Abstract
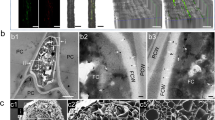
DISEASE-RESISTANT genotypes are the basis for controlling many major microbial pathogens of economic plants. Resistance is often linked with organic, antimicrobial phytoalexins, produced de novo in cells surrounding apoptotic or 'hypersensitive' cells that die rapidly after contact with incompatible pathogens1–3. Here we report the production by resistant genotypes of Theobroma cacaoof four phytoalexins in response to a xylem-invading fungal pathogen; these comprised two phenolics, a triterpenoid and, highly unusually in a higher eukaryote, elemental sulphur as cyclooctasulphur S8. Energy-dispersive X-ray microanalysis revealed a high accumulation of sulphur only in cells and structures in potential contact with the vascular pathogen; that is, xylem parenchyma, xylem vessel cell walls and gels occluding vessels. Our data provide a rare example of cellular localization of an antimicrobial substance and evidence for the first time for accumulation of elemental sulphur in a plant linked with a resistance response; this discovery comes centuries after man first used elemental sulphur as a potent fungicide4,5.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Mansfield, J. M. in Natural Antimicrobial Systems (eds Gould, G.W. et al.) 191–205 (Bath Univ. Press, 1986).
Bailey, J. A. & Mansfield, J. W. Phytoalexins (Blackie, Glasgow, 1982).
Darvill, A. G. & Albersheim, P. A. Rev. Pl. Physiol. 35, 243–275 (1984).
Tweedy, B. G. Residue Rev. 78, 43–68 (1981).
Legris-Delaporte, S., Ferron, F., Landry, J. & Coster, C. Pl. Physiol. 85, 1026–1030 (1987).
Holderness, M. Biochemist 2, 11–13 (1993).
Resende, M. L. V., Flood, J. & Cooper, R. M. Pl. Path. 44, 374–383 (1995).
Yaguchi, Y., Sakurai, N., Nagai, M. & Inoue, T. Chem. Pharm. Bull. 36, 1419–1424 (1988).
Davies, J. R. & Thuraisingham, S. T. J. Chromatogr. 35, 513–518 (1968).
Devys, M. et al. Tetrahedron Lett. 29, 6447–6448 (1988).
Young, L. & Maw, G. A. The Metabolism of Sulphur Compounds (Methuen, London, 1958).
Joyard, J., Fores, E., Blée, E. & Douce, R. Pl. Physiol. 88, 961–964 (1988).
Izak, R. R., Stierle, D. B. & Sims, J. J. Phytochemisfry 21, 229 (1982).
Steudel, R., Holdt, G., Visscher, P. T. & Van Gemerden, H. Arch. Microbiol. 153, 432–437 (1990).
Kylin, H., Atuma, S., Hovander, L. & Jensen, S. Experientia 50, 80–85 (1994).
Mansfield, J. W., Hargreaves, J. A. & Boyle, F. C. Nature 252, 316–317 (1974).
Mayana, S. & Tani, T. Physiol. Pl. Path. 21, 141–149 (1982).
Mace, M. E., Bell, A. A. & Beckman, C. H. Can. J. Bot. 54, 2095–2099 (1976).
Moesta, P., Seydel, U., Lindner, B. & Grisebach, H. Z. Naturf. 37c, 748–751 (1982).
Hahn, M. G., Bonhoff, A. & Grisebach, H. Pl. Physiol. 77, 591–601 (1985).
Cooper, R. M. in Plant Disease Control: Resistance and Susceptibility (eds Staples, R. C. & Toenniesen, G. H.) 105–142 (Wiley, New York, 1981).
Durrands, P. K. & Cooper, R. M. Physiol. molec. Pl. Path. 32, 343–362 (1988).
Ward, E. W. B., Stoessl, A. & Stothers, J. B. Phytochemistry 16, 2024–2025 (1977).
Jolivet, P., Bergeron, E., Zimierski, A. & Meunier, J.-C. Phytochemistry 38, 9–14 (1995).
Beffa, T. Can. J. Microbiol. 39, 736–741 (1993).
Diallo, B., et al. J. nat. Prod. 52, 879–881 (1989).
Gottstein, D. & Cross, D. Trees Struct. Funct. 6, 55–68 (1992).
Jensen, J. S. & Lokke, H. J. Pl. Dis. Prot. 97, 328–338 (1990).
Kokubun, T., Harborne, J. B. & Eagles, J. Phytochemistry 35, 331–333 (1994).
Ride, J. P. in Biochemical Plant Pathology (ed. Callow, J.) 215–236 (Wiley, Chichester, 1983).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cooper, R., Resende, M., Flood, J. et al. Detection and cellular localization of elemental sulphur in disease-resistant genotypes of Theobroma cacao. Nature 379, 159–162 (1996). https://doi.org/10.1038/379159a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/379159a0
This article is cited by
-
How do plants defend themselves against pathogens-Biochemical mechanisms and genetic interventions
Physiology and Molecular Biology of Plants (2022)
-
Integrative transcriptomic and gene co-expression network analysis of host responses upon Verticillium dahliae infection in Gossypium hirsutum
Scientific Reports (2021)
-
Physiological response and sulfur metabolism of the V. dahliae-infected tomato plants in tomato/potato onion companion cropping
Scientific Reports (2016)
-
Kinetics and Histopathology of the Cacao-Ceratocystis cacaofunesta Interaction
Tropical Plant Biology (2013)
-
Latest news about the sulfurtransferase protein family of higher plants
Amino Acids (2011)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.