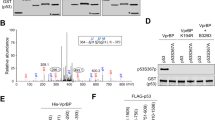
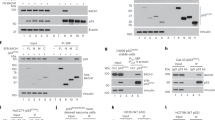
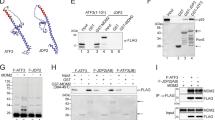
Abstract
The alternative product of the human INK4a/ARF locus, p14ARF, has the potential to act as a tumour suppressor by binding to and inhibiting the p53 antagonist MDM2. Current models propose that ARF function depends on its ability to sequester MDM2 in the nucleolus. Here we describe situations in which stabilization of MDM2 and p53 occur without relocalization of endogenous MDM2 from the nucleoplasm. Conversely, forms of ARF that do not accumulate in the nucleolus retain the capacity to stabilize MDM2 and p53. We therefore propose that nucleolar localization is not essential for ARF function but may enhance the availability of ARF to inhibit MDM2.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Ruas, M. & Peters, G. The p16INK4a/CDKN2A tumor suppressor and its relatives. Biochim. Biophys. Acta 1378, 115–177 (1998).
Sherr, C. J. Tumor surveillance via the ARF-p53 pathway. Genes Dev. 12, 2984–2991 (1998).
Sherr, C. J. & Roberts, J. M. CDK inhibitors: positive and negative regulators of G1-phase progression. Genes Dev. 13, 1501–1512 (1999).
James, M. C. & Peters, G. Alternative product of the p16/CDKN2A locus connects the Rb and p53 tumor suppressors. Prog. Cell Cycle Res. 4, 71–81 (2000).
Sharpless, N. E. & DePinho, R. A. The INK4A/ARF locus and its two gene products. Curr. Opin. Genet. Dev. 9, 22–30 (1999).
Kamijo, T. et al. Functional and physical interactions of the ARF tumor suppressor with p53 and Mdm2. Proc. Natl Acad. Sci. USA 95, 8292–8297 (1998).
Pomerantz, J. et al. The Ink4a tumor suppressor gene product, p19Arf, interacts with MDM2 and neutralizes MDM2's inhibition of p53. Cell 92, 713–723 (1998).
Stott, F. J. et al. The alternative product from the human CDKN2A locus, p14ARF, participates in a regulatory feedback loop with p53 and MDM2. EMBO J. 17, 5001–5014 (1998).
Zhang, Y., Xiong, Y. & Yarbrough, W. G. ARF promotes MDM2 degradation and stabilizes p53:ARF-INK4a locus deletion impairs both the Rb and p53 tumor suppression pathways. Cell 92, 725–734 (1998).
Honda, R. & Yasuda, H. Association of p19ARF with Mdm2 inhibits ubiquitin ligase activity of Mdm2 for tumour suppressor p53. EMBO J. 18, 22–27 (1999).
Midgley, C. A. et al. An N-terminal p14ARF peptide blocks Mdm2-dependent ubiquitination in vitro and can activate p53 in vivo. Oncogene 19, 2312–2323 (2000).
DeGregori, J., Leone, G., Miron, A., Jakoi, L. & Nevins, J. R. Distinct roles for E2F proteins in cell growth control. Proc. Natl Acad. Sci. USA 94, 7245–7250 (1997).
Bates, S. et al. p14ARF links the tumour suppressors RB and p53. Nature 395, 124–125 (1998).
Lohrum, M. A. E., Ashcroft, M., Kubbutat, M. H. G. & Vousden, K. H. Contribution of two independent MDM2-binding domains in p14ARF to p53 stabilization. Curr. Biol. 10, 539–542 (2000).
Weber, J. D. et al. Cooperative signals governing ARF-MDM2 interaction and nucleolar localization of the complex. Mol. Cell. Biol. 20, 2517–2528 (2000).
Quelle, D. E., Zindy, F., Ashmun, R. A. & Sherr, C. J. Alternative reading frames of the INK4a tumor suppressor gene encode two unrelated proteins capable of inducing cell cycle arrest. Cell 83, 993–1000 (1995).
Weber, J. D., Taylor, L. J., Roussel, M. F., Sherr, C. J. & Bar-Sagi, D. Nucleolar Arf sequesters Mdm2 and activates p53. Nature Cell Biol. 1, 20–26 (1999).
Zhang, Y. & Xiong, Y. Mutations in human ARF exon 2 disrupt its nucleolar localization and impair its ability to block nuclear export of MDM2 and p53. Mol. Cell 3, 579–591 (1999).
Lindström, M. S. et al. Immunolocalization of human p14ARF to the granular component of the interphase nucleus. Exp. Cell Res. 256, 400–410 (2000).
Rizos, H., Darmanian, A. P., Mann, G. J. & Kefford, R. F. Two arginine rich domains in the p14ARF tumour suppressor mediate nucleolar localization. Oncogene 19, 2978–2985 (2000).
Roth, J., Dobbelstein, M., Freedman, D. A., Shenk, T. & Levine, A. J. Nucleo–cytoplasmic shuttling of the hdm2 oncoprotein regulates the levels of the p53 protein via a pathway used by the human immunodeficiency virus rev protein. EMBO J. 17, 554–564 (1998).
Freedman, D. A. & Levine, A. J. Nuclear export is required for degradation of endogenous p53 by MDM2 and human papillomavirus E6. Mol. Cell. Biol. 18, 7288–7293 (1998).
Tao, W. & Levine, A. J. Nucleocytoplasmic shuttling of oncoprotein Hdm2 is required for Hdm2-mediated degradation of p53. Proc. Natl Acad. Sci. USA 96, 3077–3080 (1999).
Tao, W. & Levine, A. J. p19ARF stabilizes p53 by blocking nucleo-cytoplasmic shuttling of Mdm2. Proc. Natl Acad. Sci. USA 96, 6937–6941 (1999).
Diller, L. et al. p53 functions as a cell cycle control point in osteosarcoma. Mol. Cell. Biol. 10, 5772–5781 (1990).
Dimri, G. P., Itahana, K., Acosta, M. & Campisi, J. Regulation of a senescence checkpoint response by the E2F1 transcription factor and p14ARF tumor suppressor. Mol. Cell. Biol. 20, 273–285 (2000).
Kurokawa, K., Tanaka, T. & Kato, J. p19ARF prevents G1 cyclin-dependent kinase activation by interacting with MDM2 and activating p53 in mouse fibroblasts. Oncogene 18, 2718–2727 (1999).
Ochs, R. L. Methods used to study structure and function of the nucleolus. Methods Cell Biol. 53, 303–321 (1998).
Quelle, D. E., Cheng, M., Ashmun, R. A. & Sherr, C. J. Cancer-associated mutations at the INK4a locus cancel cell cycle arrest by p16INK4a but not by the alternative reading frame protein p19ARF. Proc. Natl Acad. Sci. USA 94, 669–673 (1997).
Johnston, J. A., Ward, C. L. & Kopito, R. R. Aggresomes: a cellular response to misfolded proteins. J. Cell Biol. 143, 1883–1898 (1998).
García-Mata, R., Bebök, Z., Sorscher, E. J. & Sztul, E. S. Characterization and dynamics of aggresome formation by a cytosolic GFP-chimera. J. Cell Biol. 146, 1239–1254 (1999).
Stommel, J. M. et al. A leucine-rich nuclear export signal in the p53 tetramerization domain: regulation of subcellular localization and p53 activity by NES masking. EMBO J. 18, 1660–1672 (1999).
Lohrum, M. A. E., Ashcroft, M., Kubbutat, M. H. G. & Vousden, K. H. Identification of a cryptic nucleolar-localization signal in MDM2. Nature Cell Biol. 2, 179–181 (2000).
Marechal, V., Elenbaas, B., Piette, J., Nicolas, J-C. & Levine, A. J. The ribosomal L5 protein is associated with mdm-2 and mdm-2–p53 complexes. Mol. Cell. Biol. 14, 7414–7420 (1994).
McConnell, B. B., Starborg, M., Brookes, S. & Peters, G. Inhibitors of cyclin-dependent kinases induce features of replicative senescence in early passage human diploid fibroblasts. Curr. Biol. 8, 351–354 (1998).
Dalton, S. & Treisman, R. Characterization of SAP-1, a protein recruited by serum response factor to the c-fos serum response element. Cell 68, 597–612 (1992).
Bates, S. et al. CDK6 (PLSTIRE) and CDK4 (PSK-J3) are a distinct subset of the cyclin-dependent kinases that associate with cyclin D1. Oncogene 9, 71–79 (1994).
Kubbutat, M. H. G., Jones, S. N. & Vousden, K. H. Regulation of p53 stability by Mdm2. Nature 287, 299–303 (1997).
Acknowledgements
We thank K. Vousden for materials and for communicating data before publication, and M. Fried, M. Owen and D. Ish-Horowicz for discussions and comments on the manuscript. S.L. was the recipient of a long-term EMBO fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Llanos, S., Clark, P., Rowe, J. et al. Stabilization of p53 by p14ARF without relocation of MDM2 to the nucleolus. Nat Cell Biol 3, 445–452 (2001). https://doi.org/10.1038/35074506
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/35074506
This article is cited by
-
Hypoxia-driven deSUMOylation of EXOSC10 promotes adaptive changes in the transcriptome profile
Cellular and Molecular Life Sciences (2024)
-
Farnesoid X receptor functions in cervical cancer via the p14ARF-mouse double minute 2-p53 pathway
Molecular Biology Reports (2022)
-
PKC Dependent p14ARF Phosphorylation on Threonine 8 Drives Cell Proliferation
Scientific Reports (2018)
-
p14ARF interacts with the focal adhesion kinase and protects cells from anoikis
Oncogene (2017)
-
A redox mechanism underlying nucleolar stress sensing by nucleophosmin
Nature Communications (2016)