Abstract
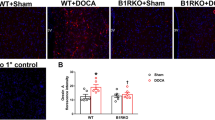
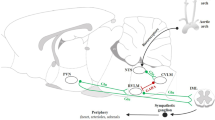
Brain angiotensin-II (Ang-II) type-1 receptors (AT1Rs), which exert profound effects on normal cardiovascular, fluid, and metabolic homeostasis, are overactivated in and contribute to chronic sympathoexcitation and hypertension. Accumulating evidence indicates that the activation of Ang-II type-2 receptors (AT2Rs) in the brain exerts effects that are opposite to those of AT1Rs, lowering blood pressure, and reducing hypertension. Thus, it would be interesting to understand the relative cellular localization of AT1R and AT2R in the brain under normal conditions and whether this localization changes during hypertension. Here, we developed a novel AT1aR-tdTomato reporter mouse strain in which the location of brain AT1aR was largely consistent with that determined in the previous studies. This AT1aR-tdTomato reporter mouse strain was crossed with our previously described AT2R-eGFP reporter mouse strain to yield a novel dual AT1aR/AT2R reporter mouse strain, which allowed us to determine that AT1aR and AT2R are primarily localized to different populations of neurons in brain regions controlling cardiovascular, fluid, and metabolic homeostasis. Using the individual AT1aR-tdTomato reporter mice, we also demonstrated that during hypertension induced by the administration of deoxycorticosterone acetate-salt, there was no shift in the expression of AT1aR from neurons to microglia or astrocytes in the paraventricular nucleus, a brain area important for sympathetic regulation. Using AT2R-eGFP reporter mice under similar hypertensive conditions, we demonstrated that the same was true of AT2R expression in the nucleus of the solitary tract (NTS), an area critical for baroreflex control. Collectively, these findings provided a novel means to assess the colocalization of AT1R and AT2R in the brain and a novel view of their cellular localization in hypertension.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Hollenberg NK. The renin-angiotensin system and sodium homeostasis. J Cardiovasc Pharm. 1984;6 Suppl :S176–183.
Dzau VJ. Circulating versus local renin-angiotensin system in cardiovascular homeostasis. Circulation. 1988;77:I4–13.
Fitzsimons JT. Angiotensin, thirst, and sodium appetite. Physiol Rev. 1998;78:583–686.
Ferguson AV. Angiotensinergic regulation of autonomic and neuroendocrine outputs: critical roles for the subfornical organ and paraventricular nucleus. Neuroendocrinology. 2009;89:370–6.
McKinley MJ, Allen AM, Mathai ML, May C, McAllen RM, Oldfield BJ, et al. Brain angiotensin and body fluid homeostasis. Jpn J Physiol. 2001;51:281–9.
Miller AJ, Arnold AC. The renin-angiotensin system in cardiovascular autonomic control: recent developments and clinical implications. Clin Auton Res. 2019;29:231–43.
Leenen FH. Actions of circulating angiotensin II and aldosterone in the brain contributing to hypertension. Am J Hypertens. 2014;27:1024–32.
Marc Y, Llorens-Cortes C. The role of the brain renin-angiotensin system in hypertension: implications for new treatment. Prog Neurobiol. 2011;95:89–103.
Young CN, Davisson RL. Angiotensin-ii, the brain, and hypertension: an update. Hypertension. 2015;66:920–6.
Lenkei Z, Palkovits M, Corvol P, Llorens-Cortès C. Expression of angiotensin type-1 (at1) and type-2 (at2) receptor mrnas in the adult rat brain: a functional neuroanatomical review. Front Neuroendocrinol. 1997;18:383.
Millan MA, Jacobowitz DM, Aguilera G, Catt KJ. Differential distribution of at1 and at2 angiotensin II receptor subtypes in the rat brain during development. Proc Natl Acad Sci USA. 1991;88:11440–4.
Tsutsumi K, Saavedra JM. Characterization and development of angiotensin II receptor subtypes (at1 and at2) in rat brain. Am J Physiol. 1991;261:R209-216.
Carter DA, Choong YT, Connelly AA, Bassi JK, Hunter NO, Thongsepee N, et al. Functional and neurochemical characterization of angiotensin type 1a receptor-expressing neurons in the nucleus of the solitary tract of the mouse. Am J Physiol Regul. 2017;313:R438–49.
Chen D, Jancovski N, Bassi JK, Nguyen-Huu TP, Choong YT, Palma-Rigo K, et al. Angiotensin type 1a receptors in c1 neurons of the rostral ventrolateral medulla modulate the pressor response to aversive stress. J Neurosci. 2012;32:2051–61.
Gonzalez AD, Wang G, Waters EM, Gonzales KL, Speth RC, Van Kempen TA, et al. Distribution of angiotensin type 1a receptor-containing cells in the brains of bacterial artificial chromosome transgenic mice. Neuroscience. 2012;226:489–509.
de Kloet AD, Wang L, Ludin JA, Smith JA, Pioquinto DJ, Hiller H, et al. Reporter mouse strain provides a novel look at angiotensin type-2 receptor distribution in the central nervous system. Brain Struct Funct. 2016;221:891–912.
Brouwers S, Smolders I, Wainford RD, Dupont AG. Hypotensive and sympathoinhibitory responses to selective central at2 receptor stimulation in spontaneously hypertensive rats. Clin Sci. 2015;129:81–92.
Dai SY, Peng W, Zhang YP, Li JD, Shen Y, Sun XF. Brain endogenous angiotensin II receptor type 2 (at2-r) protects against doca/salt-induced hypertension in female rats. J Neuroinflammation. 2015;12:47.
de Kloet AD, Steckelings UM, Sumners C. Protective angiotensin type 2 receptors in the brain and hypertension. Curr Hypertens Rep. 2017;19:46.
Gao J, Zhang H, Le KD, Chao J, Gao L. Activation of central angiotensin type 2 receptors suppresses norepinephrine excretion and blood pressure in conscious rats. Am J Hypertens. 2011;24:724–30.
Gao L, Wang W, Li H, Sumners C, Zucker IH. Effects of angiotensin type 2 receptor overexpression in the rostral ventrolateral medulla on blood pressure and urine excretion in normal rats. Hypertension. 2008;51:521–7.
Dai SY, Zhang YP, Peng W, Shen Y, He JJ. Central infusion of angiotensin II type 2 receptor agonist compound 21 attenuates doca/nacl-induced hypertension in female rats. Oxid Med Cell Longev. 2016;2016:3981790.
Blanch GT, Freiria-Oliveira AH, Speretta GF, Carrera EJ, Li H, Speth RC, et al. Increased expression of angiotensin II type 2 receptors in the solitary-vagal complex blunts renovascular hypertension. Hypertension. 2014;64:777–83.
Gao J, Zucker IH, Gao L. Activation of central angiotensin type 2 receptors by compound 21 improves arterial baroreflex sensitivity in rats with heart failure. Am J Hypertens. 2014;27:1248–56.
Gao L, Zucker IH. At2 receptor signaling and sympathetic regulation. Curr Opin Pharmacol. 2011;11:124–30.
Legat L, Smolders I, Dupont AG. Gabaergic signaling mediates central cardiovascular angiotensin II type 2 receptor effects. Trends Endocrinol Metab. 2018;29:605–6.
Ruchaya PJ, Speretta GF, Blanch GT, Li H, Sumners C, Menani JV, et al. Overexpression of at2r in the solitary-vagal complex improves baroreflex in the spontaneously hypertensive rat. Neuropeptides. 2016;60:29–36.
Speretta GF, Ruchaya PJ, Delbin MA, Melo MR, Li H, Menani JV, et al. Importance of at1 and at2 receptors in the nucleus of the solitary tract in cardiovascular responses induced by a high-fat diet. Hypertens Res. 2019;42:439–49.
Steckelings UM, Kloet A, Sumners C. Centrally mediated cardiovascular actions of the angiotensin II type 2 receptor. Trends Endocrinol Metab. 2017;28:684–93.
Han C, Rice MW, Cai D. Neuroinflammatory and autonomic mechanisms in diabetes and hypertension. Am J Physiol Endocrinol Metab. 2016;311:E32–41.
Montaniel KR, Harrison DG. Is hypertension a bone marrow disease? Circulation. 2016;134:1369–72.
Santisteban MM, Zubcevic J, Baekey DM, Raizada MK. Dysfunctional brain-bone marrow communication: a paradigm shift in the pathophysiology of hypertension. Curr Hypertens Rep. 2013;15:377–89.
Farina C, Aloisi F, Meinl E. Astrocytes are active players in cerebral innate immunity. Trends Immunol. 2007;28:138–45.
Norris GT, Kipnis J. Immune cells and cns physiology: microglia and beyond. J Exp Med. 2019;216:60–70.
de Kloet AD, Pitra S, Wang L, Hiller H, Pioquinto DJ, Smith JA, et al. Angiotensin type-2 receptors influence the activity of vasopressin neurons in the paraventricular nucleus of the hypothalamus in male mice. Endocrinology. 2016;157:3167–80.
de Kloet AD, Wang L, Pitra S, Hiller H, Smith JA, Tan Y, et al. A unique "angiotensin-sensitive" neuronal population coordinates neuroendocrine, cardiovascular, and behavioral responses to stress. J Neurosci. 2017;37:3478–90.
Grobe JL, Buehrer BA, Hilzendeger AM, Liu X, Davis DR, Xu D, et al. Angiotensinergic signaling in the brain mediates metabolic effects of deoxycorticosterone (doca)-salt in c57 mice. Hypertension. 2011;57:600–7.
Hilzendeger AM, Cassell MD, Davis DR, Stauss HM, Mark AL, Grobe JL, et al. Angiotensin type 1a receptors in the subfornical organ are required for deoxycorticosterone acetate-salt hypertension. Hypertension. 2013;61:716–22.
Jessberger S, Toni N, Clemenson GD Jr., Ray J, Gage FH. Directed differentiation of hippocampal stem/progenitor cells in the adult brain. Nat Neurosci. 2008;11:888–93.
Krause EG, de Kloet AD, Scott KA, Flak JN, Jones K, Smeltzer MD, et al. Blood-borne angiotensin II acts in the brain to influence behavioral and endocrine responses to psychogenic stress. J Neurosci. 2011;31:15009–15.
de Kloet AD, Pioquinto DJ, Nguyen D, Wang L, Smith JA, Hiller H, et al. Obesity induces neuroinflammation mediated by altered expression of the renin-angiotensin system in mouse forebrain nuclei. Physiol Behav. 2014;136:31–8.
Langlet F, Mullier A, Bouret SG, Prevot V, Dehouck B. Tanycyte-like cells form a blood–cerebrospinal fluid barrier in the circumventricular organs of the mouse brain. J Comp Neurol. 2013;521:3389–405.
Kádár A, Sánchez E, Wittmann G, Singru PS, Füzesi T, Marsili A, et al. Distribution of hypophysiotropic thyrotropin-releasing hormone (trh)-synthesizing neurons in the hypothalamic paraventricular nucleus of the mouse. J Comp Neurol. 2010;518:3948–61.
Gautron L, Rutkowski JM, Burton MD, Wei W, Wan Y, Elmquist JK. Neuronal and nonneuronal cholinergic structures in the mouse gastrointestinal tract and spleen. J Comp Neurol. 2013;521:3741–67.
Mousa SA, Shaqura M, Schäper J, Treskatsch S, Habazettl H, Schäfer M, et al. Developmental expression of δ-opioid receptors during maturation of the parasympathetic, sympathetic, and sensory innervations of the neonatal heart: early targets for opioid regulation of autonomic control. J Comp Neurol. 2011;519:957–71.
Liu M, Shi P, Sumners C. Direct anti-inflammatory effects of angiotensin-(1-7) on microglia. J Neurochem. 2016;136:163–71.
Mecca AP, Regenhardt RW, O'Connor TE, Joseph JP, Raizada MK, Katovich MJ, et al. Cerebroprotection by angiotensin-(1-7) in endothelin-1-induced ischaemic stroke. Exp Physiol. 2011;96:1084–96.
Regenhardt RW, Mecca AP, Desland F, Ritucci-Chinni PF, Ludin JA, Greenstein D, et al. Centrally administered angiotensin-(1-7) increases the survival of stroke-prone spontaneously hypertensive rats. Exp Physiol. 2014;99:442–53.
de Kloet AD, Wang L, Pitra S, Hiller H, Smith JA, Tan Y, et al. A unique ‘angiotensin sensitive' neuronal population coordinates neuroendocrine, cardiovascular and behavioral responses to stress. J Neurosci. 2017;37:3478–90.
Franklin KBJ, Paxinos G. The mouse brain: in stereotaxic coordinates. New York, NY: Elsevier; 2008.
Paxinos G, Watson C. The rat brain in stereotaxic coordinates. 7th Edition, Academic Press, San Diego, CA: Elsevier Life Sciences; 2013.
Karnik SS, Unal H, Kemp JR, Tirupula KC, Eguchi S, Vanderheyden PM, et al. International union of basic and clinical pharmacology. Xcix. Angiotensin receptors: interpreters of pathophysiological angiotensinergic stimuli [corrected]. Pharmacol Rev. 2015;67:754–819.
Shi P, Diez-Freire C, Jun JY, Qi Y, Katovich MJ, Li Q, et al. Brain microglial cytokines in neurogenic hypertension. Hypertension. 2010;56:297–303.
Coote JH, Yang Z, Pyner S, Deering J. Control of sympathetic outflows by the hypothalamic paraventricular nucleus. Clin Exp Pharmacol Physiol. 1998;25:461–3.
Ciriello J, Kline RL, Zhang TX, Caverson MM. Lesions of the paraventricular nucleus alter the development of spontaneous hypertension in the rat. Brain Res. 1984;310:355–9.
Nakata T, Takeda K, Itho H, Hirata M, Kawasaki S, Hayashi J, et al. Paraventricular nucleus lesions attenuate the development of hypertension in doca/salt-treated rats. Am J Hypertens. 1989;2:625–30.
Gutkind JS, Kurihara M, Castren E, Saavedra JM. Increased concentration of angiotensin II binding sites in selected brain areas of spontaneously hypertensive rats. J hypertens. 1988;6:79–84.
Lenkei Z, Corvol P, Llorens-Cortes C. Comparative expression of vasopressin and angiotensin type-1 receptor mrna in rat hypothalamic nuclei: a double in situ hybridization study. brain Res Mol brain Res. 1995;34:135–42.
Santisteban MM, Ahmari N, Carvajal JM, Zingler MB, Qi Y, Kim S, et al. Involvement of bone marrow cells and neuroinflammation in hypertension. Circ Res. 2015;117:178–91.
de Kloet AD, Pati D, Wang L, Hiller H, Sumners C, Frazier CJ, et al. Angiotensin type 1a receptors in the paraventricular nucleus of the hypothalamus protect against diet-induced obesity. J Neurosci. 2013;33:4825–33.
Obermuller N, Unger T, Culman J, Gohlke P, de Gasparo M, Bottari SP. Distribution of angiotensin II receptor subtypes in rat brain nuclei. Neurosci Lett. 1991;132:11–15.
Daniels D. Diverse roles of angiotensin receptor intracellular signaling pathways in the control of water and salt intake. In: De Luca LA, Jr., Menani JV, Johnson AK, editors. Neurobiology of body fluid homeostasis: transduction and integration. Boca Raton, FL: CRC Press/Taylor & Francis; 2014.
Ferguson AV, Bains JS. Actions of angiotensin in the subfornical organ and area postrema: implications for long term control of autonomic output. Clin Exp Pharmacol Physiol. 1997;24:96–101.
McKinley MJ, McAllen RM, Pennington GL, Smardencas A, Weisinger RS, Oldfield BJ. Physiological actions of angiotensin II mediated by at1 and at2 receptors in the brain. Clin Exp Pharm Physiol Suppl. 1996;3:S99–104.
Vieira AA, Nahey DB, Collister JP. Role of the organum vasculosum of the lamina terminalis for the chronic cardiovascular effects produced by endogenous and exogenous ang ii in conscious rats. Am J Physiol Regul. 2010;299:R1564–71.
Aguilera G, Young WS, Kiss A, Bathia A. Direct regulation of hypothalamic corticotropin-releasing-hormone neurons by angiotensin-ii. Neuroendocrinology. 1995;61:437–44.
Bains JS, Ferguson AV. Paraventricular nucleus neurons projecting to the spinal cord receive excitatory input from the subfornical organ. Am J Physiol. 1995;268:R625–33.
Zhu GQ, Patel KP, Zucker IH, Wang W. Microinjection of ang ii into paraventricular nucleus enhances cardiac sympathetic afferent reflex in rats. Am J Physiol Heart Circ Physiol. 2002;282:H2039–2045.
Cunningham JT, Beltz T, Johnson RF, Johnson AK. The effects of ibotenate lesions of the median preoptic nucleus on experimentally-induced and circadian drinking behavior in rats. Brain Res. 1992;580:325–30.
McKinley MJ, Yao ST, Uschakov A, McAllen RM, Rundgren M, Martelli D. The median preoptic nucleus: front and centre for the regulation of body fluid, sodium, temperature, sleep and cardiovascular homeostasis. Acta Physiol. 2015;214:8–32.
Abegaz B, Davern PJ, Jackson KL, Nguyen-Huu TP, Bassi JK, Connelly A, et al. Cardiovascular role of angiotensin type 1a receptors in the nucleus of the solitary tract of mice. Cardiovascular Res. 2013;100:181–91.
Colombari E, Colombari DS. Nts at 1a receptor on long-term arterial pressure regulation: putative mechanism. Cardiovasc Res. 2013;100:173–4.
Hasser EM, Cunningham JT, Sullivan MJ, Curtis KS, Blaine EH, Hay M. Area postrema and sympathetic nervous system effects of vasopressin and angiotensin ii. Clin Exp Pharmacol Physiol. 2000;27:432–6.
Nahey DB, Collister JP. Ang ii-induced hypertension and the role of the area postrema during normal and increased dietary salt. Am J Physiol Heart Circ Physiol. 2007;292:H694–700.
Oldfield BJ, Davern PJ, Giles ME, Allen AM, Badoer E, McKinley MJ. Efferent neural projections of angiotensin receptor (at1) expressing neurones in the hypothalamic paraventricular nucleus of the rat. J Neuroendocrinol. 2001;13:139–46.
Rowe BP, Saylor DL, Speth RC. Analysis of angiotensin II receptor subtypes in individual rat brain nuclei. Neuroendocrinology. 1992;55:563–73.
MacGregor DP, Murone C, Song K, Allen AM, Paxinos G, Mendelsohn FA. Angiotensin II receptor subtypes in the human central nervous system. Brain Res. 1995;675:231–40.
Song K, Allen AM, Paxinos G, Mendelsohn FA. Mapping of angiotensin II receptor subtype heterogeneity in rat brain. J Comp Neurol. 1992;316:467–84.
Guimond MO, Gallo-Payet N. The angiotensin II type 2 receptor in brain functions: an update. Int J Hypertens. 2012;2012:351758.
Lenkei Z, Palkovits M, Corvol P, Llorens-Cortes C. Distribution of angiotensin II type-2 receptor (at2) mrna expression in the adult rat brain. J Comp Neurol. 1996;373:322–39.
Haspula D, Clark MA. Neuroinflammation and sympathetic overactivity: mechanisms and implications in hypertension. Auton Neurosci. 2018;210:10–17.
Santisteban MM, Kim S, Pepine CJ, Raizada MK. Brain-gut-bone marrow axis: implications for hypertension and related therapeutics. Circ Res. 2016;118:1327–36.
Biancardi VC, Stranahan AM, Krause EG, de Kloet AD, Stern JE. Cross talk between at1 receptors and toll-like receptor 4 in microglia contributes to angiotensin ii-derived ros production in the hypothalamic paraventricular nucleus. Am J Physiol Heart Circ Physiol. 2016;310:H404–415.
Stern JE, Son S, Biancardi VC, Zheng H, Sharma N, Patel KP. Astrocytes contribute to angiotensin II stimulation of hypothalamic neuronal activity and sympathetic outflow. Hypertension. 2016;68:1483–93.
Joglar B, Rodriguez-Pallares J, Rodriguez-Perez AI, Rey P, Guerra MJ, Labandeira-Garcia JL. The inflammatory response in the mptp model of parkinson's disease is mediated by brain angiotensin: relevance to progression of the disease. J Neurochem. 2009;109:656–69.
Lanz TV, Ding Z, Ho PP, Luo J, Agrawal AN, Srinagesh H, et al. Angiotensin II sustains brain inflammation in mice via tgf-beta. J Clin Investig. 2010;120:2782–94.
Negussie S, Lymperopoulos A, Clark MA. Role of betaarrestin1 in at1 r-mediated mitogen-activated protein kinase activation in wistar and shr brainstem astrocytes. J Neurochem. 2019;148:46–62.
Sumners C, Tang W, Zelezna B, Raizada MK. Angiotensin II receptor subtypes are coupled with distinct signal-transduction mechanisms in neurons and astrocytes from rat brain. Proc Natl Acad Sci USA. 1991;88:7567–71.
Tallant EA, Higson JT. Angiotensin II activates distinct signal transduction pathways in astrocytes isolated from neonatal rat brain. Glia. 1997;19:333–42.
Wu CY, Zha H, Xia QQ, Yuan Y, Liang XY, Li JH, et al. Expression of angiotensin II and its receptors in activated microglia in experimentally induced cerebral ischemia in the adult rats. Mol Cell Biochem. 2013;382:47–58.
O'Callaghan EL, Bassi JK, Porrello ER, Delbridge LM, Thomas WG, Allen AM. Regulation of angiotensinogen by angiotensin II in mouse primary astrocyte cultures. J Neurochem. 2011;119:18–26.
Acknowledgements
This work was supported by AHA grant 17GRNT33660969 and NIH grants HL-125805 (ADdK), HL-145028 (ADdK), HL-093186 (CS), HL-136595 (EGK/CS), HL-096830 (EGK), and HL-122494 (EGK).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. Animal procedures were approved by the Institutional Animal Care and Use Committee at the University of Florida, complied with the National Institutes of Health guidelines and were performed in accordance with the Guide for the Care and Use of Laboratory Animals (eight edition, 2011, published by National Academics Press, 500 Fifth Street NW, Washington, DC, 20001, USA) (AALAC #: 000023; OLAW Assurance #: A3377-01).
Animals were housed in temperature- and humidity-controlled rooms on a 12:12 h light/dark cycle with food and water available ad libitum.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Sumners, C., Alleyne, A., Rodríguez, V. et al. Brain angiotensin type-1 and type-2 receptors: cellular locations under normal and hypertensive conditions. Hypertens Res 43, 281–295 (2020). https://doi.org/10.1038/s41440-019-0374-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41440-019-0374-8
Keywords
This article is cited by
-
The Renin Angiotensin System as a Therapeutic Target in Traumatic Brain Injury
Neurotherapeutics (2023)
-
Angiotensin II increases the firing activity of pallidal neurons and participates in motor control in rats
Metabolic Brain Disease (2023)
-
The impact of excessive salt intake on human health
Nature Reviews Nephrology (2022)
-
ACE2, Circumventricular Organs and the Hypothalamus, and COVID-19
NeuroMolecular Medicine (2022)
-
A colorful view of the brain renin–angiotensin system
Hypertension Research (2020)