification and characterization of a novel bacterial non-heme chloroperoxidase from Pseudomonas pyrrocinia. J. Biol. Chem. 263, 13725–13732 (1988).
Bantleon, R., Altenbuchner, J., & van Pée, K.-H. Chloroperoxidase from Streptomyces lividans: isolation and characterization of the enzyme and the corresponding gene. J. Bacteriol. 176, 2339–2347 (1994).
Neidleman, S.L. & Geigert, J. in Biohalogenation: Principles, basic roles and applications (Ellis Horwood, Chichester, 1986).
de Boer, E., & Wever, R. The reaction mechanism of the novel vanadium-bromoperoxidase. J. biol. Chem. 263, 12326–12332 (1988).
Haag, T., Lingens, F., van Pee, K.-H. A metal-ion and cofactor independent enzymatic redox reaction: halogenation by bacterial nonheme haloperoxidases. Angew. Chem. Int. Ed. Engl. 30, 1487–1488 (1991).
Sobek, H. et al. Crystallization and preliminary X-ray data of bromoperoxidase from Streptomyces aureofaciens ATCC 10762. J. molec. Biol. 221, 35–37 (1991).
Ollis, D.L. et al. The α/β hydrolase fold. Prot. Engng. 5, 197–211 (1992).
Sussman, J.L. et al. Atomic structure of acetylcholinesterase from Torpedo californica: aprototypic acetylcholin-binding protein. Science 253, 872–879 (1991).
Bernstein, F.C. et al. The Protein Data Bank: a computer-based archival file for macromolecular structures. J. molec. Biol. 112, 535–542 (1977).
Franken, S.M., Rozeboom, H.J., Kalk, K.H., & Dijkstra, B.W. Crystal structure of haloalkane dehalogenase: an enzyme to detoxify halogenated alkanes. EMBO J. 10, 1297–1302 (1991).
Derewenda, U., Brzozowski, A.M., Lawson, D.M., & Derewenda, Z.S Catalysis at the interface: The anatomy of a conformational change in a triglyceride lipase. Biochemistry 31, 1532–1541 (1992).
Jones, T.A., Zou, J.-Y., Cowan, S.W., & Kjeldgaard, M. Improved methods for building protein models in electron density maps and the location of errors in these models. Acta Crystallogr. A47, 110–119 (1991).
Genetics Computer Group Program Manual for the GCG Package, Version 7 (575 Science Drive, Madison, Wisconsin, 1991).
Ookuni, I. & Fry, A. Hydrogen chloride catalyzed oxygen-18 exchange between para-substituted phenyl methyl sulfoxides and water. J. org. Chem. 36, 4097–4101 (1971).
Norne, J.-E., Lilja, H., Lindman, B., Einarsson, R., & Zeppezauer, M. Pt(CN)42- and Au(CN)2-: potential general probes for anion-binding sites of proteins. Eur. J. Biochem. 59, 463–473 (1975).
Weng, M., Pfeifer, O., Krauss, S., Lingens, F. & van Pée, K.-H. Purification, characterization and comparison of non-haem bromoperoxidases from Streptomyces aureofaciens ATCC 10762. J. gen. Microbiol. 137, 2539–2546 (1991).
Howard, A.J. et al. The use of an imaging proportional counter in macromolecular crystallography. J. Appl. Crystallogr. 20, 383–387 (1987).
CCP4 The SERC (UK) collaborative computing project no. 4, a suite of programs for protein crystallography (Daresbury Laboratory, UK, 1979).
Cowtan, K.D., & Main, P. Improvement of macromolecular electron-density maps by the simultaneous application of real and reciprocal space constraints. Acta Crystallogr. D49, 148–157 (1993).
Jones, T.A A graphics model building and refinement system for macromolecules. J. appl. Crystallosgr. 11, 268–272 (1978).
Brünger, A.T., Kuriyan, J., & Karplus, M. Refinement by simulated annealing. Science 235, 458–460 (1987).
Kabsch, W., & Sander, S. Dictionary of protein secondary structure: pattern recognition of hydrogen-bonded and geometrical features. Biopolymers 22, 2577–2637 (1983).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hecht, H., Sobek, H., Haag, T. et al. The metal-ion-free oxidoreductase from Streptomyces aureofaciens has an α/β hydrolase fold. Nat Struct Mol Biol 1, 532–537 (1994). https://doi.org/10.1038/nsb0894-532
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nsb0894-532
This article is cited by
-
Rationalizing perhydrolase activity of aryl-esterase and subtilisin Carlsberg mutants by molecular dynamics simulations of the second tetrahedral intermediate state
Theoretical Chemistry Accounts (2010)
-
Interaction of the SPG21 protein ACP33/maspardin with the aldehyde dehydrogenase ALDH16A1
neurogenetics (2009)
-
A novel variant of the catalytic triad in the Streptomyces scabies esterase
Nature Structural & Molecular Biology (1995)