Abstract
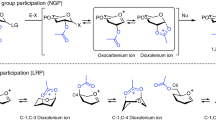
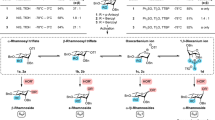
Glycosyl cations are universally accepted key ionic intermediates in the mechanism of glycosylation, the reaction that covalently links carbohydrates to other molecules. These ions have remained hypothetical species so far because of their extremely short life in organic media as a consequence of their very high reactivity. Here, we report the use of liquid hydrofluoric acid–antimony pentafluoride (HF/SbF5) superacid to generate and stabilize the glycosyl cations derived from peracetylated 2-deoxy and 2-bromoglucopyranose in a condensed phase. Their persistence in this superacid medium allows their three-dimensional structure to be studied by NMR, aided by complementary computations. Their deuteration further confirms the impact of the structure of the glycosyl cation on the stereochemical outcome of its trapping.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Transforming Glycoscience: A Roadmap for the Future (The National Academies Press, 2012).
Boltje, T. J., Buskas, T. & Boons, G. J. Opportunities and challenges in synthetic oligosaccharide and glycoconjugate research. Nature Chem. 1, 611–622 (2009).
Demchenko, A. V. Handbook of Chemical Glycosylation: Advances in Stereoselectivity and Therapeutic Relevance (Wiley, 2008).
Zhu, X. & Schmidt, R. R. New principles for glycosidic bond formation. Angew. Chem. Int. Ed. 48, 1900–1934 (2009).
Michael, A. On the synthesis of helicin and phenol glucoside. Am. Chem. J. 1, 305–312 (1879).
Fischer, E. Ueber die glucoside der alkohole. Ber. Deutsch Chem. Ges. 26, 2400–2412 (1893).
Lemieux, R. U., Hendriks, K. B., Stick, R. V. & James, K. Halide ion catalyzed glycosylation reactions. Syntheses of α-linked disaccharides. J. Am. Chem. Soc. 97, 4056–4062 (1975).
Ayala, L., Lucero, C. G., Romero, J. A. C., Tabacco, S. A. & Woerpel, K. A. Stereochemistry of nucleophilic substitution reactions depending upon substituent: evidence for electrostatic stabilization of pseudoaxial conformers of oxocarbenium ions by heteroatom substituents. J. Am. Chem. Soc. 125, 15521–15528 (2003).
Frihed, T. G., Bols, M. & Pedersen, C. M. Mechanisms of glycosylation reactions studied by low-temperature nuclear magnetic resonance. Chem. Rev. 115, 4963–5013 (2015).
Saito, K. et al. Indirect cation-flow method flash generation of alkoxycarbenium ions and studies on the stability of glycosyl cations. Angew. Chem. Int. Ed. 50, 5153–5156 (2011).
Whitfield, D. M. Computational studies of the role of the glycopyranosyl oxacarbenium ions in glycobiology and glycochemistry. Adv. Carbohydr. Chem. Biochem. 62, 83–159 (2009).
Denekamp, C. & Sandlers, Y. Anomeric distinction and oxonium ion formation in acetylated glycosides. J. Mass Spectrom. 40, 765–771 (2005).
Huang, M. et al. Dissecting the mechanisms of a class of chemical glycosylation using primary 13C kinetic isotope effects. Nature Chem. 4, 663–667 (2012).
Huang, M., Retailleau, P., Bohé, L. & Crich, D. Cation clock permits distinction between the mechanisms of α- and β-O- and β-C-glycosylation in the mannopyranose series: evidence for the existence of a mannopyranosyl oxocarbenium ion. J. Am. Chem. Soc. 134, 14746–14749 (2012).
Bohé, L. & Crich, D. A propos of glycosyl cations and the mechanism of chemical glycosylation. C.R. Chimie 14, 3–16 (2011).
Bohé, L. & Crich, D. A propos of glycosyl cations and the mechanism of chemical glycosylation the current state of the art. Carbohydr. Res. 403, 48–59 (2015).
Amyes, T. L. & Jencks, W. P. Lifetimes of oxocarbenium ions in aqueous solution from common ion inhibition of the solvolysis of α-azido ethers by added azide ion. J. Am. Chem. Soc. 111, 7888–7900 (1989).
Olah, G. A., Prakash, G. K. S., Molnar, A. & Sommer, J. Superacid Chemistry (Wiley, 2009).
Olah, G. A. et al. Stable carbonium ions. Alkylcarbonium hexafluoroantimonates. J. Am. Chem. Soc. 86, 1360–1373 (1964).
Olah, G. A. & Bollinger, J. M. Stable carbonium ions. Primary alkoxycarbonium ions. J. Am. Chem. Soc. 89, 2993–2996 (1967).
Akien, G. R. & Subramaniam, B. In 247th ACS National Meeting and Exhibition, paper CARB-96 (2014).
Matsumoto, K., Ueoka, K., Suzuki, S., Suga, S. & Yoshida, J. Direct and indirect electrochemical generation of alkoxycarbenium ion pools from thioacetals. Tetrahedron 65, 10901–10907 (2009).
Olah, G. A. et al. 17O and 13C NMR/ab initio/IGLO/GIAO-MP2 study of oxonium and carboxonium ions (dications) and comparison with experimental data. J. Am. Chem. Soc. 119, 8035–8042 (1997).
Olah, G. A., O'Brien, D. H. & White, A. M. Stable carbonium ions. LII. Protonated esters and their cleavage in fluorosulfonic acid–antimony pentafluoride solution. J. Am. Chem. Soc. 89, 5694–5700 (1967).
Lemieux, R. U. Some implications in carbohydrate chemistry of theories relating to mechanisms of replacement reactions. Adv. Carbohydr. Chem. 9, 1–57 (1954).
Zeng, Y., Wang, Z., Whitfield, D. & Huang, X. Installation of electron-donating protective groups, a strategy for glycosylating unreactive thioglycosyl acceptors using the preactivation-based glycosylation method. J. Org. Chem. 73, 7952–7962 (2008).
Hodosi, G. & Krepinsky, J. J. Polymer-supported solution synthesis of oligosaccharides: probing glycosylations of MPEG-DOX-OH with 2-acetamidoglycopyranosyl derivatives. Synlett 159–162 (1996).
Mertens, A., Lammertsma, K., Arvanaghi, M. & Olah, G. A. Onium ions. 26. Aminodiazonium ions: preparation, 1H, 13C, and 15N NMR structural studies, and electrophilic amination of aromatics. J. Am. Chem. Soc. 105, 5657–5660 (1983).
Hou, D. & Lowary, T. L. Recent advances in the synthesis of 2-deoxy-glycosides. Carbohydr. Res. 344, 1911–1940 (2009).
Bucher, C. & Gilmour, R. Fluorine-directed glycosylation. Angew. Chem. Int. Ed. 49, 8724–8728 (2010).
Woods, R. J., Andrews, C. W. & Bowen, J. P. Molecular mechanical investigations of the properties of oxocarbenium ions. II. Application to glycoside hydrolysis. J. Am. Chem. Soc. 114, 859–864 (1992).
Miljkovic, M., Yeagley, D., Deslongchamps, P. & Dory, Y. L. Experimental and theoretical evidence of through-space electrostatic stabilization of the incipient oxocarbenium ion by an axially oriented electronegative substituent during glycopyranoside acetolysis. J. Org. Chem. 62, 7597–7604 (1997).
Olah, G. A., Laali, K. K., Wang, Q. & Prakash, G. K. S. Onium Ions (Wiley, 1998).
Walwoort, M. T. et al. The impact of oxacarbenium ion conformers on the stereochemical outcome of glycosylations. Carbohydr. Res. 345, 1252–1263 (2010).
Nukada, T., Bérces, A. & Withfield, D. M. Can the stereochemical outcome of glycosylation reactions be controlled by the conformational preferences of the glycosyl donor? Carbohydr. Res. 337, 765–774 (2002).
Olah, G. A. & Klumpp, D. Superelectrophiles and their Chemistry (Wiley, 2008).
Lafitte, C., Jouannetaud, M.-P., Jacquesy, J.-C., Fahy, J. & Duflos, A. Stereoselective ionic hydrogenation of Vinca alcaloids and vinorelbine in superacids: an access to 4′R-reduced analogs. Tetrahedron Lett. 39, 8281–8282 (1998).
Sommer, J. & Bukala, J. Selective electrophilic activation of alkanes. Acc. Chem. Res. 26, 370–376 (1993).
Cumpstey, I. On a so-called ‘kinetic anomeric effect’ in chemical glycosylation. Org. Biomol. Chem. 10, 2503–2508 (2012).
Hammond, G. S. A correlation of reaction rates. J. Am. Chem. Soc. 77, 334–338 (1955).
Rodriguez, M. A. et al. Stereoselective synthesis of 2-deoxy-2-iodo-glycosides from furanoses. A new route to 2-deoxy-glycosides and 2- deoxy-oligosaccharides of ribo and xylo configuration. J. Org. Chem. 70, 10297–10310 (2005).
Acknowledgements
A.M. acknowledges the Agence Nationale de la Recherche (ANR) for a PhD grant (project ANR-12-BS07-0003-01). N.P. acknowledges the Foundation for the Development of the Chemistry of Natural Products of the French Academy of Sciences for a post-doctoral fellowship. J.D., A.M.-M., S.T. and Y.B. acknowledge the French Centre National de la Recherche Scientifique, (PICS program), the ANR (project ANR-12-BS07-0003-01), the Regional Council Poitou-Charentes and the University of Poitiers for financial support. A.A. and J.J.-B. acknowledge the Centro de Supercomputación de Galicia (CESGA) for computational resources and the Ministery of Economy and Competitiveness of Spain for funding (project CTQ2012-32025).
Author information
Authors and Affiliations
Contributions
Y.B. and P.S. initiated the study. Y.B. and S.T. designed the study. A.M., N.P. and J.D. synthesized the sugar precursors. A.M., A.M.M. and N.P. performed the superacid experiments. A.M., A.M.M., J.D., S.T., Y.B., J.J.-B. and A.A. analysed the NMR spectra. J.J.-B. and A.A. performed the computation. Y.B., J.J.B. and S.T. wrote the paper. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 3371 kb)
Rights and permissions
About this article
Cite this article
Martin, A., Arda, A., Désiré, J. et al. Catching elusive glycosyl cations in a condensed phase with HF/SbF5 superacid. Nature Chem 8, 186–191 (2016). https://doi.org/10.1038/nchem.2399
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.2399
This article is cited by
-
Unravelling the structure of glycosyl cations via cold-ion infrared spectroscopy
Nature Communications (2018)
-
Glycosyl Sulfoxides in Glycosylation Reactions
Topics in Current Chemistry (2018)
-
Synthesis of Chrysogeside B from Halotolerant Fungus Penicillium and Its Antimicrobial Activities Evaluation
Scientific Reports (2017)
-
Glycosyl cations out on parole
Nature Chemistry (2016)