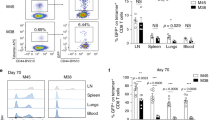
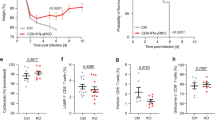
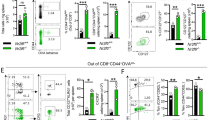
Abstract
Immunization in the absence of CD4+ T cell help results in defective CD8+ T cell memory, deficient recall responses and diminished protective immunity. Here we investigated at what stage during the immune response to pathogen CD4+ T cells are essential in the promotion of functional CD8+ T cell memory. Memory CD8+ T cell numbers decreased gradually in the absence of CD4+ T cells despite the presence of similar numbers of memory cell precursors at the peak of the effector phase. Adoptive transfer of effector or memory CD8+ T cells into wild-type or CD4+ T cell–deficient mice demonstrated that the presence of CD4+ T cells was important only after, not during, the early CD8+ T cell programming phase. In the absence of CD4+ T cells, memory CD8+ T cells became functionally impaired and decreased in quantity over time. We conclude that in the context of an acute infection, CD4+ T cells are required only during the maintenance phase of long-lived memory CD8+ T cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Mercado, R. et al. Early programming of T cell populations responding to bacterial infection. J. Immunol. 165, 6833–6839 (2000).
Kaech, S.M. & Ahmed, R. Memory CD8+ T cell differentiation: initial antigen encounter triggers a developmental program in naive cells. Nat. Immunol. 2, 415–422 (2001).
van Stipdonk, M.J., Lemmens, E.E. & Schoenberger, S.P. Naive CTLs require a single brief period of antigenic stimulation for clonal expansion and differentiation. Nat. Immunol. 2, 423–429 (2001).
van Stipdonk, M.J. et al. Dynamic programming of CD8+ T lymphocyte responses. Nat. Immunol. 4, 361–365 (2003).
Badovinac, V.P., Porter, B.B. & Harty, J.T. Programmed contraction of CD8+ T cells after infection. Nat. Immunol. 3, 619–626 (2002).
Bevan, M.J. & Fink, P.J. The CD8 response on autopilot. Nat. Immunol. 2, 381–382 (2001).
Masopust, D., Kaech, S.M., Wherry, E.J. & Ahmed, R. The role of programming in memory T-cell development. Curr. Opin. Immunol. 16, 217–225 (2004).
Sun, J.C. & Bevan, M.J. Defective CD8 T cell memory following acute infection without CD4 T cell help. Science 300, 339–342 (2003).
Shedlock, D.J. & Shen, H. Requirement for CD4 T cell help in generating functional CD8 T cell memory. Science 300, 337–339 (2003).
Belz, G.T., Wodarz, D., Diaz, G., Nowak, M.A. & Doherty, P.C. Compromised influenza virus-specific CD8+-T-cell memory in CD4+-T-cell-deficient mice. J. Virol. 76, 12388–12393 (2002).
Janssen, E.M. et al. CD4+ T cells are required for secondary expansion and memory in CD8+ T lymphocytes. Nature 421, 852–856 (2003).
Lanzavecchia, A. Immunology. Licence to kill. Nature 393, 413–414 (1998).
Tanchot, C. & Rocha, B. CD8 and B cell memory: same strategy, same signals. Nat. Immunol. 4, 431–432 (2003).
Marrack, P. & Kappler, J. Control of T cell viability. Annu. Rev. Immunol. 22, 765–787 (2004).
Sprent, J. & Surh, C.D. T cell memory. Annu. Rev. Immunol. 20, 551–579 (2002).
Schluns, K.S. & Lefrancois, L. Cytokine control of memory T-cell development and survival. Nat. Rev. Immunol. 3, 269–279 (2003).
Kaech, S.M. et al. Selective expression of the interleukin 7 receptor identifies effector CD8 T cells that give rise to long-lived memory cells. Nat. Immunol. 4, 1191–1198 (2003).
Huster, K.M. et al. Selective expression of IL-7 receptor on memory T cells identifies early CD40L-dependent generation of distinct CD8+ memory T cell subsets. Proc. Natl. Acad. Sci. USA 101, 5610–5615 (2004).
Klonowski, K.D. et al. Dynamics of blood-borne CD8 memory T cell migration in vivo. Immunity 20, 551–562 (2004).
Bourgeois, C., Rocha, B. & Tanchot, C. A role for CD40 expression on CD8+ T cells in the generation of CD8+ T cell memory. Science 297, 2060–2063 (2002).
Khanolkar, A., Fuller, M.J. & Zajac, A.J. CD4 T cell-dependent CD8 T cell maturation. J. Immunol. 172, 2834–2844 (2004).
Bennett, S.R. et al. Help for cytotoxic-T-cell responses is mediated by CD40 signalling. Nature 393, 478–480 (1998).
Ridge, J.P., Di Rosa, F. & Matzinger, P. A conditioned dendritic cell can be a temporal bridge between a CD4+ T-helper and a T-killer cell. Nature 393, 474–478 (1998).
Schoenberger, S.P., Toes, R.E., van der Voort, E.I., Offringa, R. & Melief, C.J. T-cell help for cytotoxic T lymphocytes is mediated by CD40-CD40L interactions. Nature 393, 480–483 (1998).
Wang, J.C. & Livingstone, A.M. Cutting edge: CD4+ T cell help can be essential for primary CD8+ T cell responses in vivo. J. Immunol. 171, 6339–6343 (2003).
Sun, J.C. & Bevan, M.J. Cutting edge: long-lived CD8 memory and protective immunity in the absence of CD40 expression on CD8 T cells. J. Immunol. 172, 3385–3389 (2004).
Le Bon, A. et al. Cross-priming of CD8+ T cells stimulated by virus-induced type I interferon. Nat. Immunol. 4, 1009–1015 (2003).
Lee, B.O., Hartson, L. & Randall, T.D. CD40-deficient, influenza-specific CD8 memory T cells develop and function normally in a CD40-sufficient environment. J. Exp. Med. 198, 1759–1764 (2003).
Jameson, S.C. Maintaining the norm: T-cell homeostasis. Nat. Rev. Immunol. 2, 547–556 (2002).
Prlic, M., Lefrancois, L. & Jameson, S.C. Multiple choices: regulation of memory CD8 T cell generation and homeostasis by interleukin (IL)-7 and IL-15. J. Exp. Med. 195, F49–F52 (2002).
Schluns, K.S., Kieper, W.C., Jameson, S.C. & Lefrancois, L. Interleukin-7 mediates the homeostasis of naive and memory CD8 T cells in vivo. Nat. Immunol. 1, 426–432 (2000).
Goldrath, A.W. et al. Cytokine requirements for acute and basal homeostatic proliferation of naive and memory CD8+ T cells. J. Exp. Med. 195, 1515–1522 (2002).
Tan, J.T. et al. Interleukin (IL)-15 and IL-7 jointly regulate homeostatic proliferation of memory phenotype CD8+ cells but are not required for memory phenotype CD4+ cells. J. Exp. Med. 195, 1523–1532 (2002).
Kieper, W.C. et al. Overexpression of interleukin (IL)-7 leads to IL-15-independent generation of memory phenotype CD8+ T cells. J. Exp. Med. 195, 1533–1539 (2002).
Becker, T.C. et al. Interleukin 15 is required for proliferative renewal of virus-specific memory CD8 T cells. J. Exp. Med. 195, 1541–1548 (2002).
Schluns, K.S., Williams, K., Ma, A., Zheng, X.X. & Lefrancois, L. Cutting edge: requirement for IL-15 in the generation of primary and memory antigen-specific CD8 T cells. J. Immunol. 168, 4827–4831 (2002).
Judge, A.D., Zhang, X., Fujii, H., Surh, C.D. & Sprent, J. Interleukin 15 controls both proliferation and survival of a subset of memory-phenotype CD8+ T cells. J. Exp. Med. 196, 935–946 (2002).
Lodolce, J.P. et al. IL-15 receptor maintains lymphoid homeostasis by supporting lymphocyte homing and proliferation. Immunity 9, 669–676 (1998).
Ku, C.C., Murakami, M., Sakamoto, A., Kappler, J. & Marrack, P. Control of homeostasis of CD8+ memory T cells by opposing cytokines. Science 288, 675–678 (2000).
Schluns, K.S., Klonowski, K.D. & Lefrancois, L. Transregulation of memory CD8 T-cell proliferation by IL-15Rα+ bone marrow-derived cells. Blood 103, 988–994 (2004).
Burkett, P.R. et al. IL-15Rα expression on CD8+ T cells is dispensable for T cell memory. Proc. Natl. Acad. Sci. USA 100, 4724–4729 (2003).
Schluns, K.S. et al. Distinct cell types control lymphoid subset development by means of IL-15 and IL-15 receptorα expression. Proc. Natl. Acad. Sci. USA 101, 5616–5621 (2004).
Dubois, S., Mariner, J., Waldmann, T.A. & Tagaya, Y. IL-15Rα recycles and presents IL-15 in trans to neighboring cells. Immunity 17, 53–547 (2002).
Shen, H. et al. Recombinant Listeria monocytogenes as a live vaccine vehicle for the induction of protective anti-viral cell-mediated immunity. Proc. Natl. Acad. Sci. USA 92, 3987–3991 (1995).
Altman, J.D. et al. Phenotypic analysis of antigen-specific T lymphocytes. Science 274, 94–96 (1996).
Murali-Krishna, K. et al. Counting antigen-specific CD8 T cells: a reevaluation of bystander activation during viral infection. Immunity 8, 177–187 (1998).
Acknowledgements
Supported by the National Institutes of Health (AI19335, AI056809 and CA09537) and the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
GP(33-41)-specific CD8+ T cell numbers in the liver and lymph nodes of MHC class II-deficient and wild-type mice following infection. (PDF 22 kb)
Supplementary Fig. 2
Changes in surface phenotype and cytokine production in CD8+ T cells during the effector and memory phases in wild-type and MHC class II-deficient mice. (PDF 60 kb)
Rights and permissions
About this article
Cite this article
Sun, J., Williams, M. & Bevan, M. CD4+ T cells are required for the maintenance, not programming, of memory CD8+ T cells after acute infection. Nat Immunol 5, 927–933 (2004). https://doi.org/10.1038/ni1105
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1105
This article is cited by
-
Activin-A impedes the establishment of CD4+ T cell exhaustion and enhances anti-tumor immunity in the lung
Journal of Experimental & Clinical Cancer Research (2021)
-
Eosinopenia and neutrophil-to-lymphocyte count ratio as prognostic factors in exacerbation of COPD
Scientific Reports (2021)
-
cDC1 prime and are licensed by CD4+ T cells to induce anti-tumour immunity
Nature (2020)
-
Memory T cells: strategies for optimizing tumor immunotherapy
Protein & Cell (2020)
-
Metabolomics and transcriptomics pathway approach reveals outcome-specific perturbations in COPD
Scientific Reports (2018)