Abstract
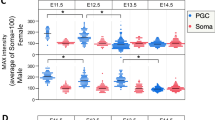
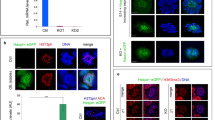
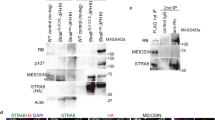
The transition from mitosis to meiosis is a defining juncture in the life cycle of sexually reproducing organisms. In yeast, the decision to enter meiosis is made before the single round of DNA replication that precedes the two meiotic divisions1. We present genetic evidence of an analogous decision point in the germ line of a multicellular organism. The mouse Stra8 gene is expressed in germ cells of embryonic ovaries, where meiosis is initiated, but not in those of embryonic testes, where meiosis does not begin until after birth2. Here we report that in female embryos lacking Stra8 gene function, the early, mitotic development of germ cells is normal, but these cells then fail to undergo premeiotic DNA replication, meiotic chromosome condensation, cohesion, synapsis and recombination. Combined with previous findings, these genetic data suggest that active differentiation of ovarian germ cells commences at a regulatory point upstream of premeiotic DNA replication.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Marston, A.L. & Amon, A. Meiosis: cell-cycle controls shuffle and deal. Nat. Rev. Mol. Cell Biol. 5, 983–997 (2004).
Menke, D.B., Koubova, J. & Page, D.C. Sexual differentiation of germ cells in XX mouse gonads occurs in an anterior-to-posterior wave. Dev. Biol. 262, 303–312 (2003).
McLaren, A. & Southee, D. Entry of mouse embryonic germ cells into meiosis. Dev. Biol. 187, 107–113 (1997).
Adams, I.R. & McLaren, A. Sexually dimorphic development of mouse primordial germ cells: switching from oogenesis to spermatogenesis. Development 129, 1155–1164 (2002).
Koubova, J. et al. Retinoic acid regulates sex-specific timing of meiotic initiation in mice. Proc. Natl. Acad. Sci. USA 103, 2474–2479 (2006).
Bowles, J. et al. Retinoid signaling determines germ cell fate in mice. Science 312, 596–600 (2006).
Yao, H.H., DiNapoli, L. & Capel, B. Meiotic germ cells antagonize mesonephric cell migration and testis cord formation in mouse gonads. Development 130, 5895–5902 (2003).
Bullejos, M. & Koopman, P. Germ cells enter meiosis in a rostro-caudal wave during development of the mouse ovary. Mol. Reprod. Dev. 68, 422–428 (2004).
Oulad-Abdelghani, M. et al. Characterization of a premeiotic germ cell-specific cytoplasmic protein encoded by Stra8, a novel retinoic acid-responsive gene. J. Cell Biol. 135, 469–477 (1996).
Hartung, M. & Stahl, A. Preleptotene chromosome condensation in mouse oogenesis. Cytogenet. Cell Genet. 18, 309–319 (1977).
Devictor, M., Luciani, J.M. & Stahl, A. Do male germ cells begin meiosis during fetal life? J. Genet. Hum. 27, 21–28 (1979).
Borum, K. Oogenesis in the mouse. A study of the meiotic prophase. Exp. Cell Res. 24, 495–507 (1961).
Speed, R.M. Meiosis in the foetal mouse ovary. I. An analysis at the light microscope level using surface-spreading. Chromosoma 85, 427–437 (1982).
Eijpe, M., Offenberg, H., Jessberger, R., Revenkova, E. & Heyting, C. Meiotic cohesin REC8 marks the axial elements of rat synaptonemal complexes before cohesins SMC1beta and SMC3. J. Cell Biol. 160, 657–670 (2003).
Lee, J., Iwai, T., Yokota, T. & Yamashita, M. Temporally and spatially selective loss of Rec8 protein from meiotic chromosomes during mammalian meiosis. J. Cell Sci. 116, 2781–2790 (2003).
Moens, P.B. & Spyropoulos, B. Immunocytology of chiasmata and chromosomal disjunction at mouse meiosis. Chromosoma 104, 175–182 (1995).
Prieto, I. et al. Cohesin component dynamics during meiotic prophase I in mammalian oocytes. Chromosome Res. 12, 197–213 (2004).
Rogakou, E.P., Pilch, D.R., Orr, A.H., Ivanova, V.S. & Bonner, W.M. DNA double-stranded breaks induce histone H2AX phosphorylation on serine 139. J. Biol. Chem. 273, 5858–5868 (1998).
Pittman, D.L. et al. Meiotic prophase arrest with failure of chromosome synapsis in mice deficient for Dmc1, a germline-specific RecA homolog. Mol. Cell 1, 697–705 (1998).
Yoshida, K. et al. The mouse RecA-like gene Dmc1 is required for homologous chromosome synapsis during meiosis. Mol. Cell 1, 707–718 (1998).
Baudat, F., Manova, K., Yuen, J.P., Jasin, M. & Keeney, S. Chromosome synapsis defects and sexually dimorphic meiotic progression in mice lacking Spo11. Mol. Cell 6, 989–998 (2000).
Romanienko, P.J. & Camerini-Otero, R.D. The mouse Spo11 gene is required for meiotic chromosome synapsis. Mol. Cell 6, 975–987 (2000).
Crone, M., Levy, E. & Peters, H. The duration of the premeiotic DNA synthesis in mouse oocytes. Exp. Cell Res. 39, 678–688 (1965).
Toyooka, Y. et al. Expression and intracellular localization of mouse Vasa-homologue protein during germ cell development. Mech. Dev. 93, 139–149 (2000).
Bannister, L.A., Reinholdt, L.G., Munroe, R.J. & Schimenti, J.C. Positional cloning and characterization of mouse mei8, a disrupted allelle of the meiotic cohesin Rec8. Genesis 40, 184–194 (2004).
Yuan, L. et al. The murine SCP3 gene is required for synaptonemal complex assembly, chromosome synapsis, and male fertility. Mol. Cell 5, 73–83 (2000).
Xu, H., Beasley, M.D., Warren, W.D., van der Horst, G.T. & McKay, M.J. Absence of mouse REC8 cohesin promotes synapsis of sister chromatids in meiosis. Dev. Cell 8, 949–961 (2005).
Brennan, J. & Capel, B. One tissue, two fates: molecular genetic events that underlie testis versus ovary development. Nat. Rev. Genet. 5, 509–521 (2004).
Fujiwara, S. & Kawamura, K. Acquisition of retinoic acid signaling pathway and innovation of the chordate body plan. Zoolog. Sci. 20, 809–818 (2003).
Albrecht, K.H. & Eicher, E.M. Evidence that Sry is expressed in pre-Sertoli cells and Sertoli and granulosa cells have a common precursor. Dev. Biol. 240, 92–107 (2001).
Acknowledgements
We thank R. Jaenisch (Whitehead Institute for Biomedical Research) for v6.5 ES cells; A. Bortvin, T. Hassold and T. Ashley for advice; C. Heyting (Department of Genetics, Agricultural University, Wageningen) for SCP3 and REC8 antisera; T. Noce (Mitsubishi Kagaku Institute of Life Science) for MVH antisera and E. Anderson, G. Baltus, M. Capelson, M. Gill, J. Koubova, J. Lange, Y. Lim, J. Mueller and J. Potash for comments on the manuscript. Microscopy and image capture were conducted in part at the W.M. Keck Foundation Biological Imaging Facility at the Whitehead Institute. A.E.C. is a Novartis Fellow of the Life Sciences Research Foundation. This work was supported by the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Targeted disruption of the Stra8 locus. (PDF 781 kb)
Supplementary Fig. 2
Germ cell loss and reduced gonadal size in Stra8-deficient mice. (PDF 876 kb)
Supplementary Fig. 3
Alignment of predicted amino acid sequences of STRA8 homologs identified electronically. (PDF 105 kb)
Supplementary Table 1
Oligonucleotide sequences. (PDF 35 kb)
Rights and permissions
About this article
Cite this article
Baltus, A., Menke, D., Hu, YC. et al. In germ cells of mouse embryonic ovaries, the decision to enter meiosis precedes premeiotic DNA replication. Nat Genet 38, 1430–1434 (2006). https://doi.org/10.1038/ng1919
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng1919
This article is cited by
-
Multiple ageing effects on testicular/epididymal germ cells lead to decreased male fertility in mice
Communications Biology (2024)
-
Landscape of pathogenic mutations in premature ovarian insufficiency
Nature Medicine (2023)
-
In vitro differentiation of primed human induced pluripotent stem cells into primordial germ cell-like cells
Molecular Biology Reports (2023)
-
PRC1-mediated epigenetic programming is required to generate the ovarian reserve
Nature Communications (2022)
-
Haploidy in somatic cells is induced by mature oocytes in mice
Communications Biology (2022)