Key Points
-
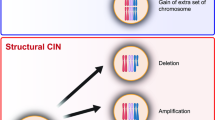
As core components of spindle poles, centrosomes have a key role in directing the formation of bipolar mitotic spindles, which is crucial for accurate segregation of chromosomes during cytokinesis.
-
Numeral and functional abnormalities of centrosomes result in mitotic spindle defects, leading to chromosome segregation errors, and are the major causes of chromosome instability in cancer, which accelerates the step-wise tumour progression.
-
Several oncogenic and tumour-suppressor proteins are found to be involved in the regulation of centrosome duplication and function, and mutations of those proteins result in mitotic defects that are associated with numeral and functional abnormalities of centrosomes.
-
The centrosome regulation activities of these oncogenic and tumour-suppressor proteins are crucial parts of their overall oncogenic, tumour-suppressing potential.
Abstract
Chromosome instability, which is equated to mitotic defects and consequential chromosome segregation errors, provides a formidable basis for the acquisition of further malignant phenotypes during tumour progression. Centrosomes have a crucial role in the formation of bipolar mitotic spindles, which are essential for accurate chromosome segregation. Mutations of certain oncogenic and tumour-suppressor proteins directly induce chromosome instability by disrupting the normal function and numeral integrity of centrosomes. How these proteins control centrosome duplication and function, and how their mutational activation and/or inactivation results in numeral and functional centrosome abnormalities, is discussed in this Review.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Mazia, D. The chromosome cycle and the centrosome cycle in the mitotic cycle. Int. Rev. Cytology 100, 49–92 (1987).
Hinchcliffe, E. H. & Sluder, G. Two for two: Cdk2 and its role in centrosome doubling. Oncogene 21, 6154–6160 (2002).
Fukasawa, K. Centrosome amplification, chromosome instability and cancer development. Cancer Lett. 230, 6–19 (2005).
D'Assoro, A. B., Lingle, W. L. & Salisbury, J. L. Centrosome amplification and the development of cancer. Oncogene 21, 6146–6153 (2002).
Uetake, Y. & Sluder, G. Cell cycle progression after cleavage failure: mammalian somatic cells do not possess a “tetraploidy checkpoint”. J. Cell Biol. 165, 609–615 (2004).
Levine, D. S., Sanchez, C. A., Rabinovitch, P. S. & Reid, B. J. Formation of the tetraploid intermediate is associated with the development of cells with more than four centrioles in the elastase-simian virus 40 tumor antigen transgenic mouse model of pancreatic cancer. Proc. Natl Acad. Sci. USA 88, 6427–6431 (1991).
Quintyne, N. J., Reing, J. E., Hoffelder, D. R., Gollin, S. M. & Saunders, W. S. Spindle multipolarity is prevented by centrosomal clustering. Science 307, 127–129 (2005). This study explores the mechanistic basis of the formation of pseudo-bipolar mitotic spindle in cells containing amplified centrosomes.
Tucker, J. D. & Preston, R. J. Chromosome aberrations, micronuclei, aneuploidy, sister chromatid exchanges, and cancer risk assessment. Mutat. Res. 365, 147–159 (1996).
Heichman, K. A. & Roberts, J. M. Rules to replicate by. Cell 79, 557–562 (1994).
Hinchcliffe, E. H., Li, C., Thompson, E. A., Maller, J. L. & Sluder, G. Requirement of Cdk2–cyclin E activity for repeated centrosome reproduction in Xenopus egg extracts. Science 283, 851–854 (1999).
Lacey, K. R., Jackson, P. K. & Stearns, T. Cyclin-dependent kinase control of centrosome duplication. Proc. Natl Acad. Sci. USA 96, 2817–2822 (1999).
Matsumoto, Y., Hayashi, K. & Nishida, E. Cyclin-dependent kinase 2 (Cdk2) is required for centrosome duplication in mammalian cells. Curr. Biol. 9, 429–432 (1999).
Mussman, J. G. et al. Synergistic induction of centrosome hyperamplification by loss of p53 and cyclin E overexpression. Oncogene 19, 1635–1946 (2000).
Balczon, R. et al. Dissociation of centrosome replication events from cycles of DNA synthesis and mitotic division in hydroxyurea-arrested Chinese hamster ovary cells. J. Cell Biol. 130, 105–115 (1995). This study provides the basis of the experimental system commonly used today for examination of the regulation of centrosome duplication, and also indicates the requirement of acquisition of duplication competency for centrosomes to initiate duplication.
Wong, C. & Stearns, T. Centrosome number is controlled by a centrosome-intrinsic block to reduplication. Nature Cell Biol. 5, 539–544 (2003). This study demonstrates clearly the requirement of acquisition of duplication competency for centrosomes to initiate duplication.
Tsou, M. F. & Stearns, T. Controlling centrosome number: licenses and blocks. Curr. Opin. Cell Biol. 18, 74–78 (2006).
Keyomarsi, K. & Herliczek, T. W. The role of cyclin E in cell proliferation, development and cancer. Prog. Cell Cycle Res. 3, 171–191 (1997).
Spruck, C. H., Won, K. A. & Reed, S. I. Deregulated cyclin E induces chromosome instability. Nature 401, 297–300 (1999).
Kawamura, K. et al. Induction of centrosome amplification and chromosome instability in human bladder cancer cells by p53 mutation and cyclin E overexpression. Cancer Res. 64, 4800–4809 (2004).
Okuda, M. et al. Nucleophosmin/B23 is a target of CDK2-cyclin E in centrosome duplication. Cell 103, 127–140 (2000).
Yung, B. Y., Busch, H. & Chan, P. K. Translocation of nucleolar phosphoprotein B23 (37 kDa/pI 5.1) induced by selective inhibitors of ribosome synthesis. Biochim. Biophys. Acta 826, 167–173 (1985).
Szebeni, A. & Olson, M. O. Nucleolar protein B23 has molecular chaperone activities. Protein Sci. 8, 905–912 (1999).
Savkur, R. S. & Olson, M. O. Preferential cleavage in pre-ribosomal RNA by protein B23 endoribonuclease. Nucleic Acids Res. 26, 4508–4515 (1998).
Umekawa, H. et al. The carboxyl terminal sequence of nucleolar protein B23.1 is important in its DNA polymerase α-stimulatory activity. J. Biochem. 130, 199–205 (2001).
Borer, R. A., Lehner, C. F., Eppenberger, H. M. & Nigg, E. A. Major nucleolar proteins shuttle between nucleus and cytoplasm. Cell 56, 379–390 (1989).
Tarapore, P. et al. Thr199 phosphorylation targets nucleophosmin to nuclear speckles and represses pre-mRNA processing. FEBS Lett. 580, 399–409 (2006).
Palaniswamy, V., Moraes, K. C., Wilusz, C. J. & Wilusz, J. Nucleophosmin is selectively deposited on mRNA during polyadenylation. Nature Struct. Mol. Biol. 13, 429–435 (2006).
Grisendi, S., Mecucci, C., Falini, B. & Pandolfi, P. P. Nucleophosmin and cancer. Nature Rev. Cancer 6, 493–505 (2006).
Kondo, T. et al. Identification and characterization of nucleophosmin/B23/numatrin which binds the anti-oncogenic transcription factor IRF-1 and manifests oncogenic activity. Oncogene 15, 1275–1281 (1997).
Grisendi, S. et al. Role of nucleophosmin in embryonic development and tumorigenesis. Nature 437, 147–153 (2005). The first demonstration of the role of nucleophosmin in the control of numeral integrity of centrosomes and genomic instability in vivo.
Shinmura, K., Tarapore, P. Tokuyama, Y., George, K. R. & Fukasawa, K. Characterization of centrosomal association of Nucleophosmin/B23 linked to Crm1 activity. FEBS Lett. 579, 6621–6634 (2005).
Wang, W., Budhu, A., Forgues, M. & Wang X. W. Temporal and spatial control of nucleophosmin by the Ran-Crm1 complex in centrosome duplication. Nature Cell Biol. 7, 823–830 (2005).
Nishimura, Y., Itoh, K., Yoshioka, K., Tokuda. K. & Himeno, M. Overexpression of ROCK in human breast cancer cells: evidence that ROCK activity mediates intracellular membrane traffic of lysosomes. Pathol. Oncol. Res. 9, 83–95 (2003).
Kamai, T. et al. Significant association of Rho/ROCK pathway with invasion and metastasis of bladder cancer. Clin. Cancer Res. 9, 2632–2641 (2003).
Ma, Z. et al. Interaction between ROCK II and nucleophosmin/B23 in the regulation of centrosome duplication. Mol. Cell Biol. 26, 9016–9034 (2006).
Chevrier, V. et al. The Rho-associated protein kinase p160ROCK is required for centrosome positioning. J. Cell Biol. 157, 807–817. (2002).
Yoneda, A., Multhaupt, A. B. & Couchman, J. R. The Rho kinases I and II regulate different aspects of myosin II activity. J. Cell Biol. 170, 443–553 (2005).
Fisk, H. A. & Winey, A. The mouse Mps1p-like kinase regulates centrosome duplication. Cell 106, 95–104 (2001).
Fisk, H. A., Mattison, C. P. & Winey, M. Human Mps1 protein kinase is required for centrosome duplication and normal mitotic progression. Proc. Natl Acad. Sci. USA 100, 14875–14880 (2003).
Kanai, M. et al. Physical and functional interaction between mortalin and Mps1 kinase. Genes Cells 12, 797–810 (2007).
Czarnecka, A. M., Campanella, C., Zummo, G. & Cappello, F. Mitochondrial chaperones in cancer: from molecular biology to clinical diagnostics. Cancer Biol. Ther. 5, 714–720 (2006).
Meraldi, P., Lukas, J., Fry, A. M., Bartek, J. & Nigg, E. A. Centrosome duplication in mammalian somatic cells requires E2F and Cdk2-cyclin A. Nature Cell Biol. 1, 88–93 (1999).
Tokuyama, Y., Horn, H. F., Kawamura, K., Tarapore, P. & Fukasawa, K. Specific phosphorylation of nucleophosmin on Thr(199) by cyclin-dependent kinase 2–cyclin E and its role in centrosome duplication. J. Biol. Chem. 276, 21529–21537 (2001).
Reed, S. I. Control of the G1/S transition. Cancer Surv. 29, 7–23 (1997).
Duensing, A. et al. RNA polymerase II transcription is required for human papillomavirus type 16 E7- and hydroxyurea-induced centriole overduplication. Oncogene 26, 215–223 (2007).
Ussar, S. & Voss, T. MEK1 and MEK2, different regulators of the G1/S transition. J. Biol. Chem. 279, 43861–43869 (2004).
Nelsen, C. J. et al. Short term cyclin D1 overexpression induces centrosome amplification, mitotic spindle abnormalities, and aneuploidy. J. Biol. Chem. 280, 768–776 (2005).
Giacinti, C. & Giordano, A. RB and cell cycle progression. Oncogene 25, 5220–5227 (2006).
Duensing, S. et al. The human papillomavirus type 16 E6 and E7 oncoproteins cooperate to induce mitotic defects and genomic instability by uncoupling centrosome duplication from the cell division cycle. Proc. Natl Acad. Sci. USA 97, 10002–10007 (2000).
Balsitis, S. J. et al. Recapitulation of the effects of the human papillomavirus type 16 E7 oncogene on mouse epithelium by somatic Rb deletion and detection of pRb-independent effects of E7 in vivo. Mol. Cell Biol. 23, 9094–9103 (2003).
Iovino, F., Lentini, L., Amato, A. & Di Leonardo, A. RB acute loss induces centrosome amplification and aneuploidy in murine primary fibroblasts. Mol. Cancer 5, 8 (2006).
Ortega, S., Malumbres, M. & Barbacid, M. Cyclin D-dependent kinases, INK4 inhibitors and cancer. Biochim. Biophys. Acta 1602, 73–87 (2002).
Rudolph, J. Inhibiting transient protein–protein interactions: lessons from the Cdc25 protein tyrosine phosphatases. Nature Rev. Cancer 7, 202–211 (2007).
Boutros, R., Lobjois, V. & Ducommun, B. CDC25 phosphatases in cancer cells: key players? Good targets? Nature Rev. Cancer 7, 495–507 (2007).
Dutertre, S. et al. Phosphorylation of CDC25B by Aurora-A at the centrosome contributes to the G2-M transition. J. Cell Sci. 117, 2523–2531 (2004).
Balint, E. E. & Vousden, K. H. Activation and activities of the p53 tumour suppressor protein. Br. J. Cancer 85, 1813–1823 (2001).
Harper, J. W. Cyclin dependent kinase inhibitors. Cancer Surv. 29, 91–107 (1997).
Fukasawa, K., Choi, T, Kuriyama, R, Rulong, S. & Vande Woude, G. F. Abnormal centrosome amplification in the absence of p53. Science 271, 1744–1747 (1996). The first demonstration of the disturbance of the numeral integrity of centrosomes and the consequential destabilization of chromosomes by loss of p53.
Fukasawa, K., Wiener, F., Vande Woude, G. F. & Mai, S. Genomic instability and apoptosis are frequent in p53 deficient mice. Oncogene 15, 1295–1302 (1997).
Tarapore, P., Horn, H. F., Tokuyama, Y. & Fukasawa, K. Direct regulation of the centrosome duplication cycle by the p53–p21 (Waf1/Cip1) pathway. Oncogene 20, 3173–3184 (2001).
Sherr, C. J. Divorcing ARF and p53: an unsettled case. Nature Rev. Cancer 6, 663–673 (2006).
Blair Zajdel, M. E. & Blair, G. E. The intracellular distribution of the transformation-associated protein p53 in adenovirus-transformed rodent cells. Oncogene 2, 579–584 (1988).
Brown, C. R., Doxsey, S. J., White, E. & Welch, W. J. Both viral (adenovirus E1B) and cellular (hsp 70, p53) components interact with centrosomes. J. Cell Physiol. 160, 47–60 (1994).
Morris, V. B., Brammall, J., Noble, J. & Reddel, R. p53 localizes to the centrosomes and spindles of mitotic cells in the embryonic chick epiblast, human cell lines, and a human primary culture: an immunofluorescence study. Exp. Cell Res. 256, 122–130 (2000).
Tarapore, P., Tokuyama, Y., Horn, H. F. & Fukasawa, K. Difference in the centrosome duplication regulatory activity among p53 'hot spot' mutants: potential role of Ser 315 phosphorylation-dependent centrosome binding of p53. Oncogene 20, 6851–6863 (2001).
Tritarelli, A. et al. p53 localization at centrosomes during mitosis and postmitotic checkpoint are ATM-dependent and require serine 15 phosphorylation. Mol. Biol. Cell 15, 3751–3757 (2004).
Shinmura, K., Bennett, R. A., Tarapore, P. & Fukasawa, K. Direct evidence for the role of centrosomally localized p53 in the regulation of centrosome duplication. Oncogene 26, 2939–2944 (2007). This study provides evidence for the role of centrosomally localized p53 in the regulation of centrosome duplication, independent of its transactivation function.
Vargas, D. A., Takahashi, S. & Ronai, Z. Mdm2: a regulator of cell growth and death. Adv. Cancer Res. 89, 1–34 (2003).
Carroll, P. E. et al. Centrosome hyperamplification in human cancer: chromosome instability induced by p53 mutation and/or Mdm2 overexpression. Oncogene 18, 1935–1944 (1999).
Wolf, G. et al. Prognostic significance of polo-like kinase (PLK) expression in non-small cell lung cancer. Oncogene 14, 543–549 (1997).
Knecht, R. et al. Prognostic significance of polo-like kinase (PLK) expression in squamous cell carcinomas of the head and neck. Cancer Res. 59, 2794–2797 (1999).
Macmillan, J. C., Hudson, J. W., Bull, S., Dennis, J. W. & Swallow, C. J. Comparative expression of the mitotic regulators SAK and PLK in colorectal cancer. Ann. Surg. Oncol. 8, 729–740 (2001).
Li, J. J. & Li, S. A. Mitotic kinases: the key to duplication, segregation, and cytokinesis errors, chromosomal instability, and oncogenesis. Pharmacol. Ther. 111, 974–984 (2006).
Warnke, S. et al. Polo-like kinase-2 is required for centriole duplication in mammalian cells. Curr. Biol. 14, 1200–1207 (2004).
Habedanck, R., Stierhof, Y. D., Wilkinson, C. J. & Nigg, E. A. The Polo kinase Plk4 functions in centriole duplication. Nature Cell Biol. 7, 1140–1146 (2005).
Gopalan, G., Chan, C. S. M. & Donovan, P. J. A novel mammalian, mitotic spindle-associated kinase is related to yeast and fly chromosome segregation regulators. J. Cell Biol. 138, 643–656 (1997).
Zhou, H. et al. Tumour amplified kinase STK15/BTAK induces centrosome amplification, aneuploidy and transformation. Nature Genet. 20, 189–193 (1998).
Katayama, H. et al. Phosphorylation by aurora kinase A induces Mdm2-mediated destabilization and inhibition of p53. Nature Genet. 36, 55–62 (2004). This study provides an insight into the molecular mechanism underlying the regulation of centrosome duplication by Aurora A by modulation of the stability of p53.
Thacker, J. The RAD51 gene family, genetic instability and cancer. Cancer Lett. 219, 125–135 (2005).
Venkitaraman, A. R. Cancer susceptibility and the functions of BRCA1 and BRCA2. Cell 108, 171–182 (2002).
Hsu, L. C. & White, R. L. BRCA1 is associated with the centrosome during mitosis. Proc. Natl Acad. Sci. USA, 95, 12983–12988 (1998).
Xu, X. et al. Centrosome amplification and a defective G2–M cell cycle checkpoint induce genetic instability in BRCA1 exon 11 isoform-deficient cells. Mol. Cell, 3, 389–395 (1999).
Starita, L. M. et al. BRCA1-dependent ubiquitination of γ-tubulin regulates centrosome number. Mol. Cell Biol. 24, 8457–8466 (2004).
Okada, S. & Ouchi, T. Cell cycle differences in DNA damage-induced BRCA1 phosphorylation affect its subcellular localization. J. Biol. Chem. 17, 2015–2020 (2003). Reference 81 and 84 provide the initial demonstration of the involvement of BRCA1 in the regulation of numeral integrity of centrosomes.
Lotti, L. V. et al. Subcellular localization of the BRCA1 gene product in mitotic cells. Genes Chromosomes Cancer 35, 193–203 (2002).
Maul, G. G., Jensen, D. E., Ishov, A. M., Herlyn, M. & Rauscher, F. J. Nuclear redistribution of BRCA1 during viral infection. Cell Growth Differ. 9, 743–755 (1998).
Hut, H. M. et al. Dysfunctional BRCA1 is only indirectly linked to multiple centrosomes. Oncogene 24, 7619–7623 (2005).
Wilson, C. A. et al. Differential subcellular localization, expression and biological toxicity of BRCA1 and the splice variant BRCA1-Δ11b. Oncogene 14, 1–16 (1997).
Tutt, A. et al. Absence of Brca2 causes genome instability by chromosome breakage and loss associated with centrosome amplification. Curr. Biol. 9, 1107–1110 (1999).
Sung, P., Krejci, L., Van Komen, S. & Sehorn, M. G. Rad51 recombinase and recombination mediators. J. Biol. Chem. 278, 42729–42732 (2003).
Bertrand, P., Lambert, S., Joubert, C. & Lopez, B. S. Overexpression of mammalian Rad51 does not stimulate tumorigenesis while a dominant-negative Rad51 affects centrosome fragmentation, ploidy and stimulates tumorigenesis, in p53-defective CHO cells. Oncogene 22, 7587–7592 (2003).
Griffin, C. S., Simpson, P. J., Wilson, C. R. & Thacker, J. Mammalian recombination-repair genes XRCC2 and XRCC3 promote correct chromosome segregation. Nature Cell Biol. 2, 757–761 (2000).
Smiraldo, P. G., Gruver, A. M., Osborn, J. C. & Pittman, D. L. Extensive chromosomal instability in Rad51d-deficient mouse cells. Cancer Res. 65, 2089–2096 (2005).
Date, O. et al. Haploinsufficiency of RAD51B causes centrosome fragmentation and aneuploidy in human cells. Cancer Res. 66, 6018–6024 (2006).
Renglin Lindh, A., Schultz, N., Saleh-Gohari, N. & Helleday, T. RAD51C (RAD51L2) is involved in maintaining centrosome number in mitosis. Cytogenet. Genome Res. 116, 38–45 (2007).
Dodson, H. et al. Centrosome amplification induced by DNA damage occurs during a prolonged G2 phase and involves ATM. EMBO J. 23, 3864–3873 (2004). This study provides the first mechanistic insight of how centrosome amplification occurs in cells inflicted with DNA damage.
Sugihara, E. et al. Suppression of centrosome amplification after DNA damage depends on p27 accumulation. Cancer Res. 66, 4020–4029 (2006).
Sato, N., Mizumoto, K., Nakamura, M. & Tanaka, M. Radiation-induced centrosome overduplication and multiple mitotic spindles in human tumor cells. Exp. Cell Res. 255, 321–326 (2000).
D'Assoro, A. B. et al. Genotoxic stress leads to centrosome amplification in breast cancer cell lines that have an inactive G1/S cell cycle checkpoint. Oncogene 23, 4068–4075 (2004).
Kawamura, K. et al. Induction of centrosome amplification in p53 siRNA-treated human fibroblast cells by radiation exposure. Cancer Sci. 97, 252–258 (2006).
Taylor, W. R. & Stark, G. R. Regulation of the G2/M transition by p53. Oncogene 20, 1803–1815 (2001).
Nurse, P. Universal control mechanism regulating onset of M-phase. Nature 344, 503–508 (1990).
Charrier-Savournin, F. B. et al. p21-Mediated nuclear retention of cyclin B1-Cdk1 in response to genotoxic stress. Mol. Biol. Cell 15, 3965–3976 (2004).
Tang, J., Erikson, R. L. & Liu, X. Checkpoint kinase 1 (Chk1) is required for mitotic progression through negative regulation of polo-like kinase 1 (Plk1). Proc. Natl Acad. Sci. USA 103, 11964–11969 (2006).
Roshak, A. K. et al. The human polo-like kinase, PLK, regulates cdc2/cyclin B through phosphorylation and activation of the cdc25C phosphatase. Cell Signal. 12, 405–411 (2000).
Lee, J., Kumagai, A. & Dunphy, W. G. Positive regulation of wee1 by chk1 and 14-3-3 proteins. Mol. Biol. Cell 12, 551–563 (2001).
Jackman, M., Lindon, C., Nigg, E. A. & Pines, J. Active cyclin B1–Cdk1 first appears on centrosomes in prophase. Nature Cell Biol. 5, 143–148 (2003).
Hirota, T. et al. Aurora-A and an interacting activator, the LIM protein Ajuba, are required for mitotic commitment in human cells. Cell 114, 585–598 (2003).
Kramer, A. et al. Centrosome-associated Chk1 prevents premature activation of cyclin-B–Cdk1 kinase. Nature Cell Biol. 6, 884–891 (2004).
Smith, S. & de Lange, T. Cell cycle dependent localization of the telomeric PARP, tankyrase, to nuclear pore complexes and centrosomes. J. Cell Sci. 112, 3649–3656 (1999).
Kanai, M. et al. Involvement of poly(ADP-Ribose) polymerase 1 and poly(ADP-Ribosyl)ation in regulation of centrosome function. Mol. Cell Biol. 23, 2451–2462 (2003). References 107, 110 and 111 reveal a new and important aspect of centrosomes as the site of the initial activation of CDK1–cyclin B by Aurora A and Chk1 kinases.
Augustin, A. et al. PARP-3 localizes preferentially to the daughter centriole and interferes with the G1/S cell cycle progression. J. Cell Sci. 116, 1551–1562 (2003).
Chang, P., Coughlin, M. & Mitchison, T. J. Tankyrase-1 polymerization of poly(ADP-ribose) is required for spindle structure and function. Nature Cell Biol. 7, 1133–1139 (2005).
Moroianu, J. Nuclear import and export pathways. J. Cell Biochem. 75 (Suppl. 32–33), 76–83 (1999).
Harel, A. & Forbes, D. J. Importin β: conducting a much larger cellular symphony. Mol. Cell 16, 319–330 (2004).
Dasso, M. The Ran GTPase: theme and variations. Curr. Biol. 12, 502–508 (2002).
Forgues, M. et al. Involvement of Crm1 in hepatitis B virus X protein-induced aberrant centriole replication and abnormal mitotic spindles. Mol. Cell Biol. 23, 5282–5292 (2003).
Gruss, O. J. & Vernos, I. The mechanism of spindle assembly: functions of Ran and its target TPX2. J. Cell Biol. 166, 949–955 (2004).
Ciciarello, M. et al. Importin β is transported to spindle poles during mitosis and regulates Ran-dependent spindle assembly factors in mammalian cells. J. Cell Sci. 117, 6511–6522 (2004).
Di Fiore, B. et al. Mammalian RanBP1 regulates centrosome cohesion during mitosis. J. Cell Sci. 116, 3399–3411 (2003).
Joukov, V. et al. The BRCA1/BARD1 heterodimer modulates Ran-dependent mitotic spindle assembly. Cell 127, 539–552 (2006).
Kufer, T. A. et al. A. human TPX2 is required for targeting Aurora-A kinase to the spindle. J. Cell Biol. 158, 617–623 (2002).
Tsai, M. Y. et al. A Ran signalling pathway mediated by the mitotic kinase Aurora A in spindle assembly. Nature Cell Biol. 5, 242–248 (2003).
Eyers, P. A., Erikson, E., Chen, L. G. & Maller, J. L. A novel mechanism for activation of the protein kinase Aurora A. Curr. Biol. 13, 691–697 (2003).
Ouchi, M. et al. BRCA1 phosphorylation by Aurora-A in the regulation of G2 to M transition. J. Biol. Chem. 279, 19643–19648 (2004).
Mayor, T., Stierhof, Y. D., Tanaka, K., Fry, A. M. & Nigg, E. A. The centrosomal protein C-Nap1 is required for cell cycle-regulated centrosome cohesion. J. Cell Biol. 151, 837–846 (2000).
Bahe, S., Stierhof, Y. D., Wilkinson, C. J., Leiss, F. & Nigg, E. A. Rootletin forms centriole-associated filaments and functions in centrosome cohesion. J. Cell Biol. 171, 27–33 (2005).
Hayward, D. G. & Fry, A. M. Nek2 kinase in chromosome instability and cancer. Cancer Lett. 237, 155–166 (2006).
Fry, A. M. et al. C-Nap1, a novel centrosomal coiled-coil protein and candidate substrate of the cell cycle-regulated protein kinase Nek2. J. Cell Biol. 141, 1563–1574 (1998).
Faragher, A. J. & Fry, A. M. Nek2A kinase stimulates centrosome disjunction and is required for formation of bipolar mitotic spindles. Mol. Biol. Cell 14, 2876–2889 (2003).
Helps, N. R., Luo, X., Barker, H. M. & Cohen, P. T. NIMA-related kinase 2 (Nek2), a cell-cycle-regulated protein kinase localized to centrosomes, is complexed to protein phosphatase 1. Biochem. J. 349, 509–518 (2000).
Mi, J., Guo, C., Brautigan, D. L. & Larner, J. M. Protein phosphatase-1α regulates centrosome splitting through Nek2. Cancer Res. 67, 1082–1089 (2007). References 126, 129 and 132 reveal the molecular mechanism underlying centrosome separation at late G2 phase.
Meraldi, P. & Nigg, E. A. Centrosome cohesion is regulated by a balance of kinase and phosphatase activities. J. Cell Sci. 114, 3749–3757 (2001).
Dohadwala, M. et al. Phosphorylation and inactivation of protein phosphatase 1 by cyclin-dependent kinases. Proc. Natl Acad. Sci. USA 91, 6408–6412 (1994).
Kashina, A. S., Rogers, G. C. & Scholey, J. M. The bimC family of kinesins: essential bipolar mitotic motors driving centrosome separation. Biochim. Biophys. Acta. 1357, 257–271 (1997).
Blangy, A. et al. Phosphorylation by p34cdc2 regulates spindle association of human Eg5, a kinesin-related motor essential for bipolar spindle formation in vivo. Cell 83, 1159–1169 (1995).
Glover, D. M., Leibowitz, M. H., McLean, D. A. & Parry, H. Mutations in aurora prevent centrosome separation leading to the formation of monopolar spindles. Cell 81, 95–105 (1995).
Roghi, C. et al. The Xenopus protein kinase pEg2 associates with the centrosome in a cell cycle-dependent manner, binds to the spindle microtubules and is involved in bipolar mitotic spindle assembly. J. Cell Sci. 111, 557–572 (1998).
Giet, R., Uzbekov, R., Cubizolles, F., Le Guellec, K. & Prigent, C. The Xenopus laevis aurora-related protein kinase pEg2 associates with and phosphorylates the kinesin-related protein XlEg5. J. Biol. Chem. 274, 15005–15013 (1999).
Katayama, H., Zhou, H., Li, Q., Tatsuka, M. & Sen, S. Interaction and feedback regulation between STK15/BTAK/Aurora-A kinase and protein phosphatase 1 through mitotic cell division cycle. J. Biol. Chem. 276, 46219–46224 (2001).
Raff, J. W. Centrosomes and cancer: lessons from a TACC. Trends Cell Biol. 12, 222–225 (2002).
Giet, R. et al. Drosophila Aurora A kinase is required to localize D-TACC to centrosomes and to regulate astral microtubules. J. Cell Biol. 156, 437–451 (2002).
Kinoshita, K., Habermann, B. & Hyman, A. A. XMAP215: a key component of the dynamic microtubule cytoskeleton. Trends Cell Biol. 12, 267–273 (2002).
Toji, S. et al. The centrosomal protein Lats2 is a phosphorylation target of Aurora-A kinase. Genes Cells 9, 383–397 (2004).
McPherson, J. P. et al. Lats2/Kpm is required for embryonic development, proliferation control and genomic integrity, EMBO J. 23, 3677–3688 (2004).
Yabuta, N. et al. Lats2 is an essential mitotic regulator required for the coordination of cell division. J. Biol. Chem. 282, 19259–19271 (2007).
Abe, Y., Ohsugi, M., Haraguchi, K., Fujimoto, J. & Yamamoto, T. LATS2–Ajuba complex regulates γ-tubulin recruitment to centrosomes and spindle organization during mitosis. FEBS Lett. 580, 782–788 (2006).
Kumar, R., Gururaj, A. E. & Banes, C. J. p21-activated kinases in cancer. Nature Rev. Cancer 6, 459–471 (2006).
Zhao, Z. S., Lim, J. P., Ng, Y. W., Lim, L. & Manser, E. The GIT-associated kinase PAK targets to the centrosome and regulates Aurora-A. Mol. Cell 20, 237–249 (2005).
Barr, F. A., Sillje, H. H. W. & Nigg, E. A. Polo-like kinases and the orchestration of cell division. Nature Rev. Mol. Cell Biol. 5, 429–440 (2004).
Casenghi, M. et al. Polo-like kinase 1 regulates Nlp, a centrosome protein involved in microtubule nucleation. Dev. Cell 5, 113–125 (2003).
Rapley, J. et al. Coordinate regulation of the mother centriole component nlp by nek2 and plk1 protein kinases. Mol. Cell Biol. 25, 1309–1324 (2005).
McNally, K. P., Buster, D. & McNally, F. J. Katanin-mediated microtubule severing can be regulated by multiple mechanisms. Cell Motil. Cytoskeleton 53, 337–349 (2002).
Budde, P. P., Kumagai, A., Dunphy, W. G. & Heald, R. Regulation of Op18 during spindle assembly in Xenopus egg extracts. J. Cell Biol. 153, 149–158 (2001).
Yarm, F. R. Plk phosphorylation regulates the microtubule-stabilizing protein TCTP. Mol. Cell Biol. 22, 6209–6221 (2002).
do Carmo Avides, M., Tavares, A. & Glover, D. M. Polo kinase and Asp are needed to promote the mitotic organizing activity of centrosomes. Nature Cell Biol. 3, 421–424 (2001).
Oshimori, N., Ohsugi, M. & Yamamoto, T. The Plk1 target Kizuna stabilizes mitotic centrosomes to ensure spindle bipolarity. Nature Cell Biol. 8, 1095–1101 (2006). This study identifies the target protein of PLK1, Kizuna, which has a crucial role in the maintenance of the structural integrity of centrosomes during mitosis, suggesting a new and important method of regulation of centrosomes and chromosome stability.
Simizu, S. & Osada, H. Mutations in the Plk gene lead to instability of Plk protein in human tumour cell lines. Nature Cell Biol. 2, 852–854 (2000).
Musacchio, A. & Hardwick, K. G. The spindle checkpoint: structural insights into dynamic signaling. Nature Rev. Mol. Cell Biol. 3, 731–741 (2002).
Yu, H. Regulation of APC-Cdc20 by the spindle checkpoint. Curr. Opin. Cell Biol. 14, 706–714 (2002).
Oikawa, T. et al. Transcriptional control of BubR1 by p53 and suppression of centrosome amplification by BubR1. Mol. Cell Biol. 25, 4046–4061 (2005).
Bornens, M. Centrosome composition and microtubule anchoring mechanisms. Curr. Opin. Cell Biol. 14, 25–34 (2002).
Moritz, M. & Agard, D. A. γ-Tubulin complexes and microtubule nucleation. Curr. Opin. Struct. Biol. 11, 174–181 (2001).
Wiese, C. & Zheng, Y. γ-Tubulin complexes and their interaction with microtubule-organizing centers. Curr. Opin. Struct. Biol. 9, 250–259 (1999).
Delgehyr, N., Sillibourne, J. & Bornens, M. Microtubule nucleation and anchoring at the centrosome are independent processes linked by ninein function. J. Cell Sci. 118, 1565–1575 (2005). This study proposes the distinction between the microtubule nucleation and anchoring activities of centrosomes.
Lane, H. A. & Nigg, E. A. Antibody microinjection reveals an essential role for human polo-like kinase 1 (Plk1) in the functional maturation of mitotic centrosomes. J. Cell Biol. 135, 1701–1713 (1996).
Acknowledgements
I apologize for not being able to cite many important studies owing to space limitations. Preparation of this article is supported in part by grants from the National Institutes of Health (Bethesda, Maryland, USA).
Author information
Authors and Affiliations
Supplementary information
Supplementary information S1 (Table)
A complete list of cancer-associated proteins which are involved in the control numeral integrity of centrosomes, centrosome duplication, and centrosome function/behavior. (PDF 375 kb)
Glossary
- Pericentriolar material
-
(PCM). Aggregates of many different proteins surrounding the paired centrioles. The protein composition of PCM is dynamic: some proteins are permanent residents of the centrosome, whereas some localize to the centrosome in a manner that is specific to the cell-cycle stage.
- Mitotic spindle
-
The term commonly and synonymously used for describing the dynamic mitotic spindle apparatus.
- Aneuploid
-
Having a change in the number of chromosomes. It should be distinguished from polyploid (presence of more than two homologous sets of chromosomes).
- Centrosome amplification
-
The generation of more than two centrosomes, primarily resulting from deregulated centrosome duplication — duplication of centrosomes more than once in a single cell cycle.
- Checkpoint
-
The control mechanisms that ensure the fidelity of cell division in eukaryotic cells by monitoring whether the processes at each phase of the cell cycle have been accurately completed before progression into the next phase.
- Polyploid
-
Having more than two homologous sets of chromosomes (for example, triploid (three sets), tetraploid (four sets)).
- Centrosome clustering
-
The phenomenon of amplified centrosomes clustering at two opposing poles, in which dynein, a multi-subunit microtubule-based motor protein that moves towards the minus ends of microtubules, has been shown to have a role.
- Centrosome maturation
-
A process taking place in S and G2 phases that establishes the microtubule nucleation or anchoring activities of centrosomes required for the formation of mitotic spindles by recruiting critical pericentriolar material components.
- Mitotic catastrophe
-
A form of (apoptotic) cell death resulting from abnormal mitosis due to DNA damage.
- Hydroxyurea
-
A potent ribonucleotide reductase inhibitor, hydroxyurea exposure results in deprivation of dNTP, leading to inhibition of S-phase entry and S-phase progression.
- γ-Tubulin
-
A major centrosomal protein that functions in the microtubule nucleation or anchoring at the centrosome through forming a protein complex with several other proteins, known as γ-tubulin ring complex (γ-TuRC). γ-TuRC is believed to function as a template for microtubules to grow.
- Poly(ADP-ribosyl)ation
-
One of the major post-translational modifications of proteins, catalysed by the poly(ADP-ribose) polymerase family of proteins. The attachment of ADP-ribose polymers adds a high negative charge, affecting the activities of the target proteins.
- Kinesin
-
A family of microtubule-based motor proteins that move towards the plus ends of microtubules.
Rights and permissions
About this article
Cite this article
Fukasawa, K. Oncogenes and tumour suppressors take on centrosomes. Nat Rev Cancer 7, 911–924 (2007). https://doi.org/10.1038/nrc2249
Issue Date:
DOI: https://doi.org/10.1038/nrc2249
This article is cited by
-
The Nek2 centrosome-mitotic kinase contributes to the mesenchymal state, cell invasion, and migration of triple-negative breast cancer cells
Scientific Reports (2021)
-
Acquired resistance to combined BET and CDK4/6 inhibition in triple-negative breast cancer
Nature Communications (2020)
-
Centrosome dysfunction: a link between senescence and tumor immunity
Signal Transduction and Targeted Therapy (2020)
-
Functional genetic variants in centrosome-related genes CEP72 and YWHAG confer susceptibility to gastric cancer
Archives of Toxicology (2020)
-
p53 mitotic centrosome localization preserves centrosome integrity and works as sensor for the mitotic surveillance pathway
Cell Death & Disease (2019)