Abstract
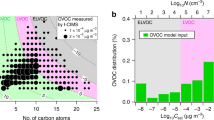
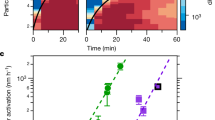
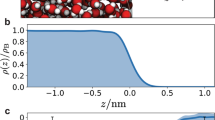
Atmospheric aerosols directly and indirectly affect the radiative balance of the Earth’s atmosphere1. Nanoparticles are a key component of atmospheric aerosols, growing rapidly under ambient conditions2,3,4. Organic species are thought to lead to the growth of nanoparticles smaller than 20 nm (refs 5, 6), but the identity of these species and the underlying chemical mechanisms remain elusive. Here we exposed nanoparticles to a range of organic vapours—2,4-hexadienal, glyoxal and trimethylamine—and monitored particle size to determine the contribution of organic vapours to nanoparticle growth. We show that organic species enhance the growth of nanoparticles, producing non-volatile oligomers, polymers and alkylaminium sulphates in the particle phase. Nanoparticle growth increased with relative humidity in the presence of glyoxal and trimethylamine, but decreased at higher relative humidities in the presence of 2,4-hexadienal, dependent on the reaction mechanism of the organic species involved. Oligomerization and polymerization were largely suppressed in particles smaller than 4 nm and nanoparticle growth increased with particle size. Our findings help to explain the presence of previously measured, but unidentified non-volatile compounds in atmospheric nanoparticles and to improve model simulations of new particle formation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Solomon, S. et al. IPCC Climate Change 2007: The Physical Science Basis (Cambridge Univ. Press, 2007).
McMurry, P. H. et al. A criterion for new particle formation in the sulfur-rich Atlanta atmosphere. J. Geophys. Res. 110, D22S02 (2005).
Zhang, Q. et al. Insights into the chemistry of new particle formation and growth events in Pittsburgh based on aerosol mass spectrometry. Environ. Sci. Technol. 38, 4797–4809 (2004).
Russell, L. M. et al. Nanoparticle growth following photochemical alpha- and beta-pinene oxidation at Appledore Island during International Consortium for Research on Transport and Transformation/Chemistry of Halogens at the Isles of Shoals 2004. J. Geophys. Res. 112, D10S21 (2007).
Ehn, M. et al. Non-volatile residuals of newly formed atmospheric particles in the boreal forest. Atmos. Chem. Phys. 7, 677–684 (2007).
Wehner, B. et al. The contribution of sulfuric acid and non-volatile compounds on the growth of freshly formed atmospheric aerosols. Geophys. Res. Lett. 32, L17810 (2005).
Ball, S. M., Hanson, D. R., Eisele, F. L. & McMurry, P. H. Laboratory studies of particle nucleation: Initial results for H2SO4, H2O, and NH3 vapours. J. Geophys. Res. 104, 23709–23718 (1999).
Zhang, R. et al. Atmospheric new particle formation enhanced by organic acids. Science 304, 1487–1490 (2004).
Yu, F. Q. & Turco, R. P. From molecular clusters to nanoparticles: Role of ambient ionization in tropospheric aerosol formation. J. Geophys. Res. 106, 4797–4814 (2001).
Lee, S. H. et al. Particle formation by ion nucleation in the upper troposphere and lower stratosphere. Science 301, 1886–1889 (2003).
Zhang, R. et al. Formation of nano-sized particles of blue haze enhanced by anthropogenic pollution. Proc. Natl Acad. Sci. USA 106, 17650–17654 (2009).
Zhang, K. M. & Wexler, A. S. A hypothesis for growth of fresh atmospheric nuclei. J. Geophys. Res. 107, 4577 (2002).
Laaksonen, A. et al. The role of VOC oxidation products in continental new particle formation. Atmos. Chem. Phys. 8, 2657–2665 (2008).
Smith, J. N. et al. Chemical composition of atmospheric nanoparticles formed from nucleation in Tecamac, Mexico: Evidence for an important role for organic species in nanoparticles growth. Geophys. Res. Lett. 35, L04808 (2008).
Zhao, J., Levitt, N. P. & Zhang, R. Heterogeneous chemistry of octanal and 2,4-hexadienal with sulfuric acid. Geophys. Res. Lett. 32, L09802 (2005).
Zhao, J., Levitt, N. P., Zhang, R. & Chen, J. Heterogeneous reactions of methylglyoxal in acidic media: Implications for secondary organic aerosol formation. Environ. Sci. Technol. 40, 7682–7687 (2006).
Krizner, H. E., De Haan, D. O. & Kua, J. Thermodynamics and kinetics of methylglyoxal dimer formation: A computational study. J. Phys. Chem. 113, 6994–7001 (2009).
Barsanti, K. C., McMurry, P. H. & Smith, J. N. The potential contribution of organic salts to new particle growth. Atmos. Chem. Phys. 9, 2949–2957 (2009).
Zhang, R. et al. Variability in morphology, hygroscopic and optical properties of soot aerosols during internal mixing in the atmosphere. Proc. Natl Acad. Sci. USA 105, 10291–10296 (2008).
Fortner, E. C., Zhao, J. & Zhang, R. Development of ion drift-chemical ionization mass spectrometry. Anal. Chem. 76, 5436–5440 (2004).
Smith, J. N., Moore, K. F., McMurry, P. H. & Eisele, F. L. Atmospheric measurements of sub-20 nm diameter particle chemical composition by thermal desorption chemical ionization mass spectrometry. Aerosol Sci. Technol. 38, 100–110 (2004).
Liggio, J., Li, S. M. & McLaren, R. Heterogeneous reactions of glyoxal on particulate matter: Identification of acetals and sulfate esters. Environ. Sci. Technol. 39, 1532–1541 (2005).
Sellegri, K. et al. Measurements of organic gases during aerosol formation events in the boreal forest atmosphere during QUEST. Atmos. Chem. Phys. 5, 373–384 (2005).
Yu, Y. & Turco, R. Case studies of particle formation events observed in boreal forests: Implications for nucleation mechanisms. Atmos. Chem. Phys. 8, 6085–6102 (2008).
Fu, T. M., Jacob, D. J. & Heald, C. L. Aqueous-phase reactive uptake of dicarbonyls as a source of organic aerosol over eastern North America. Atmos. Environ. 43, 1814–1822 (2009).
Fan, J. & Zhang, R. Atmospheric oxidation mechanism of isoprene. Environ. Chem. 1, 140–149 (2004).
Suh, I., Lei, W. & Zhang, R. Experimental and theoretical studies of isoprene reaction with NO3 . J. Phys. Chem. 105, 6471–6478 (2001).
Lloyd, J. A., Heaton, K. J. & Johnston, M. V. Reactive uptake of trimethylamine into ammonium nitrate particles. J. Phys. Chem. A 113, 4840–4843 (2009).
Smith, J. N. et al. Observations of aminium salts in atmospheric nanoparticles and possible climatic implications. Proc. Natl Acad. Sci. USA (in the press).
Volkamer, R. et al. A missing sink for gas-phase glyoxal in Mexico City: Formation of secondary organic aerosol. Geophys. Res. Lett. 34, L19807 (2007).
Acknowledgements
This work was supported by the Robert A. Welch Foundation (Grant A-1417) and the US National Science Foundation (AGS-0938352 and CBET-0932705). R.Z. acknowledges further support from the National Natural Science Foundation of China Grant (40728006).
Author information
Authors and Affiliations
Contributions
R.Z. and L.W. wrote the paper. R.Z., L.W. and A.F.K. designed the study. All seven authors performed the experiments. R.Z., L.W. and A.F.K. analysed the data.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 443 kb)
Rights and permissions
About this article
Cite this article
Wang, L., Khalizov, A., Zheng, J. et al. Atmospheric nanoparticles formed from heterogeneous reactions of organics. Nature Geosci 3, 238–242 (2010). https://doi.org/10.1038/ngeo778
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ngeo778
This article is cited by
-
Health impact assessment from exposure to trace metals present in atmospheric PM10 at Ahmedabad, a big city in western India
Environmental Monitoring and Assessment (2021)
-
Formation and growth of sub-3-nm aerosol particles in experimental chambers
Nature Protocols (2020)
-
Impact of neutral and acidic species on cycloalkenes nucleation
Structural Chemistry (2019)
-
Gain Insight into Chemical Components Driving New Particle Growth on a Basis of Particle Hygroscopicity and Volatility Measurements: a Short Review
Current Pollution Reports (2017)
-
The role of low-volatility organic compounds in initial particle growth in the atmosphere
Nature (2016)