Abstract
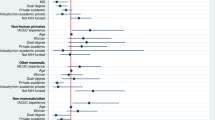
Support for Institutional Animal Care and Use Committees (IACUCs) varies among those in animal use–related professions. The authors designed and carried out an anonymous survey to solicit opinions on the structure and organizational functions of IACUCs. They found that most respondents believed a single, institution-based IACUC was an appropriate venue for institutional approval of animal care and use, that their IACUCs represented their institutions' constituencies and that the unaffiliated IACUC members adequately represented their surrounding communities. Respondents believed that members came prepared for IACUC meetings, and a majority agreed that full committee reviews were more thorough than designated member reviews. The quality of veterinary care for animals was deemed to be very good. Participants reported that the status of the person submitting an animal use protocol, the perceived monetary value of a grant associated with a protocol and pressure for a rapid protocol review did not alter the quality of the protocol review. On many of the survey items, opinions of IACUC members differently significantly from those of non-members, and opinions of non-member IACUC administrators differed from those of IACUC chairpersons, perhaps owing to differences in responsibilities and perceived status.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
We are sorry, but there is no personal subscription option available for your country.
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Steneck, N.H. Role of the institutional animal care and use committee in monitoring research. Ethics Behav. 7, 173–184 (1997).
Shapiro, K. Institutional oversight of animal-based research: tossing a bad coin. PSYETA News 21, 7–8 (2001).
Hsin, C. & Tillman, T. Federal regulations hound animal research. Yale Daily News (3 March 2010).
Rozmiarek, H. in The IACUC Handbook 2nd edn. (eds. Silverman, J., Suckow, M.A. & Murthy, S. ) 1–9 (CRC Press, Boca Raton, FL, 2007).
Plous, S. & Herzog, H. Animal research. Reliability of protocol reviews for animal research. Science 293, 608–609 (2001).
Dresser, R. Developing standards in animal research review. J. Am. Vet. Med. Assoc. 194, 1184–1191 (1989).
Dresser, R. Review standards for animal research: A closer look. ILAR News 32, 2–7 (1990).
Greene, M.E., Pitts, M.E. & James, M.L. Training strategies for IACUC members and the institutional official. ILAR J. 48, 131–142 (2007).
Collins, J.G. Postapproval monitoring and the IACUC. ILAR J. 49, 388–392 (2008).
Galvin, S.L. & Herzog, H.A. The ethical judgment of animal research. Ethics Behav. 2, 263–286 (1992).
Orlans, F.B. Animals, science, and ethics—Section V. Policy issues in the use of animals in research, testing, and education. Hastings Cent. Rep. 20, S25–30 (1990).
Orlans, F.B. Ethical decision making about animal experiments. Ethics Behav. 7, 163–171 (1997).
Orlans, F.B., Beauchamp, T.M., Dresser, R., Morton, D.B. & Gluck, J.P. The Human Use of Animals: Case Studies in Ethical Choice (Oxford University Press, NY, 1998).
Houde, L., Dumas, C. & Leroux, T. Animal ethical evaluation: an observational study of Canadian IACUCs. Ethics Behav. 13, 333–350 (2003).
Silverman, J., Baker, S.P. & Lidz, C.W. A self-assessment survey of the Institutional Animal Care and Use Committee, Part 1: animal welfare and protocol compliance. Lab Anim. (NY) 41, 230–235 (2012).
European Commission. Directive 2010/63/EU of the European Parliament and of the Council of 22 September on the protection of animals used for scientific purposes. Off. J. EU L276, 33–79 (2010).
Rollin, B.E. & Loew, F.M. Assessing the reviewers of animal research. Science 294, 1831–1832 (2001).
Gingery, D. Single IRB policy for multi-site trials may soon get White House OK; NCI hopeful. The Pink Sheet 73, 24–25 (2011).
Newcomer, C.E. in The IACUC Handbook 2nd edn. (eds. Silverman, J., Suckow, M.A. & Murthy, S. ) 37–60 (CRC Press, Boca Raton, FL, 2007).
Code of Federal Regulations, Title 9, Chapter 1, Subchapter A, Part 2.
Public Health Service. Policy on Humane Care and Use of Laboratory Animals (US Department of Health and Human Services, Washington, DC, 1986; amended, 2002).
Keith-Spiegel, P. & Koocher, G.P. The IRB paradox: could the protectors also encourage deceit? Ethics Behav. 15, 339–349 (2005).
Schuppli, C.A. & Fraser, D. Factors influencing the effectiveness of research ethics committees. J. Med. Ethics 33, 294–301 (2007).
Theran, P. The SCAW IACUC survey part II: The unaffiliated member. Lab Anim. (NY) 26, 31–32 (1997).
Häyry, H. Should the decisions of ethics committees be based on community values? Med. Health Care Philos. 1, 57–60 (1998).
Graham, K. A study of three IACUCs and their views of scientific merit and alternatives. J. Appl. Anim. Welf. Sci. 5, 75–81 (2002).
Institute for Laboratory Animal Research. Guide for the Care and Use of Laboratory Animals 8th edn. (National Academies Press, Washington, DC, 2011).
Silverman, J. The attending veterinarian as an ally and leader of the IACUC? Lab Anim (NY) 29, 26–27 (2000).
Silverman, J. Do pressure and prejudice influence the IACUC? Lab Anim (NY) 26, 23–25 (1997).
Acknowledgements
We thank Molly Greene and Christopher Lyons for their help in determining the number of listserv name duplications.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Silverman, J., Baker, S. & Lidz, C. A self-assessment survey of the Institutional Animal Care and Use Committee, Part 2: structure and organizational functions. Lab Anim 41, 289–294 (2012). https://doi.org/10.1038/laban1012-289
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/laban1012-289