Abstract
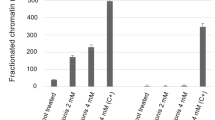
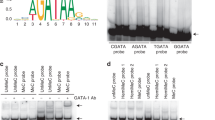
Apaf-1 protein deficiency occurs in human leukaemic blasts and confers resistance to cytochrome-c-dependent apoptosis. Demethylation treatment with 5-aza-2′-deoxycytidine (5aza2dc) increased the sensitivity of the K562 leukaemic cell line to UV light-induced apoptosis in association with increased Apaf-1 protein levels. There was no correlation between Apaf-1 protein expression and Apaf-1 mRNA levels after the demethylation treatment. Methylation-specific polymerase chain reaction was used to show that the methylation can occur within the Apaf-1 promoter region in leukaemic blasts. Apaf-1 DNA methylation was demonstrated in acute myeloid leukaemia, chronic myeloid leukaemia and acute lymphoid leukaemia, suggesting that it is not specific to a particular leukaemia subtype. Apaf-1 protein expression did not correlate with Apaf-1 mRNA levels in human leukaemic blasts. Some leukaemic cells expressed high levels of Apaf-1 mRNA but low levels of Apaf-1 protein. This study suggests that Apaf-1 DNA promoter methylation might contribute to the inactivation of Apaf-1 expression. However, Apaf-1 protein levels might also be controlled at post-transcription level.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Baylin SB and Herman JG . (2000). DNA Trends Genet., 16, 168–174.
Chitnis S, Monteiro J, Glass D, Apatoff B, Salmon J, Concannon P and Gregersen PK (2000). Arthritis Res., 2, 399–406.
Feinberg AP . (2001). Proc. Natl. Acad. Sci. USA, 98, 392–394.
Fu WN, Kelsey SM, Newland AC and Jia L . (2001). Biochem. Biophys. Res. Commun., 282, 268–272.
Fulda S, Kufer MU, Meyer E, van Valen F, Dockhorn-Dworniczak B and Debatin KM . (2001). Oncogene, 20, 5865–5877.
Herman JG, Graff JR, Myohanen S, Nelkin BD and Baylin SB . (1996). Proc. Natl. Acad. Sci. USA, 93, 9821–9826.
Jia L, Patwari Y, Srinivasula SM, Newland AC, Fernandes-Alnemri T, Alnemri ES and Kelsey SM . (2001a). Oncogene, 20, 4817–4826.
Jia L, Srinivasula SM, Liu FT, Newland AC, Fernandes-Alnemri T, Alnemri ES and Kelsey SM . (2001b). Blood, 98, 414–421.
Jones PA . (2001). Nature, 409, 141–144.
Laird PW and Jaenisch R . (1994). Hum. Mol. Genet., 3, 1487–1495.
Li E, Bestor TH and Jaenisch R . (1992). Cell, 69, 915–926.
Li P, Nijhawan D, Budihardjo I, Srinivasula SM, Ahmad M, Alnemri ES and Wang X . (1997). Cell, 91, 479–489.
Liu FT, Kelsey SM, Newland AC and Jia L . (2002). Br. J. Haematol., 117, 333–342.
Lopatina N, Haskell JF, Andrews LG, Poole JC, Saldanha S and Tollefsbol T . (2002). J. Cell Biochem., 84, 324–334.
Perkins C, Kim CN, Fang G and Bhalla KN . (1998). Cancer Res., 58, 4561–4566.
Rountree MR, Bachman KE, Herman JG, and Baylin SB . (2001). Oncogene, 20, 3156–3165.
Shinoura N, Sakurai S, Asai A, Kirino T and Hamada H . (2001). Int. J. Cancer, 93, 252–261.
Soengas MS, Alarcon RM, Yoshida H, Giaccia AJ, Hakem R, Mak TW and Lowe SW . (1999). Science, 284, 156–159.
Soengas MS, Capodieci P, Polsky D, Mora J, Esteller M, Opitz-Araya X, McCombie R, Herman JG, Gerald WL, Lazebnik YA, Cordon-Cardo C and Lowe SW . (2001). Nature, 409, 207–211.
van Noesel MM, van Bezouw S, Salomons GS, Voute PA, Pieters R, Baylin SB, Herman JG and Versteeg R . (2002). Cancer Res., 62, 2157–2161.
Wolf BB, Schuler M, Li W, Eggers-Sedlet B, Lee W, Tailor P, Fitzgerald P, Mills GB and Green DR . (2001). J. Biol. Chem., 276, 34244–34251.
Zou H, Henzel WJ, Liu X, Lutschg A and Wang X . (1997). Cell, 90, 405–413.
Acknowledgements
We are pleased to acknowledge the Cancer Research UK Medical Oncology Unit for collection and storage of peripheral blood, Dr Jude Fitzgibbon for reading the manuscript, and Mr Gary Wright for helping in DNA sequencing.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fu, WN., Bertoni, F., Kelsey, S. et al. Role of DNA methylation in the suppression of Apaf-1 protein in human leukaemia. Oncogene 22, 451–455 (2003). https://doi.org/10.1038/sj.onc.1206147
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1206147
Keywords
This article is cited by
-
Molecular changes in solitary fibrous tumor progression
Journal of Molecular Medicine (2019)
-
Oncogenic miR-23a in Pancreatic Ductal Adenocarcinogenesis Via Inhibiting APAF1
Digestive Diseases and Sciences (2015)
-
Expression of c-Jun, p73, Casp9, and N-ras in thymic epithelial tumors: relationship with the current WHO classification systems
Diagnostic Pathology (2012)
-
Big wheel keeps on turning: apoptosome regulation and its role in chemoresistance
Cell Death & Differentiation (2008)
-
A gene expression profile of tumor suppressor genes commonly methylated in bladder cancer
Journal of Cancer Research and Clinical Oncology (2007)