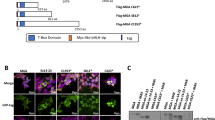
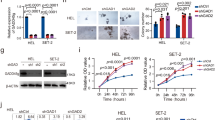
Abstract
Loss-of-function mutations of RUNX1 have been found in acute myeloid leukemia (AML) and myelodysplastic syndromes (MDSs). Although several reports have suggested roles for RUNX1 as a tumor suppressor, its precise function remains unknown. Because gene alterations of RUNX1 by themselves do not lead to the development of leukemia in mouse models, additional mutation(s) would be required for leukemia development. Here, we report that the C-terminal deletion mutant of RUNX1, RUNX1dC, attenuates DNA-damage repair responses in hematopoietic stem/progenitor cells. γH2AX foci, which indicate the presence of DNA double-strand breaks, were more abundantly accumulated in RUNX1dC-transduced lineage−Sca1+c-kit+ (LSK) cells than in mock-transduced LSK cells both in a steady state and after γ-ray treatment. Expression profiling by real-time -PCR array revealed RUNX1dC represses the expression of Gadd45a, a sensor of DNA stress. Furthermore, bone marrow cells from MDS/AML patients harboring the RUNX1-C-terminal mutation showed significantly lower levels of GADD45A expression compared with those from MDS/AML patients with wild-type RUNX1. As for this mechanism, we found that RUNX1 directly regulates the transcription of GADD45A and that RUNX1 and p53 synergistically activate the GADD45A transcription. Together, these results suggest Gadd45a dysfunction due to RUNX1 mutations can cause additional mutation(s) required for multi-step leukemogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Hirai H . Molecular mechanisms of myelodysplastic syndrome. Jpn J Clin Oncol 2003; 33: 153–160.
Quesnel B, Guillerm G, Vereecque R, Wattel E, Preudhomme C, Bauters F et al. Methylation of the p15(INK4b) gene in myelodysplastic syndromes is frequent and acquired during disease progression. Blood 1998; 91: 2985–2990.
Sugimoto K, Hirano N, Toyoshima H, Chiba S, Mano H, Takaku F et al. Mutations of the p53 gene in myelodysplastic syndrome (MDS) and MDS-derived leukemia. Blood 1993; 81: 3022–3026.
Russell M, List A, Greenberg P, Woodward S, Glinsmann B, Parganas E et al. Expression of EVI1 in myelodysplastic syndromes and other hematologic malignancies without 3q26 translocations. Blood 1994; 84: 1243–1248.
Harada Y, Harada H . Molecular pathways mediating MDS/AML with focus on AML1/RUNX1 point mutations. J Cell Physiol 2009; 220: 16–20.
Yoneda-Kato N, Look AT, Kirstein MN, Valentine MB, Raimondi SC, Cohen KJ et al. The t(3;5)(q25.1;q34) of myelodysplastic syndrome and acute myeloid leukemia produces a novel fusion gene, NPM-MLF1. Oncogene 1996; 12: 265–275.
Delhommeau F, Dupont S, Della Valle V, James C, Trannoy S, Masse A et al. Mutation in TET2 in myeloid cancers. N Engl J Med 2009; 360: 2289–2301.
Speck NA, Gilliland DG . Core-binding factors in haematopoiesis and leukaemia. Nat Rev Cancer 2002; 2: 502–513.
Tang JL, Hou HA, Chen CY, Liu CY, Chou WC, Tseng MH et al. AML1/RUNX1 mutations in 470 adult patients with de novo acute myeloid leukemia: prognostic implication and interaction with other gene alterations. Blood 2009; 114: 5352–5361.
Song WJ, Sullivan MG, Legare RD, Hutchings S, Tan X, Kufrin D et al. Haploinsufficiency of CBFA2 causes familial thrombocytopenia with propensity to develop acute myelogenous leukaemia. Nat Genet 1999; 23: 166–175.
Ichikawa M, Asai T, Saito T, Seo S, Yamazaki I, Yamagata T et al. AML-1 is required for megakaryocytic maturation and lymphocytic differentiation, but not for maintenance of hematopoietic stem cells in adult hematopoiesis. Nat Med 2004; 10: 299–304.
Higuchi M, O’Brien D, Kumaravelu P, Lenny N, Yeoh EJ, Downing JR . Expression of a conditional AML1-ETO oncogene bypasses embryonic lethality and establishes a murine model of human t(8;21) acute myeloid leukemia. Cancer Cell 2002; 1: 63–74.
Jackson SP, Bartek J . The DNA-damage response in human biology and disease. Nature 2009; 461: 1071–1078.
Lindahl T, Barnes DE . Repair of endogenous DNA damage. Cold Spring Harb Symp Quant Biol 2000; 65: 127–133.
Hoeijmakers JH . Genome maintenance mechanisms for preventing cancer. Nature 2001; 411: 366–374.
Kundu M, Compton S, Garrett-Beal L, Stacy T, Starost MF, Eckhaus M et al. Runx1 deficiency predisposes mice to T-lymphoblastic lymphoma. Blood 2005; 106: 3621–3624.
Liebermann DA, Hoffman B . Gadd45 in stress signaling. J Mol Signal 2008; 3: 15.
Bennett JM, Catovsky D, Daniel MT, Flandrin G, Galton DA, Gralnick HR et al. Proposals for the classification of the myelodysplastic syndromes. Br J Haematol 1982; 51: 189–199.
Harada H, Harada Y, Niimi H, Kyo T, Kimura A, Inaba T . High incidence of somatic mutations in the AML1/RUNX1 gene in myelodysplastic syndrome and low blast percentage myeloid leukemia with myelodysplasia. Blood 2004; 103: 2316–2324.
Satoh Y, Matsumura I, Tanaka H, Ezoe S, Fukushima K, Tokunaga M et al. AML1/RUNX1 works as a negative regulator of c-Mpl in hematopoietic stem cells. J Biol Chem 2008; 283: 30045–30056.
Harada H, Harada Y, Tanaka H, Kimura A, Inaba T . Implications of somatic mutations in the AML1 gene in radiation-associated and therapy-related myelodysplastic syndrome/acute myeloid leukemia. Blood 2003; 101: 673–680.
Tanaka T, Tanaka K, Ogawa S, Kurokawa M, Mitani K, Nishida J et al. An acute myeloid leukemia gene, AML1, regulates hemopoietic myeloid cell differentiation and transcriptional activation antagonistically by two alternative spliced forms. EMBO J 1995; 14: 341–350.
Rogakou EP, Pilch DR, Orr AH, Ivanova VS, Bonner WM . DNA double-stranded breaks induce histone H2AX phosphorylation on serine 139. J Biol Chem 1998; 273: 5858–5868.
Mah LJ, El-Osta A, Karagiannis TC . gammaH2AX: a sensitive molecular marker of DNA damage and repair. Leukemia 2010; 24: 679–686.
Nojima K, Hochegger H, Saberi A, Fukushima T, Kikuchi K, Yoshimura M et al. Multiple repair pathways mediate tolerance to chemotherapeutic cross-linking agents in vertebrate cells. Cancer Res 2005; 65: 11704–11711.
Mori T, Nakane M, Hattori T, Matsunaga T, Ihara M, Nikaido O . Simultaneous establishment of monoclonal antibodies specific for either cyclobutane pyrimidine dimer or (6-4)photoproduct from the same mouse immunized with ultraviolet-irradiated DNA. Photochem Photobiol 1991; 54: 225–232.
Perugini M, Kok CH, Brown AL, Wilkinson CR, Salerno DG, Young SM et al. Repression of Gadd45alpha by activated FLT3 and GM-CSF receptor mutants contributes to growth, survival and blocked differentiation. Leukemia 2009; 23: 729–738.
Hollander MC, Alamo I, Jackman J, Wang MG, McBride OW, Fornace Jr AJ . Analysis of the mammalian gadd45 gene and its response to DNA damage. J Biol Chem 1993; 268: 24385–24393.
Kastan MB, Zhan Q, el-Deiry WS, Carrier F, Jacks T, Walsh WV et al. A mammalian cell cycle checkpoint pathway utilizing p53 and GADD45 is defective in ataxia-telangiectasia. Cell 1992; 71: 587–597.
Hollander MC, Sheikh MS, Bulavin DV, Lundgren K, Augeri-Henmueller L, Shehee R et al. Genomic instability in Gadd45a-deficient mice. Nat Genet 1999; 23: 176–184.
Yamashita N, Osato M, Huang L, Yanagida M, Kogan SC, Iwasaki M et al. Haploinsufficiency of Runx1/AML1 promotes myeloid features and leukaemogenesis in BXH2 mice. Br J Haematol 2005; 131: 495–507.
Krejci O, Wunderlich M, Geiger H, Chou FS, Schleimer D, Jansen M et al. p53 signaling in response to increased DNA damage sensitizes AML1-ETO cells to stress-induced death. Blood 2008; 111: 2190–2199.
Alcalay M, Meani N, Gelmetti V, Fantozzi A, Fagioli M, Orleth A et al. Acute myeloid leukemia fusion proteins deregulate genes involved in stem cell maintenance and DNA repair. J Clin Invest 2003; 112: 1751–1761.
Vairapandi M, Balliet AG, Hoffman B, Liebermann DA . GADD45b and GADD45g are cdc2/cyclinB1 kinase inhibitors with a role in S and G2/M cell cycle checkpoints induced by genotoxic stress. J Cell Physiol 2002; 192: 327–338.
Smith ML, Chen IT, Zhan Q, Bae I, Chen CY, Gilmer TM et al. Interaction of the p53-regulated protein Gadd45 with proliferating cell nuclear antigen. Science 1994; 266: 1376–1380.
Smith ML, Ford JM, Hollander MC, Bortnick RA, Amundson SA, Seo YR et al. p53-mediated DNA repair responses to UV radiation: studies of mouse cells lacking p53, p21, and/or gadd45 genes. Mol Cell Biol 2000; 20: 3705–3714.
Gupta M, Gupta SK, Balliet AG, Hollander MC, Fornace AJ, Hoffman B et al. Hematopoietic cells from Gadd45a- and Gadd45b-deficient mice are sensitized to genotoxic-stress-induced apoptosis. Oncogene 2005; 24: 7170–7179.
Steensma DP, Gibbons RJ, Mesa RA, Tefferi A, Higgs DR . Somatic point mutations in RUNX1/CBFA2/AML1 are common in high-risk myelodysplastic syndrome, but not in myelofibrosis with myeloid metaplasia. Eur J Haematol 2005; 74: 47–53.
Chen CY, Lin LI, Tang JL, Ko BS, Tsay W, Chou WC et al. RUNX1 gene mutation in primary myelodysplastic syndrome—the mutation can be detected early at diagnosis or acquired during disease progression and is associated with poor outcome. Br J Haematol 2007; 139: 405–414.
Acknowledgements
We thank the Department of Dermatology, Osaka University Graduate School of Medicine for help with UV-B-exposure experiments, Y Sowa and T Sakai for help with reporter assays of Gadd45a, and J Yamauchi for technical advice. We appreciate discussions with and suggestions of C Masutani, K Oritani, T Yokota and S Ezoe. This research was supported in part by a Grant-in Aid for Scientific Research (C) (to Y Satoh), a Grant-in Aid for Scientific Research (B) (to I Matsumura) from MEXT and by the Sankyo Foundation of Life Science.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Leukemia website
Rights and permissions
About this article
Cite this article
Satoh, Y., Matsumura, I., Tanaka, H. et al. C-terminal mutation of RUNX1 attenuates the DNA-damage repair response in hematopoietic stem cells. Leukemia 26, 303–311 (2012). https://doi.org/10.1038/leu.2011.202
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2011.202
Keywords
This article is cited by
-
RUNX1 mutations mitigate quiescence to promote transformation of hematopoietic progenitors in Fanconi anemia
Leukemia (2023)
-
Myeloid neoplasms and clonal hematopoiesis from the RUNX1 perspective
Leukemia (2022)
-
Activating c-KIT mutations confer oncogenic cooperativity and rescue RUNX1/ETO-induced DNA damage and apoptosis in human primary CD34+ hematopoietic progenitors
Leukemia (2015)
-
The RUNX family: developmental regulators in cancer
Nature Reviews Cancer (2015)
-
Recurrent CDC25C mutations drive malignant transformation in FPD/AML
Nature Communications (2014)