Abstract
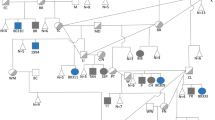
Familial hypercholanemia (FHC) is characterized by elevated serum bile acid concentrations, itching, and fat malabsorption1,2. We show here that FHC in Amish individuals is associated with mutations in tight junction protein 2 (encoded by TJP2, also known as ZO-2) and bile acid Coenzyme A: amino acid N-acyltransferase (encoded by BAAT). The mutation of TJP2, which occurs in the first PDZ domain, reduces domain stability and ligand binding in vitro. We noted a morphological change in hepatic tight junctions. The mutation of BAAT, a bile acid–conjugating enzyme3, abrogates enzyme activity; serum of individuals homozygous with respect to this mutation contains only unconjugated bile acids. Mutations in both TJP2 and BAAT may disrupt bile acid transport and circulation. Inheritance seems to be oligogenic, with genotype at BAAT modifying penetrance in individuals homozygous with respect to the mutation in TJP2.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Morton, D.H. et al. Abnormal hepatic sinusoidal bile acid transport in an Amish kindred is not linked to FIC1 and is improved by ursodiol. Gastroenterology 119, 188–195 (2000).
Shneider, B.L. et al. Hepatic basolateral sodium-dependent-bile acid transporter expression in two unusual cases of hypercholanemia and in extrahepatic biliary atresia. Hepatology 25, 1176–1183 (1997).
Falany, C.N., Johnson, M.R., Barnes, S. & Diasio, R.B. Glycine and taurine conjugation of bile acids by a single enzyme. Molecular cloning and expression of human liver bile acid CoA:amino acid N-acyltransferase. J. Biol. Chem. 269, 19375–19379 (1994).
Letunic, I. et al. Recent improvements to the SMART domain-based sequence annotation resource. Nucleic Acids Res. 30, 242–244 (2002).
Harris, B.Z. & Lim, W.A. Mechanism and role of PDZ domains in signaling complex assembly. J. Cell Sci. 114, 3219–3231 (2001).
Itoh, M. et al. Direct binding of three tight junction-associated MAGUKs, ZO-1, TJP2, and ZO-3, with the COOH termini of claudins. J. Cell Biol. 147, 1351–1363 (1999).
Mitic, L.L., Van Itallie, C.M. & Anderson, J.M. Molecular physiology and pathophysiology of tight junctions I. Tight junction structure and function: lessons from mutant animals and proteins. Am. J. Physiol. Gastrointest. Liver Physiol. 279, G250–G254 (2000).
Jesaitis, L.A. & Goodenough, D.A. Molecular characterization and tissue distribution of ZO-2, a tight junction protein homologous to ZO-1 and the Drosophila discs-large tumor suppressor protein. J. Cell Biol. 124, 949–961 (1994).
Tsukita, S., Furuse, M. & Itoh, M. Multifunctional strands in tight junctions. Nat. Rev. Mol. Cell Biol. 2, 285–293 (2001).
McCarthy, K.M. et al. Inducible expression of claudin-1-myc but not occludin-VSV-G results in aberrant tight junction strand formation in MDCK cells. J. Cell Sci. 113 Pt 19, 3387–3398 (2000).
Heiskala, M., Peterson, P.A. & Yang, Y. The roles of claudin superfamily proteins in paracellular transport. Traffic 2, 93–98 (2001).
Dawson, P.A. Bile Secretion and the Enterohepatic Circulation of Bile Acids. in Gastrointestinal and Liver Disease (eds. Feldman, M., Friedman, L.S. & Sleisenger, M.H.) 1051–1062 (Saunders, Philadelphia, 2002).
Rahner, C., Stieger, B. & Landmann, L. Structure–function correlation of tight junctional impairment after intrahepatic and extrahepatic cholestasis in rat liver. Gastroenterology 110, 1564–1578 (1996).
Koga, A. & Todo, S. Morphological and functional changes in the tight junctions of the bile canaliculi induced by bile duct ligation. Cell Tissue Res. 195, 267–276 (1978).
Setchell, K.D. & O'Connell, N.C. Disorders of Bile Acid Synthesis and Metabolism: A Metabolic Basis for Liver Disease. in Liver Disease in Children (eds. Suchy, F.J., Sokol, R.J. & Balistreri, W.F.) 701–734 (Lippincott Williams & Wilkins, Philadelphia, 2001).
Noe, J., Stieger, B. & Meier, P.J. Functional expression of the canalicular bile salt export pump of human liver. Gastroenterology 123, 1659–1666 (2002).
Ronchi, G. & Desmet, V.J. Histochemical study of gamma glutamyl transpeptidase (GGT) in experimental intrahepatic and extrahepatic cholestasis. Beitr. Pathol. 150, 316–321 (1973).
Mahapatra, A.K. & Suri, A. Anterior encephaloceles: a study of 92 cases. Pediatr. Neurosurg. 36, 113–118 (2002).
Chen, C.M. et al. Polychaetoid is required to restrict segregation of sensory organ precursors from proneural clusters in Drosophila. Mech. Dev. 57, 215–227 (1996).
Colas, J.F. & Schoenwolf, G.C. Towards a cellular and molecular understanding of neurulation. Dev. Dyn. 221, 117–145 (2001).
Bull, L.N. et al. A gene encoding a P-type ATPase mutated in two forms of hereditary cholestasis. Nat. Genet. 18, 219–224 (1998).
Hostetler, J.A. Amish Society (The Johns Hopkins University Press, Baltimore, 1993).
Terwilliger, J.D. A powerful likelihood method for the analysis of linkage disequilibrium between trait loci and one or more polymorphic marker loci. Am J. Hum. Genet. 56, 777–787 (1995).
Hillier, B.J., Christopherson, K.S., Prehoda, K.E., Bredt, D.S. & Lim, W.A. Unexpected modes of PDZ domain scaffolding revealed by structure of nNOS-syntrophin complex. Science 284, 812–815 (1999).
Harris, B.Z., Hillier, B.J. & Lim, W.A. Energetic determinants of internal motif recognition by PDZ domains. Biochemistry 40, 5921–5930 (2001).
Acknowledgements
The authors thank the individuals with FHC and their family members for their participation; I. Herskowitz for support and contributions; S. Service for software and advice; C. Hendrickson, J. Doherty, J. Woo, H. Yoon, E. Song, J. Vargas, R. Senkus, M. Maksimak, E. Cunningham, D. Agard, A. Frankel, V. Calabro, K. Novak and R. K. Guy for advice, assistance and the use of equipment; and N. Risch, N. Freimer, C. Slates, W. Hsueh, A. Slavotinek, R. Kelley and F. Collins for discussions. This work was supported by grants from the US National Institutes of Health, the Howard Hughes Medical Institute, the University of California San Francisco Program in Human Genetics and the Tetrad Graduate Program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Carlton, V., Harris, B., Puffenberger, E. et al. Complex inheritance of familial hypercholanemia with associated mutations in TJP2 and BAAT. Nat Genet 34, 91–96 (2003). https://doi.org/10.1038/ng1147
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng1147
This article is cited by
-
ZO-2/Tjp2 suppresses Yap and Wwtr1/Taz-mediated hepatocyte to cholangiocyte transdifferentiation in the mouse liver
npj Regenerative Medicine (2022)
-
New paradigms of USP53 disease: normal GGT cholestasis, BRIC, cholangiopathy, and responsiveness to rifampicin
Journal of Human Genetics (2021)
-
Impaired bile acid metabolism with defectives of mitochondrial-tRNA taurine modification and bile acid taurine conjugation in the taurine depleted cats
Scientific Reports (2020)