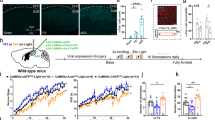
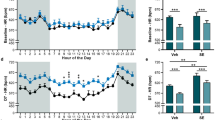
Abstract
Antagonists of γ-aminobutyric acid (GABA)- or glycine-mediated neurotransmission, muscarinic cholinergic agonists, and excitatory amino acids and their analogues are all considered to be potent chemoconvulsant agents. However, although systemic injections of these agents have been used to create experimental models of generalized epilepsy, there has been no identification of a specific locus at which any of these drugs act to initiate generalized seizures. We recently located a forebrain region from which seizures can be elicited by the GABA antagonist bicuculline1, and now report that manipulations of excitatory amino acid transmission and cholinergic transmission can also elicit seizures from this site. Bilateral clonic seizures can be elicited after unilateral application of picomole amounts of bicuculline, kainic acid or carbachol and micromole amounts of glutamate. Local application of the GABA agonist muscimol prevents the appearance of seizures on subsequent microinjection of all convulsant agents examined, whereas local application of the muscarinic antagonist, atropine, only prevents seizures induced by carbachol. This region is therefore a site of action for the epileptogenic effects of neuroactive agents with diverse mechanisms of action; it may also represent a site at which GABA agonists could function therapeutically to control epileptogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Piredda, S., Lim, C. R. & Gale, K. Life Sci. 36, 1295–1298 (1985).
Ben-Ari, Y., Tremblay, E., Riche, E., Glilini, G. & Naquet, R. Neuroscience 6, 1361–1391 (1981).
Evans, M. L. & Meldrum, B. S. Brain Res. 297, 235–245 (1984).
Lothman, E. W., Collins, R. C. & Ferrendelli, J. A. Neurology 31, 806–812 (1981).
Baker, W. W. & Benedict, F. Int. J. Neuropharmac. 7, 135–147 (1968).
Wasterlain, C. G., Masuoka, D. & Jonec, V. in Kindling Vol. 2 (ed. Wada, J. A.) 315–329 (Raven, New York, 1981).
Grossman, S. P. Science 142, 409–410 (1963).
Ben-Ari, Y., Tremblay, E. & Ottersen, O. P. Neuroscience 5, 515–528 (1980).
Schwarcz, R., Zaczek, R. & Coyle, J. Eur. J. Pharmac. 59, 209–220 (1978).
Ben-Ari, Y., Tremblay, E., Ottersen, O. P. & Meldrum, B. S. Brain Res. 191, 79–97 (1980).
Baglioni, S. & Magnini, M. Archs Fisiol. 6, 240–249 (1909).
Labruyere, J., Olney, J. W., Collins, R. C. Soc. Neurosci. Abstr. 1012 (1982).
Baxter, B. L. Expl Neurol. 19, 412–432 (1967).
Vosu, H. & Wise, R. A. Behav. Biol. 13, 491–495 (1975).
Wasterlain, C. G. & Jonec, V. Brain Res. 271, 311–323 (1983).
Racine, R. J. Electroenceph. clin. Neurophys. 32, 281–294 (1972).
Haberly, L. B. & Price, J. L. J. comp. Neurol. 178, 711–740 (1978).
Pellegrino, L. J. & Cushman, A. J. A Stereotaxic Atlas of the Rat Brain (Appleton-Century-Crofts, New York, 1967).
Engel, J. Jr, Wolfson, L. & Brown, L. Ann. Neurol. 3, 538–544 (1978).
Le Gal La Salle, G. in Kindling 2 (ed. Wada, J. A.) 31–47 (Raven, New York, 1981).
McNamara, J. O., Byrne, N. C., Dasheif, R. M. & Fitz, J. G. Prog. Neurobiol. 15, 139–159 (1980).
Racine, R. J. & Burnham, W. M. in Electrophysiology of Epilepsy (eds Schwartzkroin, P. A. & Wheal, H. V.) 153–171 (Academic, London, 1984).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Piredda, S., Gale, K. A crucial epileptogenic site in the deep prepiriform cortex. Nature 317, 623–625 (1985). https://doi.org/10.1038/317623a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/317623a0
This article is cited by
-
Design and synthesis of novel cycloalkanecarboxamide parabanic acid hybrids as anticonvulsants
Medicinal Chemistry Research (2024)
-
Specific contribution of neurons from the Dbx1 lineage to the piriform cortex
Scientific Reports (2021)
-
Quantitative T2 MRI is predictive of neurodegeneration following organophosphate exposure in a rat model
Scientific Reports (2020)
-
Effects of Prolonged Seizures on Basal Forebrain Cholinergic Neurons: Evidence and Potential Clinical Relevance
Neurotoxicity Research (2020)
-
Advances of Molecular Imaging in Epilepsy
Current Neurology and Neuroscience Reports (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.