Abstract
Impaired consciousness has long been considered the hallmark of epileptic seizures. Both generalized seizures and complex partial seizures are characterized by a multifaceted spectrum of altered conscious states, in terms of the general level of awareness and the subjective contents of consciousness. Complete loss of consciousness occurs when epileptic activity involves both cortical and subcortical structures, as in tonic–clonic seizures and absence seizures. Medial temporal lobe discharges can selectively impair experience in complex partial seizures (with affected responsiveness) and certain simple partial seizures (with unaffected responsiveness). Electrical stimulation of temporal lobe structures has been shown to evoke similar subjective experiences. Findings from neurophysiological and brain-imaging studies in epilepsy have now demonstrated that involvement of the bilateral thalamus and upper brainstem leads to selective impairment of frontoparietal association cortices and midline 'default mode' networks, which results in ictal loss of consciousness. The spread of epileptic discharges from the medial temporal lobe to the same subcortical structures can ultimately cause impairment in the level of consciousness in the late ictal and immediate postictal phase of complex partial seizures. This paper reviews novel insights into the brain mechanisms that underlie alterations of consciousness during epileptic seizures and the implications for clinical practice in terms of diagnosis and management.
Key Points
-
A patient's level of general awareness and subjective contents of consciousness can both be altered to some degree during epileptic seizures
-
Generalized seizures (tonic–clonic seizures and absence seizures) are characterized by complete loss of consciousness—that is, unresponsiveness in the absence of any ictal experience
-
Complex partial seizures (especially those with a medial temporal lobe focus) are associated with variable degrees of responsiveness and specific alterations in the subjective ictal experience
-
Neurophysiological and functional neuroimaging studies suggest that, in generalized and complex partial seizures, bilateral thalamus and upper brainstem involvement causes selective disruption of frontoparietal associative networks, which results in impaired consciousness
-
Ictal impairment of the general level of awareness seems related to transient disruption of frontoparietal and midline associative networks, which subserve 'default mode' brain function during the conscious resting state
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others

References
Zeman, A. Consciousness. Brain 124, 1263–1289 (2001).
Laureys, S. (ed) The Boundaries of Consciousness: Neurobiology and Neuropathology (Amsterdam: Elsevier, 2005).
Laureys, S. & Tononi, G. (eds) The Neurology of Consciousness (Amsterdam: Elsevier, 2009).
Blumenfeld, H. Consciousness and epilepsy: why are patients with absence seizures absent? Prog. Brain Res. 150, 271–286 (2005).
Monaco, F., Mula, M. & Cavanna, A. E. Consciousness, epilepsy and emotional qualia. Epilepsy Behav. 7, 150–160 (2005).
Engel, J. & International League Against Epilepsy. A proposed diagnostic scheme for people with epileptic seizures and with epilepsy: report of the ILAE Task Force on Classification and Terminology. Epilepsia 42, 796–803 (2001).
Zappulla, R. A. Epilepsy and consciousness. Semin. Neurol. 17, 113–119 (2007).
[No authors listed] Commission on Classification and Terminology of the International League Against Epilepsy Proposal for revised clinical and electroencephalographic classification of seizures. Epilepsia 22, 489–501 (1981).
Gloor, P. Consciousness as a neurological concept in epileptology: a critical review. Epilepsia 27 (Suppl. 2), 14–26 (1986).
Kalamangalam, G. P. Epilepsy and the physical basis of consciousness. Seizure 10, 484–491 (2001).
Johanson, M., Revonsuo, A., Chaplin, J. & Wedlund, J. E. Level and contents of consciousness in connection with partial epileptic seizures. Epilepsy Behav. 4, 279–285 (2003).
Johanson, M., Valli, K., Revonsuo, A., Chaplin, J. E. & Wedlund, J. E. Alterations in the contents of consciousness in partial epileptic seizures. Epilepsy Behav. 13, 366–371 (2008).
Johanson, M., Valli, K., Revonsuo, A. & Wedlund, J. E. Content analysis of subjective experiences in partial epileptic seizures. Epilepsy Behav. 12, 170–182 (2008).
Plum, F. & Posner, J. B. The Diagnosis of Stupor and Coma. 3rd edn (Davis, Philadelphia, 1980).
Blumenfeld, H. & Taylor, J. Why do seizures cause loss of consciousness? Neuroscientist 9, 1–10 (2003).
Blumenfeld, H. Epilepsy and consciousness in The Neurology of Consciousness. (eds Laureys, S. & Tononi, G.) 247–260 (Elsevier, Amsterdam, 2009).
Baars, B. J., Ramsøy, T. Z. & Laureys, S. Brain, conscious experience and the observing self. Trends Neurosci. 26, 671–675 (2003).
Laureys, S. & Boly, M. The changing spectrum of coma. Nat. Clin. Pract. Neurol. 4, 544–546 (2008).
Teasdale, G. & Jennett, B. Assessment of coma and impaired consciousness: a practical scale. Lancet 2, 81–84 (1974).
Alkire, M. T., Hudetz, A. G. & Tononi, G. Consciousness and anesthesia. Science 322, 876–880 (2008).
Critchley, H. D. Neural mechanisms of autonomic, affective, and cognitive integration. J. Comp. Neurol. 493, 154–166 (2005).
Gloor, P. Experiential phenomena of temporal lobe epilepsy: facts and hypotheses. Brain 113, 1673–1694 (1990).
Cavanna, A. E. et al. Measuring the level and contents of consciousness during epileptic seizures: the Ictal Consciousness Inventory. Epilepsy Behav. 13, 184–188 (2008).
Cavanna, A. E. Seizures and consciousness in Behavioral Aspects of Epilepsy: Principles and Practice (eds Schachter, S. C. et al.) (Demos, New York, 2008).
Tononi, G. & Koch, C. The neural correlates of consciousness: an update. Ann. NY Acad. Sci. 1124, 239–261 (2008).
Engel, J. Jr, Kuhl, D. E. & Phelps, M. E. Patterns of human local cerebral glucose metabolism during epileptic seizures. Science 218, 64–66 (1982).
Andre, V., Henry, D. & Nehlig, A. Dynamic variations of local cerebral blood flow in maximal electroshock seizures in the rat. Epilepsia 43, 1120–1128 (2002).
McCown, T. J., Duncan, G. E., Johnson, K. B. & Breese, G. R. Metabolic and functional mapping of the neural network subserving inferior collicular seizure generalization. Brain Res. 701, 117–128 (1995).
Ackermann, R. F., Engel, J. Jr & Baxter, L. Positron emission tomography and autoradiographic studies of glucose utilization following electroconvulsive seizures in humans and rats. Ann. NY Acad. Sci. 462, 263–269 (1986).
McIntyre, D. C., Don, J. C. & Edson, N. Distribution of 14C-2-deoxyglucose after various forms and durations of status epilepticus induced by stimulation of a kindled amygdala focus in rats. Epilepsy Res. 10, 119–133 (1991).
Lee, B. I. et al. HIPDM single photon emission computed tomography brain imaging in partial onset secondarily generalized tonic–clonic seizures. Epilepsia 28, 305–311 (1987).
Shin, W. C., Hong, S. B., Tae, W. S. & Kim, S. E. Ictal hyperperfusion patterns according to the progression of temporal lobe seizures. Neurology 58, 373–380 (2002).
Blumenfeld, H. From molecules to networks: cortical/subcortical interactions in the pathophysiology of idiopathic generalized epilepsy. Epilepsia 44 (Suppl. 2), 7–15 (2003).
Blumenfeld, H. et al. Cortical and subcortical networks in human secondarily generalized tonic-clonic seizures. Brain, in press (2009).
Blumenfeld, H., McNally, K. A., Ostroff, R. B. & Zubal, I. G. Targeted prefrontal cortical activation with bifrontal ECT. Psychiatry Res. 123, 165–170 (2003).
Blumenfeld, H. et al. Selective frontal, parietal, and temporal networks in generalized seizures. Neuroimage 19, 1556–1566 (2003).
McNally, K. A. & Blumenfeld, H. Focal network involvement in generalized seizures: new insights from electroconvulsive therapy. Epilepsy Behav. 5, 3–12 (2004).
Enev, M. et al. Imaging onset and propagation of ECT-induced seizures. Epilepsia 48, 238–244 (2007).
Hamandi, K. et al. EEG-fMRI of idiopathic and secondarily generalized epilepsies. Neuroimage 31, 1700–1710 (2006).
Bell, W. L., Walczak, T. S., Shin, C. & Radtke, R. A. Painful generalised clonic and tonic–clonic seizures with retained consciousness. J. Neurol. Neurosurg. Psychiatr. 63, 792–795 (1997).
Nogueira, R. G., Sheth, K. N., Duffy, F. H., Helmers, S. L. & Bromfield, E. B. Bilateral tonic–clonic seizures with temporal onset and preservation of consciousness. Neurology 70, 2188–2190 (2008).
Williams, D. A study of thalamic and cortical rhythms in petit mal. Brain 76, 50–69 (1953).
Blumenfeld, H. & McCormick, D. A. Corticothalamic inputs control the pattern of activity generated in thalamocortical networks. J. Neurosci. 20, 5153–5162 (2000).
Kostopoulos, G. K. Involvement of the thalamocortical system in epileptic loss of consciousness. Epilepsia 42, 13–19 (2001).
McCormick, D. A. & Contreras, D. On the cellular and network bases of epileptic seizures. Ann. Rev. Physiol. 63, 815–846 (2001).
Blumenfeld, H. The thalamus and seizures. Arch. Neurol. 59, 135–137 (2002).
Crunelli, V. & Leresche, N. Childhood absence epilepsy: genes, channels, neurons and networks. Nat. Rev. Neurosci. 3, 371–382 (2002).
Weir, B. The morphology of the spike–wave complex. Electroencephalogr. Clin. Neurophysiol. 19, 284–290 (1965).
Rodin, E. & Ancheta, O. Cerebral electrical fields during petit mal absences. Electroencephalogr. Clin. Neurophysiol. 66, 457–466 (1987).
Meeren, H. K., Pijn, J. P., Van Luijtelaar, E. L., Coenen, A. M. & Lopes da Silva, F. H. Cortical focus drives widespread corticothalamic networks during spontaneous absence seizures in rats. J. Neurosci. 22, 1480–1495 (2002).
Nersesyan, H., Hyder, F., Rothman, D. L. & Blumenfeld, H. Dynamic fMRI and EEG recordings during spike-wave seizures and generalized tonic–clonic seizures in WAG/Rij rats. J. Cereb. Blood Flow Metab. 24, 589–599 (2004).
Nersesyan, H., Herman, P., Erdogan, E., Hyder, F. & Blumenfeld, H. Relative changes in cerebral blood flow and neuronal activity in local microdomains during generalized seizures. J. Cereb. Blood Flow Metab. 24, 1057–1068 (2004).
Blumenfeld, H. Cellular and network mechanisms of spike-wave seizures. Epilepsia 46 (Suppl. 9), 21–33 (2005).
Van Luijtelaar, G. & Sitnikova, E. Global and focal aspects of absence epilepsy: the contribution of genetic models. Neurosci. Biobehav. Rev. 30, 983–1003 (2006).
Engel, J. Jr, Lubens, P., Kuhl, D. E. & Phelps, M. E. Local cerebral metabolic rate for glucose during petit mal absences. Ann. Neurol. 17, 121–128 (1985).
Prevett, M. C., Duncan, J. S., Jones, T. Fish, D. R. & Brooks, D. J. Demonstration of thalamic activation during typical absence seizures using H215O and PET. Neurology 45, 1396–1402 (1995).
Yeni, S. N., Kabasakal, L., Yalçinkaya, C., Nişli, C. & Dervent, A. Ictal and interictal SPECT findings in childhood absence epilepsy. Seizure 9, 265–269 (2000).
Holmes, M. D., Brown, M. & Tucker, D. M. Are “generalized” seizures truly generalized? Evidence of localized mesial frontal and frontopolar discharges in absence. Epilepsia 45, 1568–1579 (2004).
Ochs, R. F. et al. Effect of generalized spike-and-wave discharge on glucose metabolism measured by positron emission tomography. Ann. Neurol. 21, 458–464 (1987).
Diehl, B., Knecht, S., Deppe, M., Young, C. & Stodieck, S. R. Cerebral hemodynamic response to generalized spike-wave discharges. Epilepsia 39, 1284–1289 (1998).
Mirsky, A. F. & Van Buren, J. M. On the nature of the 'absence' in centrencephalic epilepsy: a study of some behavioral, electroencephalographic, and autonomic factors. Electroencephalogr. Clin. Neurophysiol. 18, 334–348 (1965).
Archer, J. S., Abbott, D. F., Waites, A. B. & Jackson, G. D. fMRI “deactivation” of the posterior cingulate during generalized spike and wave. Neuroimage 20, 1915–1922 (2003).
Salek-Haddadi, A. et al. Functional magnetic resonance imaging of human absence seizures. Ann. Neurol. 53, 663–667 (2003).
Aghakhani, Y. et al. fMRI activation during spike and wave discharges in idiopathic generalized epilepsy. Brain 127, 1127–1144 (2004).
Gotman, J. et al. Generalized epileptic discharges show thalamocortical activation and suspension of the default state of the brain. Proc. Natl Acad. Sci. USA 102, 15236–15240 (2005).
Labate, A., Briellmann, R. S., Abbott, D. F., Waites, A. B. & Jackson, G. D. Typical childhood absence seizures are associated with thalamic activation. Epileptic Disord. 7, 373–377 (2005).
Laufs, H., Lengler, U., Hamandi, K., Kleinschmidt, A. & Krakow, K. Linking generalized spike-and-wave discharges and resting state brain activity by using EEG/fMRI in a patient with absence seizures. Epilepsia 47, 444–448 (2006).
Williamson, P. D. et al. Characteristics of medial temporal lobe epilepsy: II. Interictal and ictal scalp electroencephalography, neuropsychological testing, neuroimaging, surgical results, and pathology. Ann. Neurol. 34, 781–787 (1993).
Alvarez-Silva, S. et al. Epileptic consciousness: concept and meaning of aura. Epilepsy Behav. 8, 527–533 (2006).
Hogan, R. E. & Kaiboriboon, K. The “dreamy state”: John Hughlings-Jackson's ideas of epilepsy and consciousness. Am. J. Psychiatry 160, 1740–1747 (2003).
Fried, I. Auras and experiential responses arising in the temporal lobe. J. Neuropsychiatry Clin. Neurosci. 9, 420–428 (1997).
Vignal, J. P., Maillard, L., McGonigal, A. & Chauvel, P. The dreamy state: hallucinations of autobiographic memory evoked by temporal lobe stimulations and seizures. Brain 130, 88–99 (2007).
Hughes, J. R. The idiosyncratic aspects of the epilepsy of Fyodor Dostoevsky. Epilepsy Behav. 7, 531–538 (2005).
Penfield, W. The Mystery of the Mind: a Critical Study of Consciousness and the Human Brain (Princeton University Press, 1975).
Bancaud, J., Brunet-Bourgin, F., Chauvel, P. & Halgren, E. Anatomical origin of deja vu and vivid 'memories' in human temporal lobe epilepsy. Brain 117, 71–90 (1994).
Inoue, Y. & Mihara, T. Awareness and responsiveness during partial seizures. Epilepsia 39 (Suppl. 5), 7–10 (1998).
Lee, K. H. et al. Pathophysiology of altered consciousness during seizures: subtraction SPECT study. Neurology 59, 841–846 (2002).
Lux, S. et al. The localizing value of ictal consciousness and its constituent functions: a video-EEG study in patients with focal epilepsy. Brain 125, 2691–2698 (2002).
Norden, A. D. & Blumenfeld, H. The role of subcortical structures in human epilepsy. Epilepsy Behav. 3, 219–231 (2002).
Mayanagi, Y., Watanabe, E. & Kaneko, Y. Mesial temporal lobe epilepsy: clinical features and seizure mechanism. Epilepsia 37 (Suppl. 3), 57–60 (1996).
Kaiboriboon, K., Bertrand, M. E., Osman, M. M. & Hogan, R. E. Quantitative analysis of cerebral blood flow patterns in mesial temporal lobe epilepsy using composite SISCOM. J. Nucl. Med. 46, 38–43 (2005).
Tae, W. S. et al. Cerebral perfusion changes in mesial temporal lobe epilepsy: SPM analysis of ictal and interictal SPECT. Neuroimage 24, 101–110 (2005).
Kim, J. H. et al. Ictal hyperperfusion patterns in relation to ictal scalp EEG patterns in patients with unilateral hippocampal sclerosis: a SPECT study. Epilepsia 48, 270–277 (2007).
Van Paesschen, W., Dupont, P., Van Driel, G., Van Billoen, H. & Maes, A. SPECT perfusion changes during complex partial seizures in patients with hippocampal sclerosis. Brain 126, 1103–1111 (2003).
McNally, K. A. et al. Localizing value of ictal-interictal SPECT analyzed by SPM (ISAS). Epilepsia 46, 1450–1464 (2005).
Blumenfeld, H. et al. Positive and negative network correlations in temporal lobe epilepsy. Cereb. Cortex 14, 892–902 (2004).
Blumenfeld, H. et al. Ictal neocortical slowing in temporal lobe epilepsy. Neurology 63, 1015–1021 (2004).
Gur, R. C. et al. Sex differences in regional cerebral glucose metabolism during a resting state. Science 267, 528–531 (1995).
Binder, J. R. et al. Conceptual processing during the conscious resting state: a functional MRI study. J. Cogn. Neurosci. 11, 80–93 (1999).
Raichle, M. E. et al. A default mode of brain function. Proc. Natl Acad. Sci. USA 98, 676–682 (2001).
Gusnard, D. A. & Raichle, M. E. Searching for a baseline: functional imaging and the resting human brain. Nat. Rev. Neurosci. 2, 685–694 (2001).
Mitchell, J. P., Heatherton, T. F. & Macrae, C. N. Distinct neural systems subserve person and object knowledge. Proc. Natl Acad. Sci. USA 99, 15238–15243 (2003).
Greicius, M. D., Krasnow, B., Reiss, A. L. & Menon, V. Functional connectivity in the resting brain: a network analysis of the default mode hypothesis. Proc. Natl Acad. Sci. USA 100, 253–258 (2003).
Cavanna, A. E. & Trimble, M. R. The precuneus: a review of its functional anatomy and behavioural correlates. Brain 129, 564–583 (2006).
Cavanna, A. E. The precuneus and consciousness. CNS Spectrums 12, 545–552 (2007).
Greicius, M. Resting-state functional connectivity in neuropsychiatric disorders. Curr. Opin. Neurol. 21, 424–430 (2008).
Vogt, B. A. & Laureys, S. Posterior cingulate, precuneal and retrosplenial cortices: cytology and components of the neural network correlates of consciousness. Prog. Brain Res. 150, 205–217 (2005).
Raichle, M. E. & Snyder, A. Z. Intrinsic brain activity and consciousness, in The Neurology of Consciousness Ch. 7 (eds Laureys, S. & Tononi, G.) (Elsevier, Amsterdam, 2009).
Schridde, U. et al. Negative BOLD with large increases in neuronal activity. Cereb. Cortex 18, 1814–1827 (2008).
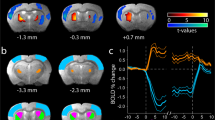
Englot, D. J. et al. Remote effects of focal hippocampal seizures on the rat neocortex. J. Neurosci. 28, 9066–9081 (2008).
Acknowledgements
The authors wish to thank Dr Hugh Rickards for his insightful comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Cavanna, A., Monaco, F. Brain mechanisms of altered conscious states during epileptic seizures. Nat Rev Neurol 5, 267–276 (2009). https://doi.org/10.1038/nrneurol.2009.38
Issue Date:
DOI: https://doi.org/10.1038/nrneurol.2009.38