Key Points
-
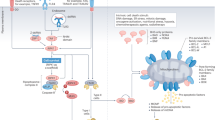
The clinical application of immunotherapy against cancer is rapidly moving forward in multiple areas, including the adoptive transfer of antitumour-reactive T cells and the use of 'therapeutic' vaccines. The latter aims at inducing cytotoxic T lymphocytes (CTLs) specific for tumour-associated antigens presented by cancer cells in the context of human leukocyte antigen (HLA) molecules.
-
Surprisingly, until recently only limited attention has been focused on identifying the most suitable targets for the induction of clinically relevant anticancer immune responses, the delineation of the most effective epitopes within these antigens and, finally, inclusion of sets of peptide epitopes best suited for targeting the disease in question.
-
The vast majority of malignancies are characterized by defects in apoptosis signalling, which is mediated by two group of apoptosis regulators: the BCL2 family (for example, BCL2, BCL-XL and MCL1) and the inhibitor of apoptosis proteins (IAP), such as survivin or melanoma IAP.
-
Such apoptosis regulators are therefore crucial cellular factors contributing to the pathogenesis and progression of cancer. Consequently, they represent very attractive targets for the design of new anticancer drugs, such as antisense oligonucleotides and small-molecule BCL2 inhibitors.
-
Notably, recently spontaneous cellular immune responses against these proteins have been identified as frequent features in cancer patients. Here, we summarize current knowledge of IAP and BCL2 family proteins as T-cell antigens, report the results of the first exploratory trials using these antigens for immunotherapy and discuss future opportunities, such as simultaneous targeting of several proteins or combination with conventional chemotherapy, because their expression in tumours is correlated with drug resistance and/or poor prognosis.
-
Immunotherapy based on survivin, BCL2 or MCL1 might also compromise endothelial-cell viability and interfere with tumour angiogenesis. Targeting survivin or BCL2 family members in angiogenic immunotherapy might therefore deliver two distinct and potentially synergistic treatment modalities using a common procedure.
-
In conclusion, a multi-epitope vaccine targeting regulators of apoptosis seems to be a promising, universal antitumour vaccine, which addresses both the tumour and the tumour stroma. Moreover, it synergistically boosts the effects of conventional cytotoxic therapies or radiation.
Abstract
Harnessing the immune system in the battle against cancer has been the focus of tremendous research efforts during the past two decades. Several means for achieving this goal, including adoptive transfer of tumour-reactive T cells, systemic or localized administration of immune modulating cytokines and the use of 'therapeutic' vaccines, have been explored. Anti-apoptotic molecules that enhance the survival of cancer cells and facilitate their escape from cytotoxic therapies represent prime candidates as vaccination antigens. Notably, spontaneous cellular immune responses against these proteins have frequently been identified in cancer patients. Here, we summarize current knowledge of IAP and BCL2 family proteins as T-cell antigens, report the results of the first explorative trial using these antigens in therapeutic vaccinations against cancer and discuss future opportunities.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Van den Eynde, B. J. & Boon, T. Tumor antigens recognized by T lymphocytes. Int. J. Clin. Lab. Res. 27, 81–86 (1997).
Andersen, M. H., Pedersen, L. O., Becker, J. C. & thor Straten, P. Identification of a cytotoxic T lymphocyte response to the apoptose inhibitor protein survivin in cancer patients. Cancer Res. 61, 869–872 (2001). First description of natural immunity against survivin in cancer patients.
Nestle, F. O. et al. Vaccination of melanoma patients with peptide- or tumor lysate-pulsed dendritic cells. Nature Med. 4, 328–332 (1998).
Smyth, M. J., Godfrey, D. I. & Trapani, J. A. A fresh look at tumor immunosurveillance and immunotherapy. Nature Immunol. 2, 293–299 (2001).
Jocham, D. et al. Adjuvant autologous renal tumour cell vaccine and risk of tumour progression in patients with renal-cell carcinoma after radical nephrectomy: phase III, randomised controlled trial. Lancet 363, 594–599 (2004). Data from the first Phase III vaccination trial suggesting that therapeutic vaccination might have an impact on disease.
Sosman, J. A. & Sondak, V. K. Melacine: an allogeneic melanoma tumor cell lysate vaccine. Expert. Rev. Vaccines. 2, 353–368 (2003).
Melief, C. J. et al. Strategies for immunotherapy of cancer. Adv. Immunol. 75, 235–282 (2000).
Novellino, L., Castelli, C. & Parmiani, G. A listing of human tumor antigens recognized by T cells: March 2004 update. Cancer Immunol. Immunother. 54, 187–207 (2004).
Riker, A. et al. Immune selection after antigen-specific immunotherapy of melanoma. Surgery 126, 112–120 (1999).
Lee, K. H. et al. Functional dissociation between local and systemic immune response during anti-melanoma peptide vaccination. J. Immunol. 161, 4183–4194 (1998).
Pawelec, G., Zeuthen, J. & Kiessling, R. Escape from host-antitumor immunity. Crit. Rev. Oncog. 8, 111–141 (1997).
Lehmann, F. et al. Differences in the antigens recognized by cytolytic T cells on two successive metastases of a melanoma patient are consistent with immune selection. Eur. J. Immunol. 25, 340–347 (1995).
Wang, Z. et al. Molecular analysis of the HLA-A2 antigen loss by melanoma cells SK-MEL- 29.1.22 and SK-MEL-29.1.29. Cancer Res. 58, 2149–2157 (1998).
Jager, E., Jager, D. & Knuth, A. Antigen-specific immunotherapy and cancer vaccines. Int. J. Cancer 106, 817–820 (2003).
Hadzantonis, M. & O'Neill, H. Review: dendritic cell immunotherapy for melanoma. Cancer Biother. Radiopharm. 14, 11–22 (1999).
Brinkman, J. A., Fausch, S. C., Weber, J. S. & Kast, W. M. Peptide-based vaccines for cancer immunotherapy. Expert. Opin. Biol. Ther. 4, 181–198 (2004).
Klinman, D. M., Currie, D., Gursel, I. & Verthelyi, D. Use of CpG oligodeoxynucleotides as immune adjuvants. Immunol. Rev. 199, 201–216 (2004).
Walden, P. & Sterry, W. New and emerging vaccination strategies for prevention and treatment of dermatological diseases. Expert. Rev. Vaccines. 3, 421–431 (2004).
Schadendorf, D. Melanoma vaccines. Drug News Perspect. 13, 85–90 (2000).
Strengell, M. et al. IL-21 in synergy with IL-15 or IL-18 enhances IFN-γ production in human NK and T cells. J. Immunol. 170, 5464–5469 (2003).
Coulie, P. G. et al. Cytolytic T-cell responses of cancer patients vaccinated with a MAGE antigen. Immunol. Rev. 188, 33–42 (2002).
Shangary, S. & Johnson, D. E. Recent advances in the development of anticancer agents targeting cell death inhibitors in the Bcl-2 protein family. Leukemia 17, 1470–1481 (2003).
Reed, J. C. Apoptosis-based therapies. Nature Rev. Drug Discov. 1, 111–121 (2002). Strategies aimed at inhibiting the expression or functions of anti-apoptotic proteins have gained considerable therapeutic attention in cancer — for example, antisense oligonucleotides or small-molecule inhibitors.
Makin, G. & Hickman, J. A. Apoptosis and cancer chemotherapy. Cell Tissue Res. 301, 143–152 (2000).
Longley, D. B. & Johnston, P. G. Molecular mechanisms of drug resistance. J. Pathol. 205, 275–292 (2005).
Kim, R., Emi, M., Tanabe, K. & Toge, T. Therapeutic potential of antisense Bcl-2 as a chemosensitizer for cancer therapy. Cancer 101, 2491–2502 (2004).
Wacheck, V. et al. Bcl-X(L) antisense oligonucleotides radiosensitise colon cancer cells. Br. J. Cancer 89, 1352–1357 (2003).
Wang, S., Yang, D. & Lippman, M. E. Targeting Bcl-2 and Bcl-XL with nonpeptidic small-molecule antagonists. Semin. Oncol. 30, 133–142 (2003).
Falkson, C. I. et al. Phase III trial of dacarbazine versus dacarbazine with interferon α-2b versus dacarbazine with tamoxifen versus dacarbazine with interferon α-2b and tamoxifen in patients with metastatic malignant melanoma: an Eastern Cooperative Oncology Group study. J. Clin. Oncol. 16, 1743–1751 (1998).
Huang, D. C. & Strasser, A. BH3-Only proteins-essential initiators of apoptotic cell death. Cell 103, 839–842 (2000).
Walensky, L. D. et al. Activation of apoptosis in vivo by a hydrocarbon-stapled BH3 helix. Science 305, 1466–1470 (2004).
Letai, A. et al. Distinct BH3 domains either sensitize or activate mitochondrial apoptosis, serving as prototype cancer therapeutics. Cancer Cell 2, 183–192 (2002).
Reed, J. C. Apoptosis-targeted therapies for cancer. Cancer Cell 3, 17–22 (2003).
Byrd, J. C. et al. The mechanism of tumor cell clearance by rituximab in vivo in patients with B-cell chronic lymphocytic leukemia: evidence of caspase activation and apoptosis induction. Blood 99, 1038–1043 (2002).
Islam, A. et al. High expression of Survivin, mapped to 17q25, is significantly associated with poor prognostic factors and promotes cell survival in human neuroblastoma. Oncogene 19, 617–623 (2000).
Swana, H. S., Grossman, D., Anthony, J. N., Weiss, R. M. & Altieri, D. C. Tumor content of the antiapoptosis molecule survivin and recurrence of bladder cancer. N. Engl. J. Med. 341, 452–453 (1999).
Kawasaki, H. et al. Inhibition of apoptosis by survivin predicts shorter survival rates in colorectal cancer. Cancer Res. 58, 5071–5074 (1998).
Altieri, D. C. Validating survivin as a cancer therapeutic target. Nature Rev. Cancer 3, 46–54 (2003). An in-depth review, which describes the potential applicability of survivin in the clinic.
Olie, R. A. et al. A novel antisense oligonucleotide targeting survivin expression induces apoptosis and sensitizes lung cancer cells to chemotherapy. Cancer Res. 60, 2805–2809 (2000).
Gazzaniga, P. et al. Expression and prognostic significance of LIVIN, SURVIVIN and other apoptosis-related genes in the progression of superficial bladder cancer. Ann. Oncol. 14, 85–90 (2003).
Fulda, S., Wick, W., Weller, M. & Debatin, K. M. Smac agonists sensitize for Apo2L/TRAIL- or anticancer drug-induced apoptosis and induce regression of malignant glioma in vivo. Nature Med. 8, 808–815 (2002).
Altieri, D. C., Marchisio, P. C. & Marchisio, C. Survivin apoptosis: an interloper between cell death and cell proliferation in cancer. Lab. Invest. 79, 1327–1333 (1999).
Ambrosini, G., Adida, C., Sirugo, G. & Altieri, D. C. Induction of apoptosis and inhibition of cell proliferation by survivin gene targeting. J. Biol. Chem. 273, 11177–11182 (1998).
Adida, C. et al. Prognostic significance of survivin expression in diffuse large B-cell lymphomas. Blood 96, 1921–1925 (2000).
Grossman, D., McNiff, J. M., Li, F. & Altieri, D. C. Expression and targeting of the apoptosis inhibitor, survivin, in human melanoma. J. Invest Dermatol. 113, 1076–1081 (1999).
Velculescu, V. E. et al. Analysis of human transcriptomes. Nature Genet. 23, 387–388 (1999).
Yang, D., Welm, A. & Bishop, J. M. Cell division and cell survival in the absence of survivin. Proc. Natl Acad. Sci. USA 101, 15100–15105 (2004).
Li, F. et al. Control of apoptosis and mitotic spindle checkpoint by survivin. Nature 396, 580–584 (1998).
Schmitz, M. et al. Generation of survivin-specific CD8+ T effector cells by dendritic cells pulsed with protein or selected peptides. Cancer Res. 60, 4845–4849 (2000).
Andersen, M. H. et al. Spontaneous cytotoxic T-cell responses against survivin-derived MHC class I-restricted T-cell epitopes in situ as well as ex vivo in cancer patients. Cancer Res. 61, 5964–5968 (2001).
Reker, S. et al. HLA-B35-restricted immune responses against survivin in cancer patients. Int. J. Cancer 108, 937–941 (2004).
Casati, C. et al. The apoptosis inhibitor protein Survivin induces tumor-specific CD8(+) and CD4(+) T cells in colorectal cancer patients. Cancer Res. 63, 4507–4515 (2003).
Reker, S. et al. Identification of novel Survivin-derived CTL epitopes. Cancer Biol. Ther. 3, (2004).
Schmidt, S. M. et al. Survivin is a shared tumor-associated antigen expressed in a broad variety of malignancies and recognized by specific cytotoxic T cells. Blood 102, 571–576 (2003). Survivin-specific CTL generated from healthy donors or leukaemia patients elicited cytolytic activity against a large number of tumour cells of different origin.
Coughlin, C. M., Vance, B. A., Grupp, S. A. & Vonderheide, R. H. RNA-transfected CD40-activated B cells induce functional T-cell responses against viral and tumor antigen targets: implications for pediatric immunotherapy. Blood 103, 2046–2054 (2004).
thor Straten, P., Guldberg, P., Grønbæk, K., Zeuthen, J. & Becker, J. C. In situ T-cell responses against melanoma comprise high numbers of locally expanded T-cell clonotypes. J. Immunol. 163, 443–447 (1999).
Otto, K. et al. Therapy-induced T cell responses against the universal tumor antigen survivin. Vaccine 23, 884–889 (2004).
Pisarev, V. et al. Full-length dominant-negative survivin for cancer immunotherapy. Clin. Cancer Res. 9, 6523–6533 (2003).
Tsuruma, T. et al. Phase I clinical study of anti-apoptosis protein, survivin-derived peptide vaccine therapy for patients with advanced or recurrent colorectal cancer. J. Transl. Med. 2, 19 (2004).
Kasof, G. M. & Gomes, B. C. Livin, a novel inhibitor of apoptosis protein family member. J. Biol. Chem. 276, 3238–3246 (2001).
Vucic, D., Stennicke, H. R., Pisabarro, M. T., Salvesen, G. S. & Dixit, V. M. ML-IAP, a novel inhibitor of apoptosis that is preferentially expressed in human melanomas. Curr. Biol. 10, 1359–1366 (2000).
Ashhab, Y., Alian, A., Polliack, A., Panet, A. & Ben Yehuda, D. Two splicing variants of a new inhibitor of apoptosis gene with different biological properties and tissue distribution pattern. FEBS Lett. 495, 56–60 (2001).
Schmollinger, J. C. et al. Melanoma inhibitor of apoptosis protein (ML-IAP) is a target for immune-mediated tumor destruction. Proc. Natl Acad. Sci. USA 100, 3398–3403 (2003). First description of ML-IAP/livin as a tumour-associated antigen.
Andersen, M. H., Becker, J. C. & thor Straten, P. Identification of an HLA-A3 restricted CTL epitope from ML-IAP. J. Invest. Dermatol. 122, 1336–1337 (2004).
Andersen, M. H., Reker, S., Becker, J. C. & thor Straten, P. The melanoma inhibitor of apoptosis protein: a target for spontaneous cytotoxic T cell responses. J. Invest. Dermatol. 122, 392–399 (2004).
D'Souza, S. et al. Circulating Melan-A/Mart-1 specific cytolytic T lymphocyte precursors in HLA-A2+ melanoma patients have a memory phenotype. Int. J. Cancer 78, 699–706 (1998).
Jäger, E. et al. Cytolytic T cell reactivity against melanoma-associated differentiation antigens in peripheral blood of melanoma patients and healthy individuals. Melanoma Res. 6, 419–425 (1996).
Griffioen, M., Borghi, M., Schrier, P. I. & Osanto, S. Detection and quantification of CD8(+) T cells specific for HLA-A*0201-binding melanoma and viral peptides by the IFN-γ-ELISPOT assay. Int. J. Cancer 93, 549–555 (2001).
Rudin, C. M. et al. A pilot trial of G3139, a bcl-2 antisense oligonucleotide, and paclitaxel in patients with chemorefractory small-cell lung cancer. Ann. Oncol. 13, 539–545 (2002).
Reed, J. C. et al. BCL-2 family proteins: regulators of cell death involved in the pathogenesis of cancer and resistance to therapy. J. Cell Biochem. 60, 23–32 (1996).
Hua, C. et al. Mechanism of bcl-2 activation in human follicular lymphoma. Oncogene 5, 233–235 (1990).
Schimmer, A. D., Munk-Pedersen, I., Minden, M. D. & Reed, J. C. Bcl-2 and apoptosis in chronic lymphocytic leukemia. Curr. Treat. Options. Oncol. 4, 211–218 (2003).
Wang, H. G., Takayama, S., Rapp, U. R. & Reed, J. C. Bcl-2 interacting protein, BAG-1, binds to and activates the kinase Raf-1. Proc. Natl Acad. Sci. USA 93, 7063–7068 (1996).
Coultas, L. & Strasser, A. The role of the Bcl-2 protein family in cancer. Semin. Cancer Biol. 13, 115–123 (2003).
Krajewska, M. et al. Immunohistochemical analysis of bcl-2, bax, bcl-X, and mcl-1 expression in prostate cancers. Am. J. Pathol. 148, 1567–1576 (1996).
Zapata, J. M. et al. Expression of multiple apoptosis-regulatory genes in human breast cancer cell lines and primary tumors. Breast Cancer Res. Treat. 47, 129–140 (1998).
Tang, L. et al. Expression of apoptosis regulators in cutaneous malignant melanoma. Clin. Cancer Res. 4, 1865–1871 (1998).
Andersen, M. H. et al. Immunogenicity of Bcl-2 in cancer patients. Blood 105, 728–734 (2005). First description of the natural immunity against a member of the BCL2 family in cancer patients.
Andersen, M. H., Becker, J. C. & thor Straten, P. The anti-apoptotic member of the Bcl-2 family Mcl-1 is a CTL target in cancer patients. Leukemia 19, 484–485, (2005). First description of the natural immunity against MCL1 in cancer patients.
Andersen, M. H., Reker, S., Kvistborg, P., Becker, J. C. & thor Straten, P. Spontaneous immunity against Bcl-XL in cancer patients. submit f. pub (2005). First description of the natural immunity against a member of BCL-X L in cancer patients.
Moshynska, O., Sankaran, K., Pahwa, P. & Saxena, A. Prognostic significance of a short sequence insertion in the MCL-1 promoter in chronic lymphocytic leukemia. J. Natl Cancer Inst. 96, 673–682 (2004).
Tanaka, K. et al. Expression of survivin and its relationship to loss of apoptosis in breast carcinomas. Clin. Cancer Res. 6, 127–134 (2000).
Nakagawara, A. Molecular basis of spontaneous regression of neuroblastoma: role of neurotrophic signals and genetic abnormalities. Hum. Cell 11, 115–124 (1998).
Cameron, R. B., Spiess, P. J. & Rosenberg, S. A. Synergistic antitumor activity of tumor-infiltrating lymphocytes, interleukin 2, and local tumor irradiation. Studies on the mechanism of action. J. Exp. Med. 171, 249–263 (1990).
Levings, M. K., Sangregorio, R. & Roncarolo, M. G. Human cd25(+)cd4(+) t regulatory cells suppress naive and memory T cell proliferation and can be expanded in vitro without loss of function. J. Exp. Med. 193, 1295–1302 (2001).
Ng, W. F. et al. Human CD4(+)CD25(+) cells: a naturally occurring population of regulatory T cells. Blood 98, 2736–2744 (2001).
Fry, T. J. & Mackall, C. L. Interleukin-7: master regulator of peripheral T-cell homeostasis? Trends Immunol. 22, 564–571 (2001).
Tandle, A., Blazer, D. G., III & Libutti, S. K. Antiangiogenic gene therapy of cancer: recent developments. J. Transl. Med. 2, 22 (2004).
Niethammer, A. G. et al. A DNA vaccine against VEGF receptor 2 prevents effective angiogenesis and inhibits tumor growth. Nature Med. 8, 1369–1375 (2002).
Dias, S., Shmelkov, S. V., Lam, G. & Rafii, S. VEGF(165) promotes survival of leukemic cells by Hsp90-mediated induction of Bcl-2 expression and apoptosis inhibition. Blood 99, 2532–2540 (2002).
Katoh, O. et al. Vascular endothelial growth factor inhibits apoptotic death in hematopoietic cells after exposure to chemotherapeutic drugs by inducing MCL1 acting as an antiapoptotic factor. Cancer Res. 58, 5565–5569 (1998).
Xiang, R. et al. A DNA vaccine targeting survivin combines apoptosis with suppression of angiogenesis in lung tumor eradication. Cancer Res. 65, 553–561 (2005). Murine model suggesting that survivin-based vaccination target both the tumour and the tumour stroma.
Nair, S. et al. Synergy between tumor immunotherapy and antiangiogenic therapy. Blood 102, 964–971 (2003).
Lange-Asschenfeldt, B. et al. The angiogenesis inhibitor vasostatin does not impair wound healing at tumor-inhibiting doses. J. Invest. Dermatol. 117, 1036–1041 (2001).
Quinn, T. E., Thurman, G. B., Sundell, A. K., Zhang, M. & Hellerqvist, C. G. CM101, a polysaccharide antitumor agent, does not inhibit wound healing in murine models. J. Cancer Res. Clin. Oncol. 121, 253–256 (1995).
Rosenberg, S. A. et al. Immunologic and therapeutic evaluation of a synthetic peptide vaccine for the treatment of patients with metastatic melanoma. Nature Med. 4, 321–327 (1998).
Bauer, S. et al. Activation of NK cells and T cells by NKG2D, a receptor for stress-inducible MICA. Science 285, 727–729 (1999).
Groh, V. et al. Broad tumor-associated expression and recognition by tumor-derived γδ T cells of MICA and MICB. Proc. Natl Acad. Sci. USA. 96, 6879–6884 (1999).
Vetter, C. S. et al. Expression of stress-induced MHC class I related chain molecules on human melanoma. J. Invest. Dermatol. 118, 600–605 (2002).
Pedersen, L. O. et al. Differential expression of inhibitory or activating CD94/NKG2 subtypes on MART-1-reactive T cells in vitiligo versus melanoma: a case report. J. Invest. Dermatol. 118, 595–599 (2002).
Cormier, J. N. et al. Comparative analysis of the in vivo expression of tyrosinase, MART-1/Melan-A, and gp100 in metastatic melanoma lesions: implications for immunotherapy. J. Immunother. 21, 27–31 (1998).
Maeurer, M. J. et al. Tumor escape from immune recognition: lethal recurrent melanoma in a patient associated with downregulation of the peptide transporter protein TAP-1 and loss of expression of the immunodominant MART-1/Melan-A antigen. J. Clin. Invest 98, 1633–1641 (1996).
Offringa, R., van der Burg, S. H., Ossendorp, F., Toes, R. E. & Melief, C. J. Design and evaluation of antigen-specific vaccination strategies against cancer. Curr. Opin. Immunol. 12, 576–582 (2000).
Kursar, M. et al. Regulatory CD4+CD25+ T cells restrict memory CD8+ T cell responses. J. Exp. Med. 196, 1585–1592 (2002).
Moss, P. A., Rosenberg, W. M. & Bell, J. I. The human T cell receptor in health and disease. Annu. Rev. Immunol. 10, 71–96 (1992).
Stockwin, L. H., McGonagle, D., Martin, I. G. & Blair, G. E. Dendritic cells: immunological sentinels with a central role in health and disease. Immunol. Cell Biol. 78, 91–102 (2000).
Castelli, C. et al. T-cell recognition of melanoma-associated antigens. J. Cell Physiol 182, 323–331 (2000).
Siegel, S. et al. Identification of a survivin-derived peptide that induces HLA-A*0201-restricted antileukemia cytotoxic T lymphocytes. Leukemia 18, 2046–2047 (2004).
Hirohashi, Y. et al. An HLA-A24-restricted Cytotoxic T Lymphocyte Epitope of a Tumor-associated Protein, Survivin. Clin. Cancer Res. 8, 1731–1739 (2002).
Siegel, S., Wagner, A., Schmitz, N. & Zeis, M. Induction of antitumour immunity using survivin peptide-pulsed dendritic cells in a murine lymphoma model. Br. J. Haematol. 122, 911–914 (2003).
Andersen, M. H., Kvistborg, P., Becker, J. C. & thor Straten, P. Identification of an HLA-A1 restricted CTL epitope from Mcl-1. Leukemia 7 April 2005 [Epub ahead of print].
Acknowledgements
This work was funded in part by grants from the Danish Medical Research Council, The Danish Cancer Society and Danish Cancer Research Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- MAJOR HISTOCOMPATIBILITY COMPLEX
-
A complex of genetic loci in higher vertebrates that encodes a family of cellular antigens that allow the immune system to recognize self from non-self.
- ADJUVANT
-
An agent mixed with an antigen that enhances the immune response to that antigen upon immunization.
- CpG DINUCLEOTIDES
-
Cytosine–guanosine pairs inDNA sequences. Oligodeoxynucleotide sequences that include CpG dinucleotides and certain flanking nucleotides have been found to induce innate immune responses.
Rights and permissions
About this article
Cite this article
Andersen, M., Becker, J. & Straten, P. Regulators of apoptosis: suitable targets for immune therapy of cancer. Nat Rev Drug Discov 4, 399–409 (2005). https://doi.org/10.1038/nrd1717
Issue Date:
DOI: https://doi.org/10.1038/nrd1717
This article is cited by
-
Tumor microenvironment antigens
Seminars in Immunopathology (2023)
-
Inducing Apoptosis and Decreasing Cell Proliferation in Human Acute Promyelocytic Leukemia Through Regulation Expression of CASP3 by Let-7a-5p Blockage
Indian Journal of Hematology and Blood Transfusion (2018)
-
Crystal structure of caspase recruiting domain (CARD) of apoptosis repressor with CARD (ARC) and its implication in inhibition of apoptosis
Scientific Reports (2015)
-
Bioinformatics for cancer immunotherapy target discovery
Cancer Immunology, Immunotherapy (2014)
-
Effector CD4 and CD8 T Cells and Their Role in the Tumor Microenvironment
Cancer Microenvironment (2013)