Abstract
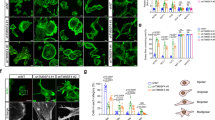
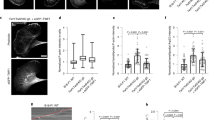
Dynamic actin remodelling processes at the leading edge of migrating tumour cells are concerted events controlled by a fine-tuned temporal and spatial interplay of kinases and phosphatases. Actin severing is regulated by actin depolymerizing factor (ADF)/cofilin, which regulates stimulus-induced lamellipodia protrusion and directed cell motility. Cofilin is activated by dephosphorylation through phosphatases of the slingshot (SSH) family. SSH activity is strongly increased by its binding to filamentous actin (F-actin); however, other upstream regulators remain unknown. Here we show that in response to RhoA activation, protein kinase D1 (PKD1) phosphorylates the SSH enzyme SSH1L at a serine residue located in its actin-binding motif. This generates a 14-3-3-binding motif and blocks the localization of SSH1L to F-actin-rich structures in the lamellipodium by sequestering it in the cytoplasm. Consequently, expression of constitutively active PKD1 in invasive tumour cells enhanced the phosphorylation of cofilin and effectively blocked the formation of free actin-filament barbed ends and directed cell migration.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Bamburg, J. R., McGough, A. & Ono, S. Putting a new twist on actin: ADF/cofilins modulate actin dynamics. Trends Cell. Biol. 9, 364–370 (1999).
Pollard, T. D. & Borisy, G. G. Cellular motility driven by assembly and disassembly of actin filaments. Cell 112, 453–465 (2003).
Yamaguchi, H. & Condeelis, J. Regulation of the actin cytoskeleton in cancer cell migration and invasion. Biochim. Biophys. Acta 1773, 642–652 (2007).
Mouneimne, G. et al. Spatial and temporal control of cofilin activity is required for directional sensing during chemotaxis. Curr. Biol. 16, 2193–2205 (2006).
Sinha, P. et al. Increased expression of epidermal fatty acid binding protein, cofilin, and 14–3-3-sigma (stratifin) detected by two-dimensional gel electrophoresis, mass spectrometry and microsequencing of drug-resistant human adenocarcinoma of the pancreas. Electrophoresis 20, 2952–2960 (1999).
Wang, W., Eddy, R. & Condeelis, J. The cofilin pathway in breast cancer invasion and metastasis. Nature Rev. Cancer 7, 429–440 (2007).
Wang, W. et al. The activity status of cofilin is directly related to invasion, intravasation, and metastasis of mammary tumors. J Cell Biol 173, 395–404 (2006).
Nishita, M. et al. Spatial and temporal regulation of cofilin activity by LIM kinase and Slingshot is critical for directional cell migration. J. Cell Biol. 171, 349–359 (2005).
Mizuno, K. et al. Identification of a human cDNA encoding a novel protein kinase with two repeats of the LIM/double zinc finger motif. Oncogene 9, 1605–1612 (1994).
Huang, T. Y., DerMardirossian, C. & Bokoch, G. M. Cofilin phosphatases and regulation of actin dynamics. Curr. Opin. Cell Biol. 18, 26–31 (2006).
Toshima, J. et al. Cofilin phosphorylation by protein kinase testicular protein kinase 1 and its role in integrin-mediated actin reorganization and focal adhesion formation. Mol. Biol. Cell 12, 1131–1145 (2001).
Agnew, B. J., Minamide, L. S. & Bamburg, J. R. Reactivation of phosphorylated actin depolymerizing factor and identification of the regulatory site. J. Biol. Chem. 270, 17582–17587 (1995).
Nagata-Ohashi, K. et al. A pathway of neuregulin-induced activation of cofilin-phosphatase Slingshot and cofilin in lamellipodia. J. Cell Biol. 165, 465–471 (2004).
Niwa, R., Nagata-Ohashi, K., Takeichi, M., Mizuno, K. & Uemura, T. Control of actin reorganization by Slingshot, a family of phosphatases that dephosphorylate ADF/cofilin. Cell 108, 233–246 (2002).
Nishita, M. et al. Phosphoinositide 3-kinase-mediated activation of cofilin phosphatase Slingshot and its role for insulin-induced membrane protrusion. J. Biol. Chem. 279, 7193–7198 (2004).
Zugaza, J. L., Waldron, R. T., Sinnett-Smith, J. & Rozengurt, E. Bombesin, vasopressin, endothelin, bradykinin, and platelet-derived growth factor rapidly activate protein kinase D through a protein kinase C-dependent signal transduction pathway. J. Biol. Chem. 272, 23952–23960 (1997).
Hausser, A. et al. Protein kinase D regulates vesicular transport by phosphorylating and activating phosphatidylinositol-4 kinase IIIβ at the Golgi complex. Nature Cell Biol. 7, 880–886 (2005).
Liljedahl, M. et al. Protein kinase D regulates the fission of cell surface destined transport carriers from the trans-Golgi network. Cell 104, 409–420 (2001).
Storz, P., Döppler, H. & Toker, A. Protein kinase D mediates mitochondrion-to-nucleus signaling and detoxification from mitochondrial reactive oxygen species. Mol. Cell. Biol. 25, 8520–8530 (2005).
von Blume, J. et al. Phosphorylation at Ser244 by CK1 determines nuclear localization and substrate targeting of PKD2. EMBO J. 26, 4619–4633 (2007).
Eiseler, T., Schmid, M. A., Topbas, F., Pfizenmaier, K. & Hausser, A. PKD is recruited to sites of actin remodelling at the leading edge and negatively regulates cell migration. FEBS Lett. 581, 4279–4287 (2007).
Jaggi, M. et al. E-cadherin phosphorylation by protein kinase D1/protein kinase Cμ is associated with altered cellular aggregation and motility in prostate cancer. Cancer Res. 65, 483–492 (2005).
Riol-Blanco, L. et al. The neuronal protein Kidins220 localizes in a raft compartment at the leading edge of motile immature dendritic cells. Eur. J. Immunol. 34, 108–118 (2004).
Yamamoto, M., Nagata-Ohashi, K., Ohta, Y., Ohashi, K. & Mizuno, K. Identification of multiple actin-binding sites in cofilin-phosphatase Slingshot-1L. FEBS Lett. 580, 1789–1794 (2006).
Döppler, H., Storz, P., Li, J., Comb, M. J. & Toker, A. A phosphorylation state-specific antibody recognizes Hsp27, a novel substrate of protein kinase D. J. Biol. Chem. 280, 15013–15019 (2005).
Döppler, H. & Storz, P. A novel tyrosine phosphorylation site in protein kinase D contributes to oxidative stress-mediated activation. J. Biol. Chem. 282, 31873–31881 (2007).
Song, J., Li, J., Lulla, A., Evers, B. M. & Chung, D. H. Protein kinase D protects against oxidative stress-induced intestinal epithelial cell injury via Rho/ROK/PKC-δ pathway activation. Am. J. Physiol. Cell Physiol. 290, C1469–C1476 (2006).
Storz, P. & Toker, A. Protein kinase D mediates a stress-induced NF-κB activation and survival pathway. EMBO J. 22, 109–120 (2003).
Hutti, J. E. et al. A rapid method for determining protein kinase phosphorylation specificity. Nature Methods 1, 27–29 (2004).
Maekawa, M. et al. Signaling from Rho to the actin cytoskeleton through protein kinases ROCK and LIM-kinase. Science 285, 895–898 (1999).
Mullin, M. J., Lightfoot, K., Marklund, U. & Cantrell, D. A. Differential requirement for RhoA GTPase depending on the cellular localization of protein kinase D. J. Biol. Chem. 281, 25089–25096 (2006).
Yuan, J., Slice, L. W. & Rozengurt, E. Activation of protein kinase D by signaling through Rho and the α subunit of the heterotrimeric G protein G13. J. Biol. Chem. 276, 38619–38627 (2001).
Mouneimne, G. et al. Phospholipase C and cofilin are required for carcinoma cell directionality in response to EGF stimulation. J. Cell Biol. 166, 697–708 (2004).
Raftopoulou, M. & Hall, A. Cell migration: Rho GTPases lead the way. Dev. Biol. 265, 23–32 (2004).
Storz, P., Döppler, H. & Toker, A. Protein kinase Cδ selectively regulates protein kinase D-dependent activation of NF-κB in oxidative stress signaling. Mol. Cell. Biol. 24, 2614–2626 (2004).
Matthews, S. A., Rozengurt, E. & Cantrell, D. Protein kinase D. A selective target for antigen receptors and a downstream target for protein kinase C in lymphocytes. J. Exp. Med. 191, 2075–2082 (2000).
Rey, O., Sinnett-Smith, J., Zhukova, E. & Rozengurt, E. Regulated nucleocytoplasmic transport of protein kinase D in response to G protein-coupled receptor activation. J. Biol. Chem. 276, 49228–49235 (2001).
Baron, C. L. & Malhotra, V. Role of diacylglycerol in PKD recruitment to the TGN and protein transport to the plasma membrane. Science 295, 325–328 (2002).
van Rheenen, J. et al. EGF-induced PIP2 hydrolysis releases and activates cofilin locally in carcinoma cells. J. Cell Biol. 179, 1247–1259 (2007).
Sidani, M. et al. Cofilin determines the migration behavior and turning frequency of metastatic cancer cells. J. Cell Biol. 179, 777–791 (2007).
Ichetovkin, I., Grant, W. & Condeelis, J. Cofilin produces newly polymerized actin filaments that are preferred for dendritic nucleation by the Arp2/3 complex. Curr. Biol. 12, 79–84 (2002).
Dawe, H. R., Minamide, L. S., Bamburg, J. R. & Cramer, L. P. ADF/cofilin controls cell polarity during fibroblast migration. Curr. Biol. 13, 252–257 (2003).
Zebda, N. et al. Phosphorylation of ADF/cofilin abolishes EGF-induced actin nucleation at the leading edge and subsequent lamellipod extension. J. Cell Biol. 151, 1119–1128 (2000).
Storz, P., Döppler, H., Johannes, F. J. & Toker, A. Tyrosine phosphorylation of protein kinase D in the pleckstrin homology domain leads to activation. J. Biol. Chem. 278, 17969–17976 (2003).
Cai, L., Marshall, T. W., Uetrecht, A. C., Schafer, D. A. & Bear, J. E. Coronin 1B coordinates Arp2/3 complex and cofilin activities at the leading edge. Cell 128, 915–929 (2007).
Jauliac, S. et al. The role of NFAT transcription factors in integrin-mediated carcinoma invasion. Nature Cell Biol. 4, 540–544 (2002).
Acknowledgements
We thank L. Lewis-Tuffin for help with the video microscopy, and R. Mooney (University of Rochester Medical Center, New York) for providing MTLn3 cells. This work was sponsored in part by funds from the Mayo Foundation and the Mayo Comprehensive Cancer Center, the Mayo Clinic Breast Cancer SPORE (CA116201-03DR4), a 'Friends for an Earlier Breast Cancer Test' Grant (all to P.S.) and by a grant-in-aid for scientific research from the Ministry of Education, Culture, Sports, Science, and Technology of Japan (to K.M.).
Author information
Authors and Affiliations
Contributions
This publication is a collaborative work of the laboratories of P.S. and K.M. Data shown were generated by T.E., H.D., I.K.Y. and K.K. The data were evaluated by P.S. The manuscript was written by P.S. and K.M.
Corresponding author
Supplementary information
Supplementary Information
Supplementary Information (PDF 1467 kb)
Rights and permissions
About this article
Cite this article
Eiseler, T., Döppler, H., Yan, I. et al. Protein kinase D1 regulates cofilin-mediated F-actin reorganization and cell motility through slingshot. Nat Cell Biol 11, 545–556 (2009). https://doi.org/10.1038/ncb1861
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1861
This article is cited by
-
Region-specific inhibition of 14-3-3 proteins induces psychomotor behaviors in mice
npj Schizophrenia (2019)
-
Mechanism of IRSp53 inhibition by 14-3-3
Nature Communications (2019)
-
Protein kinase D1: gatekeeper of the epithelial phenotype and key regulator of cancer metastasis?
British Journal of Cancer (2018)
-
Cofilin and profilin: partners in cancer aggressiveness
Biophysical Reviews (2018)
-
Src-mediated tyrosine phosphorylation of Protein Kinase D2 at focal adhesions regulates cell adhesion
Scientific Reports (2017)