Key Points
-
Glycans, which decorate all eukaryotic cell surfaces, undergo changes in structure with the onset of diseases such as cancer and inflammation. This article highlights some examples of disease-associated glycans and the possibility of exploiting these glycans for therapeutic or diagnostic strategies.
-
Cancer-associated changes in glycosylation include both the under- and overexpression of naturally-occurring glycans as well as neoexpression of glycans normally restricted to embryonic tissues. These structures most often arise from changes in the expression levels of glycosylating enzymes (glycosyltransferases and glycosidases) in cancerous versus healthy cells.
-
To dissect the roles of glycans in metastasis and tumour formation, cellular glycans have been structurally perturbed in a number of ways. The general conclusion of these studies is that certain glycans seem to play a role in cancer progression.
-
Given the functional link between aberrant glycosylation and malignancy, therapeutics that block the formation of cancer-associated glycans might have an effect on tumour progression. The immune system can be recruited to target cancer cells on the basis of their altered glycosylation.
-
Several glycan-based vaccines are presently undergoing clinical evaluation with some encouraging preliminary results.
-
Existing diagnostic methods used to monitor tumour-specific glycosylation require surgical biopsy followed by histological analysis with lectins or monoclonal antibodies. An interesting future direction in the field is to target aberrant glycosylation with probes for non-invasive imaging.
-
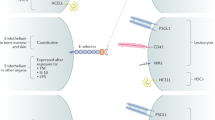
Specific carbohydrate epitopes, such as 6-sulpho sialyl Lewis x, initiate leukocyte homing to sites of chronic inflammation by enabling leukocyte-endothelial cell adhesion via the leukocyte receptor L-selectin and are specifically expressed at disease sites.
-
Drugs that block the selectins or the biosynthesis of their glycan ligands are under investigation in the pharmaceutical industry. In addition, there is an opportunity for the development of noninvasive diagnostics that might identify sites of chronic inflammation prior to the presentation of disease symptoms.
Abstract
Changes in glycosylation are often a hallmark of disease states. For example, cancer cells frequently display glycans at different levels or with fundamentally different structures than those observed on normal cells. This phenomenon was first described in the early 1970s, but the molecular details underlying such transformations were poorly understood. In the past decade advances in genomics, proteomics and mass spectrometry have enabled the association of specific glycan structures with disease states. In some cases, the functional significance of disease-associated changes in glycosylation has been revealed. This review highlights changes in glycosylation associated with cancer and chronic inflammation and new therapeutic and diagnostic strategies that are based on the underlying glycobiology.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Fabbro, D. & Garcia-Echeverria, C. Targeting protein kinases in cancer therapy. Curr. Opin. Drug Disc. Dev. 5, 701–712 (2002).
Sliva, D. Signaling pathways responsible for cancer cell invasion as targets for cancer therapy. Curr. Cancer Drug Targ. 4, 327–336 (2004).
Palladino, M. A., Bahjat, F. R., Theodorakis, E. A. & Moldawer, L. L. Anti-TNF-alpha therapies: The next generation. Nat. Rev. Drug Disc. 2, 736–746 (2003).
Meezan, E., Wu, H. C., Black, P. H. & Robbins, P. W. Comparative studies on carbohydrate-containing membrane components of normal and virus-transformed mouse fibroblasts. Separation of glycoproteins and glycopeptides by sephadex chromatography. Biochemistry 8, 2518–2524 (1969). The first demonstration that cancer glycans differ from glycans on healthy cells.
Turner, G. A. N-Glycosylation of serum-proteins in disease and its investigation using lectins. Clin. Chim. Acta 208, 149–171 (1992).
Axford, J. S. Glycosylation and rheumatic disease. Biochim. Biophys. Acta 1455, 219–229 (1999).
Mackiewicz, A. & Mackiewicz, K. Glycoforms of serum α1-acid glycoprotein as markers of inflammation and cancer. Glycoconj. J. 12, 241–247 (1995).
Gabius, H. J. Biological information transfer beyond the genetic code: the sugar code. Natur Wissenschaften 87, 108–121 (2000).
Saussez, S. et al. Quantitative glycohistochemistry defines new prognostic markers for cancers of the oral cavity. Cancer 82, 252–260 (1998).
Shriver, Z., Raguram, S. & Sasisekharan, R. Glycomics: a pathway to a class of new and improved therapeutics. Nat. Rev. Drug Disc. 3, 863–873 (2004).
Pancino, G. et al. Purification and characterization of a breast-cancer-associated glycoprotein not expressed in normal breast and identified by monoclonal antibody-83d4. Brit. J. Cancer 63, 390–398 (1991).
Matsushita, Y., Cleary, K. R., Ota, D. M., Hoff, S. D. & Irimura, T. Sialyl-dimeric Lewis-X antigen expressed on mucin-like glycoproteins in colorectal-cancer metastases. Lab. Invest. 63, 780–791 (1990).
Dennis, J. W., Laferte, S., Waghorne, C., Breitman, M. L. & Kerbel, R. S. β1-6 branching of asn-linked oligosaccharides is directly associated with metastasis. Science 236, 582–585 (1987).
Kim, Y. J. & Varki, A. Perspectives on the significance of altered glycosylation of glycoproteins in cancer. Glycoconj. J. 14, 569–576 (1997).
Sell, S. Cancer-associated carbohydrates identified by monoclonal antibodies. Human Path. 21, 1003–1019 (1990).
Hakomori, S. & Zhang, Y. Glycosphingolipid antigens and cancer therapy. Chem. Biol. 4, 97–104 (1997).
Taylorpapadimitriou, J. & Epenetos, A. A. Exploiting altered glycosylation patterns in cancer- progress and challenges in diagnosis and therapy. Trends Biotech. 12, 227–233 (1994).
Gabius, H. J. Tumor lectinology- at the intersection of carbohydrate chemistry, biochemistry, cell biology, and oncology. Angew. Chem. 27, 1267–1276 (1988).
Orntoft, T. F. & Vestergaard, E. M. Clinical aspects of altered glycosylation of glycoproteins in cancer. Electrophoresis 20, 362–371 (1999).
Hollingsworth, M. A. & Swanson, B. J. Mucins in cancer: Protection and control of the cell surface. Nat. Rev. Cancer 4, 45–60 (2004).
Hakomori, S. Traveling for the glycosphingolipid path. Glycoconj. J. 17, 627–647 (2000).
Zhang, S. L. et al. Selection of tumor antigens as targets for immune attack using immunohistochemistry. 1. Focus on gangliosides. Int. J. Cancer 73, 42–49 (1997).
Zhang, S. L. et al. Selection of tumor antigens as targets for immune attack using immunohistochemistry. 2. Blood group–related antigens. Int. J. Cancer 73, 50–56 (1997).
Fukuda, M. Possible roles of tumor-associated carbohydrate antigens. Cancer Res. 56, 2237–44 (1996).
Jurianz, K. et al. Complement resistance of tumor cells: Basal and induced mechanisms. Mol. Immunol. 36, 929–939 (1999).
Speiser, D. E. et al. Self antigens expressed by solid tumors do not efficiently stimulate naive or activated T cells: Implications for immunotherapy. J. Exp. Med. 186, 645–653 (1997).
Danishefsky, S. J. & Allen, J. R. From the laboratory to the clinic: A retrospective on fully synthetic carbohydrate-based anticancer vaccines. Angew. Chem. 39, 836–863 (2000). An excellent review of synthetic carbohydrate-based anticancer vaccines.
Livingston, P. O. Approaches to augmenting the immunogenicity of melanoma gangliosides- from whole melanoma-cells to ganglioside-KLH conjugate vaccines. Immunol. Rev. 145, 147–166 (1995).
Ragupathi, G. et al. On the power of chemical synthesis: Immunological evaluation of models for multiantigenic carbohydrate-based cancer vaccines. Proc. Nat'l Acad. Sci. U. S. A. 99, 13699–13704 (2002).
Gilewski, T. et al. Immunization of metastatic breast cancer patients with a fully synthetic globe H conjugate: A phase I trial. Proc. Nat'l Acad. Sci. U. S. A. 98, 3270–3275 (2001).
Slovin, S. F. et al. Carbohydrate vaccines in cancer: Immunogenicity of a fully synthetic globo H hexasaccharide conjugate in man. Proc. Nat'l Acad. Sci. U. S. A. 96, 5710–5715 (1999).
Chapman, P. B. et al. A phase II trial comparing five dose levels of BEC2 anti-idiotypic monoclonal antibody vaccine that mimics GD3 ganglioside. Vaccine 22, 2904–2909 (2004).
Krug, L. M. et al. Vaccination of small cell lung cancer patients with polysialic acid or N-propionylated polysialic acid conjugated to keyhole limpet hemocyanin. Clin. Cancer Res. 10, 916–923 (2004). The first demonstration in humans that vaccines containing unnatural sugars mount a more robust immune response against cancer glycans than their natural counterparts.
Ragupathi, G. et al. Consistent antibody response against ganglioside GD2 induced in patients with melanoma by a GD2 lactone-keyhole limpet hemocyanin conjugate vaccine plus immunological adjuvant QS-21. Clin. Cancer Res. 9, 5214–5220 (2003).
Dove, A. The bittersweet promise of glycobiology. Nat. Biotech. 19, 913–917 (2001).
Holmberg, L. & Sandmaier, B. Vaccination with Theratope (STn-KLH) as treatment for breast cancer. Expert Rev. Vaccines 3, 655–663 (2004).
Livingston, P. O. The unfulfilled promise of melanoma vaccines. Clin. Cancer Res. 7, 1837–1838 (2001).
Slovin, S. F. et al. Fully synthetic carbohydrate-based vaccines in biochemically relapsed prostate cancer: Clinical trial results with α-N-acetylgalactosamine-O-serine/threonine conjugate vaccine. J. Clin. Oncol. 21, 4292–4298 (2003).
Liu, T. M., Guo, Z. W., Yang, Q. L., Sad, S. & Jennings, H. J. Biochemical engineering of surface α2,8 polysialic acid for immunotargeting tumor cells. J. Biol. Chem. 275, 32832–32836 (2000). The first report on the use of unnatural sialic acid biosynthesis for tumour immunotherapy in animals.
Chefalo, P., Pan, Y. B., Nagy, N., Harding, C. & Guo, Z. W. Preparation and immunological studies of protein conjugates of N-acylneuraminic acids. Glycoconj. J. 20, 407–414 (2003).
Pan, Y. B., Chefalo, P., Nagy, N., Harding, C. & Guo, Z. W. Synthesis and immunological properties of N-modified GM3 antigens as therapeutic cancer vaccines. J. Med. Chem. 48, 875–883 (2005).
Zou, W. et al. Bioengineering of surface GD3 ganglioside for immunotargeting human melanoma cells. J. Biol. Chem. 279, 25390–25399 (2004).
Lemieux, G. A. & Bertozzi, C. R. Modulating cell surface immunoreactivity by metabolic induction of unnatural carbohydrate antigens. Chem. Biol. 8, 265–275 (2001).
Galili, U. Autologous tumor vaccines processed to express α-gal epitopes: a practical approach to immunotherapy in cancer. Cancer Immunol. Immunother. 53, 935–945 (2004).
Galili, U., Chen, Z. C. & DeGeest, K. Expression of α-gal epitopes on ovarian carcinoma membranes to be used as a novel autologous tumor vaccine. Gynecol. Oncol. 90, 100–108 (2003).
Deriy, L., Chen, Z. C., Gao, G. P. & Galili, U. Expression of α-gal epitopes on HeLa cells transduced with adenovirus containing α1,3galactosyltransferase cDNA. Glycobiology 12, 135–144 (2002).
Deriy, L., Ogawa, H., Gao, G. P. & Galili, U. In vivo targeting of vaccinating tumor cells to antigen—presenting cells by a gene therapy method with adenovirus containing the α1,3galactosyltransferase gene. Cancer Gene Ther. (in press).
Burchell, J. M., Mungul, A. & Taylor-Papadimitriou, J. O-linked glycosylation in the mammary gland: Changes that occur during malignancy. J. Mamm. Gland Biol. Neopl. 6, 355–364 (2001).
Wong, N. K. et al. Characterization of the oligosaccharides associated with the human ovarian tumor marker CA125. J. Biol. Chem. 278, 28619–28634 (2003).
Basu, P. S., Majhi, R. & Batabyal, S. K. Lectin and serum-PSA interaction as a screeninng test for prostate cancer. Clin. Biochem. 36, 373–376 (2003).
Peracaula, R. et al. Altered glycosylation pattern allows the distinction between prostate-specifc antigen (PSA) from normal and tumor origins. Glycobiology 13, 457–470 (2003).
MacDonald, J. S. Carcinoembryonic antigen screening: Pros and cons. Sem. Oncol. 26, 556–560 (1999).
Lloyd, K. O., Burchell, J., Kudryashov, V., Yin, B. W. & Taylor-Papadimitriou, J. Comparison of O-linked carbohydrate chains in MUC-1 mucin from normal breast epithelial cell lines and breast carcinoma cell lines. Demonstration of simpler and fewer glycan chains in tumor cells. J. Biol. Chem. 271, 33325–34 (1996).
Dalziel, M. et al. The relative activities of the C2GnT1 and ST3Gal-I glycosyltransferases determine O-glycan structure and expression of a tumor-associated epitope on MUC1. J. Biol. Chem. 276, 11007–11015 (2001).
Ramanathan, R. K. et al. Phase I study of a MUC1 vaccine composed of different doses of MUC1 peptide with SB-AS2 adjuvant in resected and locally advance pancreatic cancer. Cancer Immunol. Immunother. 54, 254–264 (2005).
Karanikas, V. et al. Antibody and T cell responses of patients with adenocarcinoma immunized with mannan-MUC1 fusion protein. J. Clin. Invest. 100, 2783–2792 (1997).
Xing, P. X. et al. Phase I study of synthetic MUC1 peptides in cancer. Int. J. Oncol. 6, 1283–1289 (1995).
Renkonen, J., Paavonen, T. & Renkonen, R. Endothelial and epithelial expression of sialyl Lewis(x) and sialyl Lewis(a) in lesions of breast carcinoma. Int. J. Cancer 74, 296–300 (1997).
Miyake, M., Taki, T., Hitomi, S. & Hakomori, S. Correlation of expression of H/Le(Y)/Le(B) antigens with survival in patients with carcinoma of the lung. New Eng. J. Med. 327, 14–18 (1992).
Nakamori, S. et al. Increased expression of sialyl Lewis(X) antigen correlates with poor survival in patients with colorectal-carcinoma- clinicopathological and immunohistochemical study. Cancer Res. 53, 3632–3637 (1993).
Shimodaira, K. et al. Carcinoma-associated expression of core 2 β1,6-N-acetylglucosaminyltransferase gene in human colorectal cancer: Role of O-glycans in tumor progression. Cancer Res. 57, 5201–5206 (1997).
Gorelik, E., Galili, U. & Raz, A. On the role of cell surface carbohydrates and their binding proteins (lectins) in tumor metastasis. Cancer Metast. Rev. 20, 245–277 (2001).
Guo, H. B., Lee, I., Kamar, M. & Pierce, M. N-Acetylglucosaminyltransferase V expression levels regulate cadherin-associated homotypic cell-cell adhesion and intracellular signaling pathways. J. Biol. Chem. 278, 52412–52424 (2003).
Demetriou, M., Nabi, I. R., Coppolino, M., Dedhar, S. & Dennis, J. W. Reduced contact-inhibition and substratum adhesion in epithelial-cells expressing GlcNAc-transferase-V. J. Cell Biol. 130, 383–392 (1995).
Stanley, P. Selection of lectin-resistant mutants of animal-cells. Meth. Enz. 96, 157–184 (1983).
Tao, T. W. & Burger, M. M. Non-metastasizing variants selected from metastasizing melanoma cells. Nature 270, 437–438 (1977).
Tokuyama, S. et al. Suppression of pulmonary metastasis in murine B16 melanoma cells by transfection of a sialidase cDNA. Int. J. Cancer 73, 410–415 (1997).
Granovsky, M. et al. Suppression of tumor growth and metastasis in Mgat5-deficient mice. Nature Med. 6, 306–312 (2000). This paper describes reduced mammary tumour growth and metastasis in Mgat5−/− mice than in transgenic littermates expressing Mgat5, supporting the role of the branching β1,6GlcNAc residue in cancer progression.
Tang, D. G. & Honn, K. V. Adhesion molecules and tumor-metastasis- an update. Inv. Metast. 14, 109–122 (1994).
McEver, R. P. Selectin-carbohydrate interactions during inflammation and metastasis. Glycoconj. J. 14, 585–591 (1997).
Foster, M. M., Brown, J. R., Wang, L. C. & Esko, J. D. A disaccharide precursor of sialyl Lewis X inhibits metastatic potential of tumor cells. Cancer Res. 63, 2775–2781 (2003). A description of a small molecule glycosylation inhibitor capable of decreasing metastasis in a murine tumour model.
Sarkar, A. K., Rostand, K. S., Jain, R. K., Matta, K. L. & Esko, J. D. Fucosylation of disaccharide precursors of sialyl Lewis(X) inhibit selectin-mediated cell adhesion. J. Biol. Chem. 272, 25608–25616 (1997).
Brown, J. R., Fuster, M. M., Whisenant, T. & Esko, J. D. Expression patterns of α 2,3-sialyltransferases and α1,3-fucosyltransferases determine the mode of sialyl Lewis X inhibition by disaccharide decoys. J. Biol. Chem. 278, 23352–23359 (2003).
Alper, J. Glycobiology- Turning sweet on cancer. Science 301, 159–160 (2003).
Borsig, L. et al. Heparin and cancer revisited: Mechanistic connections involving platelets, P-selectin, carcinoma mucins, and tumor metastasis. Proc. Nat'l Acad. Sci. U. S. A. 98, 3352–3357 (2001).
Borsig, L., Kim, Y. J., Varki, N. & Varki, A. The role of P-selectin and carcinoma mucins in tumor growth and metastasis. Glycobiology 8, 1143–1143 (1998). This report showed that heparin inhibits metastasis by interrupting glycan-mediated platelet tumour cell adhesion.
Kim, Y. J., Borsig, L., Varki, N. M. & Varki, A. P-selectin deficiency attenuates tumor growth and metastasis. Proc. Nat'l Acad. Sci. U. S. A. 95, 9325–9330 (1998). The authors demonstrate that P-selectin-deficient mice showed significantly slower growth of subcutaneously implanted human colon carcinoma cells and generated fewer lung metastases from intravenously injected cells than P selectin-expressing littermates.
Shirota, K., Kato, Y., Irimura, T., Konda, H. & Sugiyama, Y. Anti-metastatic effect of the sialyl lewis-X analog GSC-150 on the human colon carcinoma derived cell line KM12-HX in the mouse. Biol. Pharm. Bull. 24, 316–319 (2001).
Zacharski, L. R. & Ornstein, D. L. Heparin and cancer. Thromb. Haemost. 80, 10–23 (1998).
Nitti, D. et al. Final results of a phase III clinical trial on adjuvant intraportal infusion with heparin and 5-fluorouracil (5-FU) in resectable colon cancer. European Journal of Cancer 33, 1209–1215 (1997).
Weissleder, R. Scaling down imaging: molecular mapping of cancer in mice. Nat. Rev. Cancer 2, 11–8 (2002).
Weissleder, R. & Ntziachristos, V. Shedding light onto live molecular targets. Nat. Med. 9, 123–8 (2003).
Gambhir, S. S. Molecular imaging of cancer with positron emission tomography. Nat. Rev. Cancer 2, 683–93 (2002).
Sivolapenko, G. et al. Breast cancer imaging with radiolabelled peptide from complementary–determining region of antitumor immunity. Lancet 346 (1995).
Moore, A., Medarova, Z., Potthast, A. & Dai, G. P. In vivo targeting of underglycosylated MUC-1 tumor antigen using a multimodal imaging probe. Cancer Research 64, 1821–1827 (2004). This report describes noninvasive imaging of breast cancer in a murine tumour model using a peptide that recognizes uMUC1.
Kayser, H. et al. Biosynthesis of a nonphysiological sialic-acid in different rat organs, using N-propanoyl-D-hexosamines as precursors. J. Biol. Chem. 267, 16934–16938 (1992).
Prescher, J. P., Dube, D. H. & Bertozzi, C. R. Chemical remodeling of cell surfaces in living animals. Nature 430, 873–877 (2004). The first demonstration that the Staudinger ligation can tag azide-containing glycans in vivo.
Dube, D. H. & Bertozzi, C. R. Metabolic oligosaccharide engineering as a tool for glycobiology. Curr. Opin. Chem. Biol. 7, 616–625 (2003).
Keppler, O. T., Horstkorte, R., Pawlita, M., Schmidts, C. & Reutter, W. Biochemical engineering of the N-acyl side chain of sialic acid: biological implications. Glycobiology 11, 11R–18R (2001).
Saxon, E. & Bertozzi, C. R. Cell surface engineering by a modified Staudinger reaction. Science 287, 2007–2010 (2000).
Lemieux, G. A., Yarema, K. J., Jacobs, C. L. & Bertozzi, C. R. Exploiting differences in sialoside expression for selective targeting of MRI contrast reagents. J. Am. Chem. Soc. 121, 4278–4279 (1999).
Vocadlo, D. J., Hang, H. C., Kim, E. J., Hanover, J. A. & Bertozzi, C. R. A chemical approach for identifying O-GlcNAc-modified proteins in cells. Proc. Nat'l Acad. Sci. U. S. A. 100, 9116–9121 (2003).
Hang, H. C., Yu, C., Kato, D. L. & Bertozzi, C. R. A metabolic labeling approach toward proteomic analysis of mucin-type O-linked glycosylation. Proc. Nat'l Acad. Sci. U. S. A. 100, 14846–14851 (2003).
Kansas, G. S. Selectins and their ligands: Current concepts and controversies. Blood 88, 3259–3287 (1996).
Renkonen, J., Tynninen, O., Hayry, P., Paavonen, T. & Renkonen, R. Glycosylation might provide endothelial zip codes for organ-specific leukocyte traffic into inflammatory sites. Am. J. Pathol. 161, 543–550 (2002).
Lowe, J. B. Glycan-dependent leukocyte adhesion and recruitment in inflammation. Curr. Opin. Cell Biol. 15, 531–538 (2003).
Ley, K. The role of selectins in inflammation and disease. Trends Mol. Med. 9, 263–268 (2003).
Crocker, P. R. & Feizi, T. Carbohydrate recognition systems: Functional triads in cell-cell interactions. Curr. Opin. Struct. Biol. 6, 679–691 (1996).
Varki, A. Selectin ligands. Proc. Nat'l Acad. Sci. U. S. A. 91, 7390–7397 (1994).
Michie, S. A., Streeter, P. R., Bolt, P. A., Butcher, E. C. & Picker, L. J. The human peripheral lymph-node vascular addressin- an inducible endothelial antigen involved in lymphocyte homing. Am. J. Pathol. 143, 1688–1698 (1993).
Rosen, S. D. Endothelial ligands for L-selectin: From lymphocyte recirculation to allograft rejection. Am. J. Pathol. 155, 1013–1020 (1999).
Kannagi, R. Regulatory roles of carbohydrate ligands for selectins in the homing of lymphocytes. Curr. Opin. Struct. Biol. 12, 599–608 (2002).
Zollner, T. M. & Asadullah, K. Selectin and selectin ligand binding: a bittersweet attraction. J. Clin. Invest. 112, 980–983 (2003).
Khor, S. P., McCarthy, K., Dupont, M., Murray, K. & Timony, G. Pharmacokinetics, pharmacodynamics, allometry, and dose selection of rPSGL-Ig for phase I trial. J. Pharm. Exper. Ther. 293, 618–624 (2000).
Lee, L. V. et al. A potent and highly selective inhibitor of human α1,3-fucosyltransferase via click chemistry. J. Am. Chem. Soc. 125, 9588–9589 (2003).
Lin, C. H. et al. Enzymatic synthesis of a sialyl Lewis X dimer from egg yolk as an inhibitor of E-selectin. Bioorg. Med. Chem. 3, 1625–1630 (1995).
Dimitroff, C. J., Kupper, T. S. & Sackstein, R. Prevention of leukocyte migration to inflamed skin with a novel fluorosugar modifier of cutaneous lymphocyte-associated antigen. J. Clin. Invest. 112, 1008–1018 (2003).
Armstrong, J. I. et al. A library approach to the generation of bisubstrate analoge sulfotransferse inhibitors. Org. Lett. 23, 2657–2660 (2001).
Denis, M. C., Mahmood, U., Benoist, C., Mathis, D. & Weissleder, R. Imaging inflammation of the pancreatic islets in type 1 diabetes. Proc. Nat'l Acad. Sci. U. S. A. 101, 12634–12639 (2004).
Sibson, N. R. et al. MRI detection of early endothelial activation in brain inflammation. Mag. Reson. Med. 51, 248–252 (2004).
Schofield, L., Hewitt, M. C., Evans, K., Siomos, M. A. & Seeberger, P. H. Synthetic GPI as a candidate anti-toxic vaccine in a model of malaria. Nature 418, 785–789 (2002).
Verez-Bencomo, V. et al. A synthetic conjugate polysaccharide vaccine against Haemophilus influenzae type b. Science 305, 522–525 (2004).
Mandal, M., Dudkin, V. Y., Geng, X. D. & Danishefsky, S. In pursuit of carbohydrate-based HIV vaccines, Part 1: The total synthesis of hybrid-type gp120 fragments. Angew. Chem. 43, 2557–2561 (2004).
Geng, X. D., Dudkin, V. Y., Mandal, M. & Danishefsky, S. J. In pursuit of carbohydrate-based HIV vaccines, Part 2: The total synthesis of high-mannose-type gp120 fragments-evaluation of strategies directed to maximal convergence. Angew. Chem. 43, 2562–2565 (2004).
Borman, S. Carbohydrate vaccines: novel chemical and enzymatic oligosaccharide synthesis techniques could lead to a new generation of carbohydrate–based vaccine agents. Chem. Eng. News 82, 31–35 (2004).
Knutson, K. L. GMK (Progenics Pharmaceuticals). Curr. Opin. Invest. Drugs 3, 159–164 (2002).
Ragupathi, G. et al. A fully synthetic globo H carbohydrate vaccine induces a focused humoral response in prostate cancer patients: A proof of principle. Angew. Chem. 38, 563–566 (1999).
Krug, L. M. et al. Vaccination of patients with small-cell lung cancer with synthetic fucosyl GM-1 conjugated to keyhole limpet hemocyanin. Clin. Cancer Res. 10, 6094–6100 (2004).
Sabbatini, P. J. et al. Immunization of ovarian cancer patients with a synthetic Lewis(Y)–protein conjugate vaccine: A phase 1 trial. Int. J. Cancer 87, 79–85 (2000).
Vandintherjanssen, A., Pals, S. T., Scheper, R., Breedveld, F. & Meijer, C. Dendritic cells and high endothelial venules in the rheumatoid synovial–membrane. J. Rhematol. 17, 11–17 (1990).
Salmi, M., Granfors, K., Macdermott, R. & Jalkanen, S. Aberrant binding of lamina propria lymphocytes to vascular endothelium in inflammatory bowel diseases. Gastroenterology 106, 596–605 (1994).
Salmi, M. & Jalkanen, S. Regulation of L-selectin expression on cultured bone-marrow leukocytes and their precursors. Eur. J. Immunol. 22, 835–843 (1992).
Duijvestijn, A. M., Kerkhove, M., Bargatze, R. F. & Butcher, E. C. Lymphoid tissue-specific and inflammation-specific endothelial-cell differentiation defined by monoclonal-antibodies. J. Immunol. 138, 713–719 (1987).
Toppila, S., Paavonen, T., Laitinen, A., Laitinen, L. A. & Renkonen, R. Endothelial sulfated sialyl Lewis X glycans, putative L-selectin ligands, are preferentially expressed in bronchial asthma but not in other chronic inflammatory lung diseases. Am. J. Respir. Cell Mol. Biol. 23, 492–498 (2000).
Hanninen, A. et al. Vascular addressins are induced on islet vessels during insulitis in nonobese diabetic mice and are involved in lymphoid-cell binding to islet endothelium. J. Clin. Invest. 92, 2509–2515 (1993).
Turunen, J. P. et al. De-novo expression of endothelial sialyl Lewis(a) and sialyl Lewis(X) during cardiac transplant rejection- superior capacity of a tetravalent sialyl Lewis(X) oligosaccharide in inhibiting L-selectin–dependent lymphocyte adhesion. J. Exp. Med. 182, 1133–1141 (1995).
Slovin, S. et al. Multivalency in a phase II prostate cancer (PC) vaccine trial: Are more antigens better? Proc. Am. Soc. Clin. Oncol. 22, 167 (2003).
Acknowledgements
We thank J. Prescher, M. Paulick, I. Miller and S. Laughlin for critical reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Related links
DATABASES
Entrez Gene
OMIM
Glossary
- GLYCOSYLTRANSFERASES
-
Enzymes that form glycosidic bonds between monosaccharide units.
- GLYCOSIDASES
-
Enzymes that cleave glycosidic bonds.
- LECTINS
-
Carbohydrate-binding proteins.
- NEOEXPRESSION
-
Expression on adult tissues, when expression is normally restricted to embryonic tissues.
- GANGLIOSIDE
-
A glycosphingolipid-containing sialic acid.
- PASSIVE IMMUNIZATION
-
Treatment with an antibody, not actively recruiting the animal's immune response.
- MICROHETEROGENEITY
-
Combination of structures.
- SELECTINS
-
A family of carbohydrate-binding proteins (lectins) expressed on activated platelets (P-selectin) and endothelial cells (P- and E-selectin).
- BIOORTHOGONAL FUNCTIONAL GROUP
-
A chemical moiety that reacts selectively with a reaction partner in a physiological environment yet is inert to the biological milieu.
Rights and permissions
About this article
Cite this article
Dube, D., Bertozzi, C. Glycans in cancer and inflammation — potential for therapeutics and diagnostics. Nat Rev Drug Discov 4, 477–488 (2005). https://doi.org/10.1038/nrd1751
Issue Date:
DOI: https://doi.org/10.1038/nrd1751
This article is cited by
-
A promising target for breast cancer: B7-H3
BMC Cancer (2024)
-
An N-glycome tissue atlas of 15 human normal and cancer tissue types determined by MALDI-imaging mass spectrometry
Scientific Reports (2024)
-
The role of N-glycosylation modification in the pathogenesis of liver cancer
Cell Death & Disease (2023)
-
Targeting galectin-driven regulatory circuits in cancer and fibrosis
Nature Reviews Drug Discovery (2023)
-
Immune regulatory networks coordinated by glycans and glycan-binding proteins in autoimmunity and infection
Cellular & Molecular Immunology (2023)