Abstract
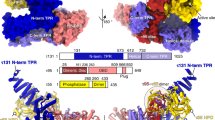
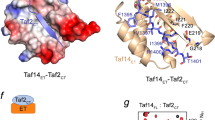
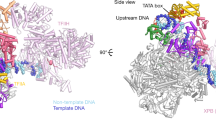
The X-ray crystal structure of the transcription factor IIA (TFIIA)in complex with the TATA-box-binding protein (TBP) and TATA-element DNA is presented at 2.5 Å resolution. TFIIA is composed of a β-barrel and a four-helix bundle motif that together have a boot-like appearance. The β-barrel extends the TBP β-sheet and bridges over the DNA major groove immediately upstream of the TATA box. The four-helix bundle contributes substantially to the surface of the complex available for interaction with additional transcription factors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Maldonado, E. & Reinberg, D. Curr. Opin. Cell Biol. 7, 352–361 (1995).
Hori, R. & Carey, M. Curr. Opin. Genet. Dev. 4, 236–244 (1994).
Conaway, R. C. & Conaway, J. W. A. Rev. Biochem. 62, 161–190 (1993).
Matsui, T., Segall, J., Weil, P. A. & Roeder, R. G. J. biol. Chem. 255, 11992–11996 (1980).
Reinberg, D., Horikoshi, M. & Roeder, R. G. J. biol. Chem. 262, 3322–3330 (1987).
Sayre, M. H., Tschochner, H. & Kornberg, R. D. J. biol. Chem. 267, 23376–23382 (1992).
Cortes, P., Flores, O. & Reinberg, D. Molec. cell. Biol. 12, 413–421 (1992).
Sun, X., Ma, D., Sheldon, M., Yeung, K. & Reinberg, D. Genes Dev. 8, 2336–2348 (1994).
Hansen, S. K. & Tjian, R. Cell 82, 565–575 (1995).
Imbalzano, A. N., Zaret, K. S. & Kingston, R. E. J. biol. Chem. 269, 8280–8286 (1994).
Ranish, J. A. & Hahn, S. J. biol. Chem. 266, 19320–19327 (1991).
Coulombe, B., Li, J. & Greenblatt, J. J. biol. Chem. 269, 19962–19967 (1994).
De Jong, J., Bernstein, R. & Roeder, R. G. Proc. natn. Acad. Sci. U.S.A. 92, 3313–3317 (1995).
Ozer, J. et al. Genes Dev. 8, 2324–2335 (1994).
Yokomori, K. et al. Genes Dev. 8, 2313–2323 (1994).
Inostroza, J. A., Mermelstein, F. H., Ha, I., Lane, W. S. & Reinberg, D. Cell 70, 477–489 (1992).
Auble, D. T. & Hahn, S. Genes Dev. 7, 844–856 (1993).
Wang, W., Gralla, J. D. & Carey, M. Genes Dev. 6, 1716–1727 (1992).
Ge, H. & Roeder, R. G. Cell 78, 513–523 (1994).
Shykind, B. M., Kim, J. & Sharp, P. A. Genes Dev. 9, 1354–1365 (1995).
Ranish, J. A., Lane, W. S. & Hahn, S. Science 255, 1127–1129 (1992).
Stargell, L. A. & Struhl, K. Science 269, 75–78 (1995).
Zeidler, M. P., Yokomori, K., Tjian, R. & Mlodzik, M. Genes Dev. 10, 50–59 (1996).
De Jong, J. & Roeder, R. G. Genes Dev. 7, 2220–2234 (1993).
Ma, D. et al. Genes Dev. 7, 2246–2257 (1993).
Yokomori, K., Admon, A., Goodrich, J. A., Chen, J. L. & Tjian, R. Genes Dev. 7, 2235–2245 (1993).
Kang, J. J., Auble, D. T., Ranish, J. A. & Hahn, S. Molec. cell. Biol. 15, 1234–1243 (1995).
Nikolov, D. B. et al. Nature 360, 40–46 (1992).
Chasman, D. I., Flaherty, K. M., Sharp, P. A. & Kornberg, R. D. Proc. natn. Acad. Sci. U.S.A. 90, 8174–8178 (1993).
Kim, Y., Geiger, J. H., Hahn, S. & Sigler, P. B. Nature 365, 512–520 (1993).
Kim, J. L., Nikolov, D. B. & Burley, S. K. Nature 365, 520–527 (1993).
Nikolov, D. B. et al. Nature 377, 119–128 (1995).
Buratowski, S. & Zhou, H. Science 255, 1130–1132 (1992).
Lee, D. K., DeJong, J., Hashimoto, S., Horikoshi, M. & Roeder, R. G. Molec. cell. Biol. 12, 5189–5196 (1992).
Tang, H., Sun, X., Reinberg, D. & Ebright, R. H. Proc. natn. Acad. Sci. U.S.A. 93, 1119–1124 (1996).
Breathnach, R. & Chambon, P. A. Rev. Biochem. 50, 349–383 (1981).
Hahn, S., Buratowski, S., Sharp, P. A. & Guarente, L. EMBO J. 8, 3379–3382 (1989).
Buratowski, S., Hahn, S., Guarente, L. & Sharp, P. A. Cell 56, 549–561 (1989).
Studier, F. W., Rosenberg, A. H., Dunn, J. J. & Dubendorff, J. W. Meth. Enzym. 185, 60–89 (1990).
Parks, T. D., Leuther, K. K., Howard, E. D., Johnston, S. A. & Dougherty, W. G. Analyt. Biochem. 216, 413–417 (1994).
Otwinowsky, Z. in Isomorphous Replacement and Anomalous Scattering (eds Wolf, W., Evans, P. R. & Leslie, A. G. W.) 80–86 (SERC, Daresbury Laboratory, Warrington, UK, 1991).
Jones, T. A., Zou, J. Y., Cowan, S. W. & Kjeldgaard, M. Acta crystallogr. A47, 110–119 (1991).
Read, R. J. Acta crystallogr. A42, 140–149 (1986).
Brünger, A. X-PLOR Version 3.1 (Yale University, New Haven, CT, 1992).
Tronrud, D. E. Acta crystallogr. A48, 912–916 (1992).
Lavery, R. & Sklenar, H. J. biomol. Struct. Dynam. 6, 63–91 (1988).
Kabsch, W. & Sander, C. Biopolymers 22, 2577–2637 (1983).
Ferrin, T. E., Huang, C. C., Jarvis, L. E. & Langridge, R. J. molec. Graph. 6, 13–27 (1988).
Nicholls, A., Sharp, K. & Honig, B. Proteins 11, 281–296 (1991).
Li, W. & Sherman, F. Molec. cell. Biol. 11, 666–676 (1991).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tan, S., Hunziker, Y., Sargent, D. et al. Crystal structure of a yeast TFIIA/TBP/DNA complex. Nature 381, 127–134 (1996). https://doi.org/10.1038/381127a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/381127a0
This article is cited by
-
Structural convergence endows nuclear transport receptor Kap114p with a transcriptional repressor function toward TATA-binding protein
Nature Communications (2023)
-
Ino2, activator of yeast phospholipid biosynthetic genes, interacts with basal transcription factors TFIIA and Bdf1
Current Genetics (2023)
-
Structural basis of the complete poxvirus transcription initiation process
Nature Structural & Molecular Biology (2021)
-
Molecular determinants underlying functional innovations of TBP and their impact on transcription initiation
Nature Communications (2020)
-
Paused RNA polymerase II inhibits new transcriptional initiation
Nature Genetics (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.