Abstract
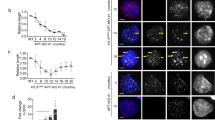
A hallmark of cancer cells is an activated telomere maintenance mechanism, which allows prolonged survival of the malignant cells. In more than 80% of tumours, telomeres are elongated by the enzyme telomerase, which adds de novo telomere repeats to the ends of chromosomes. Cancer cells are also characterized by expression of active LINE-1 elements (L1s, long interspersed nuclear elements-1). L1 elements are abundant retrotransposons in the eukaryotic genome that are primarily known for facilitating aberrant recombination. Using L1-knockdown (KD), we show for the first time that L1 is critical for telomere maintenance in telomerase-positive tumour cells. The reduced length of telomeres in the L1-KD-treated cells correlated with an increased rate of telomere dysfunction foci, a reduced expression of shelterin proteins and an increased rate of anaphase bridges. The decreased telomere length was associated with a decreased telomerase activity and decreased telomerase mRNA level; the latter was increased upon L1 overexpression. L1-KD also led to a decrease in mRNA and protein expression of cMyc and KLF-4, two main transcription factors of telomerase and altered mRNA levels of other stem-cell-associated proteins such as CD44 and hMyb, as well as a corresponding reduced growth of spheroids. The KD of KLF-4 or cMyc decreased the level of L1-ORF1 mRNA, suggesting a specific reciprocal regulation with L1. Thus, our findings contribute to the understanding of L1 as a pathogenicity factor in cancer cells. As L1 is only expressed in pathophysiological conditions, L1 now appears to be target in the rational treatment of telomerase-positive cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Moyzis RK, Buckingham JM, Cram LS, Dani M, Deaven LL, Jones MD et al. A highly conserved repetitive DNA sequence, (TTAGGG)n, present at the telomeres of human chromosomes. Proc Natl Acad Sci USA 1988; 85: 6622–6626.
Blackburn EH . Telomeres. Trends Biochem Sci 1991; 16: 378–381.
Kim NW, Piatyszek MA, Prowse KR, Harley CB, West MD, Ho PL et al. Specific association of human telomerase activity with immortal cells and cancer. Science 2015; 266: 2011–2015.
Griffith JD, Comeau L, Rosenfield S, Stansel RM, Bianchi A, Moss H et al. Mammalian telomeres end in a large duplex loop. Cell 1999; 97: 503–514.
de Lange T . Protection of mammalian telomeres. Oncogene 2002; 21: 532–540.
Verdun RE, Karlseder J . Replication and protection of telomeres. Nature 2007; 447: 924–931.
Harley CB, Futcher AB, Greider CW . Telomeres shorten during ageing of human fibroblasts. Nature 1990; 345: 458–460.
Counter CM, Avilion AA, LeFeuvre CE, Stewart NG, Greider CW et al. Telomere shortening associated with chromosome instability is arrested in immortal cells which express telomerase activity. EMBO J 1992; 11: 1921–1929.
Karlseder J, Smogorzewska A, de Lange T . Senescence induced by altered telomere state, not telomere loss. Science 2002; 295: 2446–2449.
de Lange T . Shelterin: the protein complex that shapes and safeguards human telomeres. Genes Dev 2005; 19: 2100–2110.
Choi KH, Farrell AS, Lakamp AS, Ouellette MM . Characterization of the DNA binding specificity of Shelterin complexes. Nucleic Acid Res 2011; 29: 9206–9223.
Martínez P, Blasco MA . Telomeric and extra-telomeric roles for telomerase and the telomere-binding proteins. Nat Rev Cancer 2011; 11: 161–176.
van Steensel B, Lange T . Control of telomere length by the human telomeric protein TRF1. Nature 1997; 385: 740–743.
Ahmad K, Golic KG . Telomere loss in somatic cells of Drosophila causes cell cycle arrest and apoptosis. Genetics 1999; 151: 1041–1051.
Smogorzewska A, van Steensel B, Bianchi A, Oelmann S, Schaefer MR, Schnapp G et al. Control of human telomere length by TRF1 and TRF2. Mol Cell Biol 2000; 20: 1659–1668.
Bailey SM, Meyne J, Chen DJ, Kurimasa A, Li GC, Lehnert BE et al. DNA double-strand break repair proteins are required to cap the ends of mammalian chromosomes. Proc Natl Acad Sci USA 1999; 96: 14899–14904.
Bailey SM, Cornforth MN, Kurimasa A, Chen DJ, Goodwin EH . Strand-specific postreplicative processing of mammalian telomeres. Science 2001; 293: 2462–2465.
Goytisolo FA, Samper E, Edmonson S, Taccioli GE, Blasco MA . The absence of the dna-dependent protein kinase catalytic subunit in mice results in anaphase bridges and in increased telomeric fusions with normal telomere length and G-strand overhang. Mol Cell Biol 2001; 21: 3642–3651.
Takai H, Smorgorzewska A, de Lange T . DNA damage foci at dysfunctional telomeres. Curr Biol 2003; 13: 1549–1556.
Fumagalli M, Rossiello F, Clerici M, Barozzi S, Cittaro D, Kaplunov JM et al. Telomeric DNA damage is irreparable and causes persistent DNA-damage-response activation. Nat Cell Biol 2012; 14: 355–365.
Morin GB . The human telomere terminal transferase enzyme is a ribonucleoprotein that synthesizes TTAGGG repeats. Cell 1989; 59: 521–529.
Greider CW, Blackburn EH . The telomere terminal transferase of Tetrahymena is a ribonucleoprotein enzyme with two kinds of primer specificity. Cell 1987; 51: 887–898.
Blackburn EH, Greider CW, Henderson E, Lee MS, Shampay J, Shippen-Lentz D . Recognition and elongation of telomeres by telomerase. Genome 1989; 31: 553–560.
Shippen-Lentz D, Blackburn EH . Functional evidence for an RNA template in telomerase. Science 1990; 247: 546–552.
Lewis KA, Wuttke DS . Telomerase and telomere-associated proteins: structural insights into mechanism and evolution. Structure 2012; 20: 28–39.
Lingner J, Hughes TR, Shevchenko A, Mann M, Lundblad V, Cech TR . Reverse transcriptase motifs in the catalytic subunit of telomerase. Science 1997; 276: 561–567.
Damm K, Hemmann U, Garin-Chesa P, Hauel N, Kauffmann I, Priepke H et al. A highly selective telomerase inhibitor limiting human cancer cell proliferation. EMBO J 2001; 20: 6958–6968.
Dikmen ZG, Gellert GC, Jackson S, Gryaznov S, Tressler R, Dogan P et al. In vivo inhibition of lung cancer by GRN163L: a novel human telomerase inhibitor. Cancer Res 2005; 65: 7866–7873.
Hochreiter AE, Xiao H, Goldblatt EM, Gryaznov SM, Miller KD, Badve S et al. Telomerase template antagonist GRN163L disrupts telomere maintenance, tumor growth, and metastasis of breast cancer. Clin Cancer Res 2006; 12: 3184–3192.
Harley CB . Telomerase and cancer therapeutics. Nat Rev Cancer 2008; 8: 167–179.
Fang JL, Beland FA . Long-term exposure to zidovudine delays cell cycle progression, induces apoptosis, and decreases telomerase activity in human hepatocytes. Toxicol Sci 2009; 111: 120–130.
Hao H, Nancai Y, Lei F, Xiong W, Wen S, Guofu H et al. siRNA directed against c-Myc inhibits proliferation and downregulates human telomerase reverse transcriptase in human colon cancer Colo 320 cells. J Exp Clin Cancer Res 2008; 12: 27.
Hu X, Yu N, Fu L, Su W, Huang G, Wu Y et al. Downregulation of human telomerase reverse transcriptase through anti-C-myc siRNA in human colon cancer Colo 320 cells. Nucleosides Nucleotides Nucleic Acids 2009; 28: 1–11.
Takakura M, Kyo S, Kanaya T, Hirano H, Takeda J, Yutsudo M et al. Cloning of human telomerase catalytic subunit (hTERT) gene promoter and identification of proximal core promoter sequences essential for transcriptional activation in immortalized and cancer cells. Cancer Res 1999; 59: 551–557.
Schneider-Stock R, Boltze C, Jäger V, Epplen J, Landt O, Peters B et al. Elevated telomerase activity, c-MYC-, and hTERT mRNA expression: association with tumour progression in malignant lipomatous tumours. J Pathol 2003; 199: 517–525.
Wong CW, Hou PS, Tseng SF, Chien CL, Wu KJ, Chen HF et al. Krüppel-like transcription factor 4 contributes to maintenance of telomerase activity in stem cells. Stem Cells 2010; 28: 1510–1517.
Lander ES, Linton LM, Birren B, Nusbaum C, Zody MC, Baldwin J et al. International Human Genome Sequencing Consortium Initial sequencing and analysis of the human genome. Nature 2011; 409: 860–921.
Brouha B, Schzstak J, Badge RM, Lutz-Prigge S, Farley AH, Moran JV et al. Hot L1s account for the bulk of retrotransposition in the human population. Proc Natl Acad Sci USA 2003; 100: 5280–5285.
Luan DD, Korman MH, Jakubczak JL, Eickbush TH . Reverse transcription of R2Bm RNA is primed by a nick at the chromosomal target site: a mechanism for non-LTR retrotransposition. Cell 1993; 72: 595–605.
Feng Q, Moran JV, Kazazian Jr HH, Boeke JD . Human L1 retrotransposon encodes a conserved endonuclease required for retrotransposition. Cell 1996; 87: 905–916.
Morrish TA, Gilbert N, Myers JS, Vincent BJ, Stamato TD, Taccioli GE et al. DNA repair mediated by endonuclease-independent LINE-1 retrotransposition. Nat Genet 2002; 31: 159–165.
Kolosha VO, Martin SL . High-affinity non-sequence-specific RNA binding by the open reading frame 1 (ORF1) protein from long interspersed nuclear element 1 (LINE-1). J Biol Chem 2003; 278: 8112–8117.
Ergün S, Buschmann C, Heukeshoven J, Dammann K, Schnieders F, Lauke H et al. Cell type-specific expression of LINE-1 open reading frames 1 and 2 in fetal and adult human tissues. J Biol Chem 2004; 279: 27753–27763.
Rodić N, Sharma R, Sharma R, Zampella J, Dai L, Taylor MS et al. Long interspersed element-1 protein expression is a hallmark of many human cancers. Am J Pathol 2014; 184: 1280–1286.
Chen JM, Stenson PD, Cooper DN, Fered C . A systematic analysis of LINE-1 endonuclease-dependent retrotranspositional events causing human genetic disease. Hum Genet 2005; 117: 411–427.
Babushok DV, Kazazian HH Jr . Progress in understanding the biology of the human mutagen LINE-1. Hum Mutat 2007; 28: 527–539.
Garcia-Perez JL, Marchetto MC, Muotri AR, Coufal NG, Gage FH, O'Shea KS et al. LINE-1 retrotransposition in human embryonic stem cells. Hum Mol Genet 2007; 16: 1569–1577.
Kano H, Godoy I, Courtney C, Vetter MR, Gerton GL, Ostertag EM et al. L1 retrotransposition occurs mainly in embryogenesis and creates somatic mosaicism. Genes Dev 2009; 23: 1303–1312.
Beck CR, Garcia-Perez JL, Badge RM, Moran JV . LINE-1 elements in structural variation and disease. Annu Rev Genomics Hum Genet 2011; 12: 187–215.
Sinibaldi-Vallebona P, Matteucci D, Spadafora C . Retrotransposon-encoded reverse transcriptase in the genesis, progression and cellular plasticity of human cancer. Cancers (Basel) 2011; 3: 1141–1157.
Wissing S, Munoz-Lopez M, Macia A, Yang Z, Montano M, Collins W et al. Reprogramming somatic cells into iPS cells activates LINE-1 retroelement mobility. Hum Mol Genet 2012; 21: 2008–2018.
Aoki Y, Nojima M, Suzuki H, Yasui H, Maruyama R, Yamamoto E et al. Genomic vulnerability to LINE-1 hypomethylation is a potential determinant of the clinicogenetic features of multiple myeloma. Genome Med 2012; 4: 101.
Vitullo P, Sciamanna I, Baiocchi M, Sinibaldi-Vallebona P, Spadafora C . LINE-1 retrotransposon copies are amplified during murine early embryo development. Mol Reprod Dev 2012; 79: 118–127.
Sciamanna I, Landriscina M, Pittoggi C, Quirino M, Mearelli C, Beraldi R et al. Inhibition of endogenous reverse transcriptase antagonizes human tumor growth. Oncogene 2005; 24: 3923–3931.
Oricchio E, Sciamanna I, Beraldi R, Tolstonog GV, Schumann GG, Spadafora C . Distinct roles for LINE-1 and HERV-K retroelements in cell proliferation, differentiation and tumor progression. Oncogene 2007; 26: 4226–4233.
Ward IM, Chen J . Histone H2AX is phosphorylated in an ATR-dependent manner in response to replicational stress. J Biol Chem 2002; 276: 47759–47762.
Hao LY, S M, Greider CW . Phosphorylation of H2AX at short telomeres in T cells and fibroblasts. J Biol Chem 2004; 279: 45148–45154.
Atsumi Y, Fujimori H, Fukuda H, Inase A, Shinohe K, Yoshioka Y et al. Onset of quiescence following p53 mediated down-regulation of H2AX in normal cells. PLoS One 2011; 6: e23432.
Bailey SM, Murnane JP . Telomeres, chromosome instability and cancer. Nucleic Acids Res 2006; 34: 2408–2417.
Kyo S, Takakura M, Taira T, Kanaya T, Itoh H, Yutsudo M et al. Sp1 cooperates with c-Myc to activate transcription of the human telomerase reverse transcriptase gene (hTERT). Nucleic Acid Res 2000; 28: 669–677.
Kim J, Chu J, Shen X, Wang J, Orkin SH . An extended transcriptional network for pluripotency of embryonic stem cells. Cell 2008; 132: 1049–1061.
Farkash EA, Kao GD, Horman SR, Prak ET . Gamma radiation increases endonuclease-dependent L1 retrotransposition in a cultured cell assay. Nucleic Acids Res 2006; 34: 1196–1204.
Belancio VP, Roy-Engel AM, Pochampally RR, Deininger P . Somatic expression of LINE-1 elements in human tissues. Nucleic Acids Res 2010; 38: 3909–3922.
Wallace NA, Belancio VP, Deininger PL . L1 mobile element expression causes multiple types of toxicity. Gene 2008; 419: 75–81.
Belgnaoui SM, Gosden RG, Semmes OJ, Haoudi A . Human LINE-1 retrotransposon induces DNA damage and apoptosis in cancer cells. Cancer Cell Int 2006; 6: 13.
Haoudi A, Semmes OJ, Mason JM, Cannon RE . Retrotransposition-Competent Human LINE-1 Induces Apoptosis in Cancer Cells With Intact p53. J Biomed Biotechnol 2004; 2004: 185–194.
Xi L, Cech TR . Inventory of telomerase components in human cells reveals multiple subpopulations of hTR and hTERT. Nucleic Acids Res 2014; 42: 8565–8577.
deLange T . How shelterin solves the telomere end-protection problem. Cold Spring Harbor Symp Quant Biol 2010; 75: 167–177.
Qian Y, Yang L, Cao S . Telomeres and telomerase in T cells of tumor immunity. Cell Immunol 2014; 289: 63–69.
Han JS, Boeke JD . LINE-1 retrotransposons: modulators of quantity and quality of mammalian gene expression? Bioassays 2005; 27: 775–784.
Sciamanna I, Gualtieri A, Cossetti C, Osimo EF, Ferracin M, Macchia G, Aricò E, Prosseda G, Vitullo P et al. A tumor-promoting mechanism mediated by retrotransposon-encoded reverse transcriptase is active in human transformed cell lines. Oncotarget 2013; 4: 2271–2287.
Cesare AJ, Hayashi MT, Crabbe L, Karlseder J . The telomere deprotection response is functionally distinct from the genomic DNA damage response. Mol Cell 2013; 51: 141–155.
Ohka F, Natsume A, Motomura K, Kishida Y, Kondo Y, Abe T, Nakasu Y et al. The global DNA methylation surrogate LINE-1 methylation is correlated with MGMT promoter methylation and is a better prognostic factor for glioma. PLoS One 2011; 6: e23332.
Dai L, Huang Q, Boeke JD . Effect of reverse transcriptase inhibitors on LINE-1 and Ty1 reverse transcriptase activities and on LINE-1 retrotransposition. BMC Biochem 2011; 12: 18.
Grandjenette C, Schnekenburger M, Karius T, Ghelfi J, Gaigneaux A, Henry E et al. 5-Aza-2'-deoxycytidine-mediated c-myc down-regulation triggers telomere-dependent senescence by regulating human telomerase reverse transcriptase in chronic myeloid leukemia. Neoplasia 2014; 16: 511–528.
Sampl S, Pramhas S, Stern C, Preusser M, Marosi C, Holzmann K . Expression of telomeres in astrocytoma WHO grade 2 to 4: TERRA level correlates with telomere length, telomerase activity, and advanced clinical grade. Transl Oncol 2012; 5: 56–65.
Pfaffl MW . A new mathematical model for relative quantification in realtime RTPCR. Nucleic Acid Res 2011; 29: e45.
Montoya-Durango DE, Liu Y, Teneng I, Kalbfleisch T, Lacy ME, Steffen MC et al. Epigenetic control of mammalian LINE-1 retrotransposon by retinoblastoma proteins. Mutat Res 2009; 665: 20–28.
Guiducci C, Anglana M, Wang A, Bacchetti S . Transient expression of wild-type or biologically inactive telomerase allows the formation of artificial telomeres in mortal human cells. Exp Cell Res 2001; 265: 304–311.
Grusch M, Losert A, Lackner A, Deli A, Herbacek I, Holzmann K . Microscopic analysis of adenoviral decontamination using GFP adenovirus with comparable sensitivity to flow cytometry. Methods Mol Biol 2009; 515: 125–135.
Aschacher T, Sampl S, Käser L, Bernhard D, Spittler A, Holzmann K et al. The combined use of known antiviral reverse transcriptase inhibitors AZT and DDI induce anticancer effects at low concentrations. Neoplasia 2012; 14: 44–53.
Acknowledgements
We want to thank Roger Reddel and Bert Vogelstein for providing cell lines, Silvia Bacchetti for adenovirus expressing hTERT, Satoru Kyo for hTERT promoter constructs. Furthermore, we appreciate the critical discussion with members of the Surgical Research Laboratories of the Medical University of Vienna, as well as with Igor Bondarev, Roger Reddel, Berthold Streubl and Wolfgang Köstler. Further, we appreciate the critical reading of the manuscript by Walter Berger and Christine Brostjan. SS and KH were supported by the Medical Scientific Fund of the Mayor of Vienna #10091.
Author Contributions
TA performed study plan, most of the experiments and data analysis. FE performed reporter gene assays and helped with all cell culture experiments. TA, FE, PK and SS performed real-time PCR experiments and cell cycle analysis. DM and MS helped with primer design for LINE-1 sequences, and stem cell gene expression analysis. BW helped with experimental design for TRAP-assays and telomere-specific Southern blot. KH helped with experimental design and provided use of adenoviruses. MB initiated, directed and supported the work through all levels of development. TA and MB wrote the manuscript that was revised by all the authors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Aschacher, T., Wolf, B., Enzmann, F. et al. LINE-1 induces hTERT and ensures telomere maintenance in tumour cell lines. Oncogene 35, 94–104 (2016). https://doi.org/10.1038/onc.2015.65
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2015.65
This article is cited by
-
Impact of retrotransposon protein L1 ORF1p expression on oncogenic pathways in hepatocellular carcinoma: the role of cytoplasmic PIN1 upregulation
British Journal of Cancer (2023)
-
A pilot study of LINE-1 copy number and telomere length with aging in human sperm
Journal of Assisted Reproduction and Genetics (2023)
-
Telomerase gene therapy: a remission toward cancer
Medical Oncology (2022)
-
High levels of LINE-1 transposable elements expressed in Kaposi’s sarcoma-associated herpesvirus-related primary effusion lymphoma
Oncogene (2021)
-
Cancer gene therapy by NF-κB-activated cancer cell-specific expression of CRISPR/Cas9 targeting telomeres
Gene Therapy (2020)