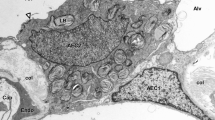
Abstract
A short-term, time-dependent smoke exposure of rats in a nose-only chamber to burning wood and 24-h recovery time revealed inflammation of the airways with varying degrees of injury from loss of cilia, degeneration of epithelium, and squamous metaplasia to submucosal edema. These histological changes were reflected in variable expression of the secretory Muc5AC and low expression of membrane-associated Muc4 mucin genes. 20-min smoke exposure in extended recovery experiments showed marked disorder of tracheal epithelium for up to 72 h of recovery with a return to normal by 7 days. Gene expressions were elevated at 24 and 48 h of recovery. 30-min smoke exposure showed a more severe degeneration of the epithelium and a longer recovery time. Muc5AC expression decreased after 72 h of recovery, while there was upregulation of Muc4 gene from 48 through 96 h. Because Muc4 upregulation and histological results correlate and it has reportedly been associated with epithelium renewal, Muc4 gene may be a useful marker for the regeneration of tracheal epithelium.
Similar content being viewed by others
References
Crapo, R. O. 1981. Smoke inhalation injuries. J. Am. Med. Assoc. 246:1694-1695.
Foley, F. D., J. A. Moncrief, and A. D. Mason. 1968. Pathology of the lung in fatally burned patients. Ann. Surg. 167:251-264.
Moylan, J. A. 1981. Inhalation injury—a primary determinant of survival following major burns. J. Burn Care Rehabil. 2:78-84.
Philips, A. W. and O. Cope. 1962. Burn therapy II. The revelation of respiratory tract damage as a principal killer of the burn patient. Ann. Surg. 155:1-19.
Pruitt, B. A., R. J. Flemma, F. C. Divincenti, and A. D. Mason. 1970. Pulmonary complications in burn patients: A comparative study of 697 patients. J. Thorac. Cardiovasc. Surg. 59:7-20.
Thomson, P. B., D. N. Herndon, and S. Abston. 1986. Effect of mortality of inhalation injury. J. Trauma 26:163.
Niehaus, G. D., R. Kimura, L. D. Traber, D. N. Herndon, J. T. Flynn, and D. L. Traber. 1990. Administration of a synthetic antiprotease reduces smoke-induced lung injury. J. Appl. Physiol. 69:694-699.
Quinn, D. A., D. Robinson, W. Jung, and C. A. Hales. 1990. Role of sulfidopeptide leukotrienes in synthetic smoke inhalation injury in sheep. J. Appl. Physiol. 68:1962-1969.
Riyami, B. M. S., R. Tree, J. Kinsella, C. J. Clark, W. H. Reid, D. Cambell, and C. J. Gemmel. 1990. Changes in alveolar macrophage, monocyte, and neutrophil cell. profiles after smoke inhalation injury. J. Clin. Pathol. 43:43-45.
Thom, S. R., I. Mendiguren, T. V. Winkle, D. Fisher, and A. B. Fisher. 1994. Smoke Inhalation with a concurrent systemic stress results in lung alveolar injury. Am. J. Respir. Crit. Care Med. 149:220-226.
Lalonde, C., U. Nayak, J. Hennigan, and R. Demling. 1997. Plasma catalase and glutathione levels are decreased in response to inhalation injury. J. Burn Care Rehabil. 18:515-519.
Leonard, S. S., Wang, S., Shi, X., Jordan, B. S., Castranova, V., and M. A. Dubick. 2000. Wood smoke particles generate free radicals and cause lipid peroxidation, DNA damage, NFκB activation and TNF-± release in macrophages. Toxicology 150:147-157.
Bhattacharyya, S. N., B. Manna, R. Smiley, P. Ashbaugh, R. Coutinho, and B. Kaufman. 1998. Smoke-induced inhalation injury: Effects of retinoic acid and antisense oligodeoxynucleotide on stability and differentiated state of the mucociliary epithelium. Inflammation 22:203-214.
Guzman, K., T. Bader, and P. Nettesheim. 1996. Regulation of MUC5 and MUC1 gene expression: Correlation with airway mucous differentiation. Am. J. Physiol. 270:L846-L853.
Braga, V. M., L. F. Pemberton, T. Duhig, and S. G. Gendler. 1992. Spatial and temporal expression of an epithelial mucin, Muc 1, during mouse development. Development 115:427-437.
Gendler, S., J. Taylor-Papadimitriou, T. Duhig, J. Rothbard, and J. Burchell. 1988. A highly immunogenic region of a human polymorphic epithelial mucin expressed by carcinomas is made up of tandem repeats. J. Biol. Chem. 263:12820-12823.
Lan, M. S., S. K. Batra, W. N. Qi, R. S. Metzgar, and M. A. Hollingsworth. 1990. Cloning and sequencing of a human pancreatic tumor mucin cDNA. J. Biol. Chem. 265:15294-15299.
Gum, J. R., J. C. Byrd, J. W. Hicks, N. W. Toribara, D. T. A. Lamport, and Y. S. Kim. 1989. Molecular cloning of human intestinal mucin cDNAs. J. Biol. Chem. 264:6480-6487.
Gum, J. R., J. W. Hicks, D. M. Swallow, R. L. Legace, J. C. Byrd, D. T. A. Lamport, B. Siddiki, and Y. S. Kim. 1990. Molecular cloning of a novel human intestinal mucin gene. Biochem. Biophys. Res. Commun. 171:407-415.
Moniaux, N., S. Nollet, P. Degand, A. Laine, and J. P. Aubert. 1999. Complete sequence of the human mucin MUC4: A putative cell membrane-associated mucin. Biochem. J. 338:325-333.
Gerard, C., R. L. Eddy, and T. B. Shows. 1990. The core polypeptide of cystic fibrosis tracheal mucin contains a tandem repeat structure. J. Clin. Invest. 86:1921-1927.
Guyonnet-Dupert, V., J. P. Audie, V. Debailleul, A. Laine, M. P. Buisine, S. Galiegue-Zouitina, P. Pigny, P. Degand, J. P. Aubert, and N. Porchet. 1995. Characterization of the human mucin gene MUC5AC: A consensus cystein-rich domain for 11p15 mucin genes? Biochem. J. 305:211-219.
Desseyn, J. L., V. Guyonnet, N. Porchet, J. P. Aubert, and A. Laine. 1997. Human mucin gene MUC5B, the 10.7-kb large central exon encodes various alternate subdomains resulting in a super-repeat. J. Biol Chem. 272:3168-3178.
Bobek, L. A., J. Liu, S. N. Sait, T. B. Shows, Y. A. Bobek, and M. J. Levine. 1996. Structure and chromosomal localization of the human salivary mucin, MUC7. Genomics 31:277-282.
Shankar, V., P. Pichan, R. L. J. Eddy, V. Tonk, N. Nowak, S. N. Sait, T. B. Shows, R. E. Schultz, G. Gotway, R. C. Elkins, M. S. Gilmore, and G. P. Sachdev. 1997. Chromosomal localization of a human mucin gene (MUC8) and cloning of the cDNA corresponding to the carboxy terminus. Am. J. Respir. Cell Mol. Biol. 16:232-241.
Williams, S. J., M. A. Mcguckin, D. C. Gotley, H. G. Eyre, G. R. Sutherland, and T. M. Antalis. 1999. Two novel mucin genes down-regulated in colorectal cancer identified by differential display. Cancer Res. 59:4083-4089.
Khorrami, A. M., A. Choudhury, M. Andrianifahanana, G. C. Varshney, S. N. Bhattacharyya, M. A. Hollingsworth, and S. K. Batra. 2002. Purification and characterization of a human pancreatic adenocarcinoma mucin. J. Biochem. 131:21-29.
Bhattacharyya, S. N., B. C. Veit, B. Manna, J. I. Enriquez, M. P. Walker, M. Khorrami, and B. Kaufman. 1990. Neutral and acidic human tracheobronchial Mucin. Isolation and characterization of core protein. Inflammation 14:355-373.
Moniaux, N., F. Escande, S. K. Batra, N. Porchet, A. Laine, and J. P. Aubert. 2000. Alternative splicing generates a family of putative secreted and membrane-associated MUC4 mucins. Eur. J. Biochem. 267:4536-4544.
Wu, K., N. Fregien, and K. L. Carraway. 1994. Molecular cloning and sequencing of the mucin subunit of a heterodimeric, bifunctional cell surface glycoprotein complex of ascites rat mammary adenocarcinoma cells. J. Biol. Chem. 269:11950-11955.
Inatomi, T., A. S. Tisdale, Q. Zhan, S. Spurr-Michaud, and I. K. Gipson. 1997. Cloning of rat MUC5AC gene: Comparison of its structure and tissue distribution to that of human and mouse homologue. Biochem. Biophys. Res. Commun. 236:789-797.
Ohmori, H., A. F. Dohrman, M. Gallup, T. Tsuda, H. Kai, J. R. Gum, Y. S. Kim, and C. S. Baum. 1994. Molecular cloning of the amino-terminal region of a rat MUC2 mucin gene homologue. J. Biol. Chem. 269:17833-17840.
Rossi, E. A., R. R. Mcneer, S. A. Price-Schiavi, J. M. H. Van Den Brande, M. Komatsu, J. F. Thomson, C. A. C. Carraway, N. L. Freign, and K. L. Carraway. 1996. Sialomucin complex, a heterodimeric glycoprotein complex. J. Biol. Chem. 271:33476-33485.
Price-Sciavi, S. A., C. A. C. Carraway, N. Fregien, and K. L. Carrawy. 1998. Post-transcriptional regulation of a milk membrane protein, the sialomucin complex (ascites sialoglycoprotein (ASGP)-1/ASGP-2. Rat Muc4), by transforming growth factor β. J. Biol. Chem. 273:35228-35237.
Choudhury, A., R. K. Singh, N. Moniaux, T. H. El-Metwally, J.-P. Aubert, and S. K. Batra. 2000. Retinoic acid-dependent transforming growth factor-β2-mediated induction of MUC4 mucin expression in human pancreatic tumor cells follows retinoic acid receptor-± signaling pathway. J. Biol. Chem. 275:33929-33936.
Levine, S. J., P. Larivee, C. Logun, C. W. Angus, F. P. Ognibene, and J. H. Shelhamer. 1995. Tumor necrosis factor-± induces mucin hypersecretion and mucin gene expression by human Airway epithelial cells. Am. J. Respir. Cell Mol. Biol. 12:196-204.
Jany, B., M. Gallup, T. Tsuda, and C. Basbaum. 1991. Mucin gene expression in airways following infection and irritation. Biochem. Biophys. Res. Commun. 181:1-8.
Bhattacharyya, S. N., B. Manna, R. Smiley, P. Ashbaugh, R. Coutinho, and B. Kaufman. 1998. Smoke-induced inhalation injury: Effects of retinoic acid and antisense oligodeoxynucleotide on stability and differentiated state of the mucociliary epithelium. Inflammation 22:203-214.
Borchers, M. T., S. E. Wert, and G. D. Lleikauf. 1998. Acrolein-induced MUC5AC in rat airways. Am. J. Physiol. 18:L573-L581.
Sakano, T., C. V. Okerberg, R. L. Shippe, J. Sanchez, A. D. Maso, and B. A. Pruitt. 1993. A rabbit model of inhalation injury. J. Trauma 34:411-416.
Levin, B. S., A. J. Fowell, M. M. Birky, M. Paabo, A. Stolte, and D. Malek. 1982. In: Further Development of a Test Method for the Assessment of the Acute Inhalation Toxicity of Combustion Products, National Bureau of Standards (publication 82–2532), Washington, DC.
Chomczynski, P. and N. Sacchi. 1987. Single-step method of RNA isolation by guanidine thiocyanate-phenol-chloroform extraction. Anal. Biochem. 162:156-159.
Manna, B., M. Lund, P. Ashbaugh, B. Kaufman, and S. N. Bhattacharyya. 1994. Effect of retinoic acid on mucin gene expression in rat airways in vitro. Biochem. J. 297:309-313.
Naito, C., M. Kawamura, and Y. Ymamot. 1993. Lipid peroxides as the initiation factor of atherosclerosis. Ann. N. Y. Acad. Sci. 676:27-45.
Benzie, I. F. F. and J. J. Strain. 1996. Ferric reducing ability of plasma (FRAP) as a measure of antioxidant power: The FRAP assay. Anal. Biochem. 239:70-76.
Lawrence, R. A. and R. F. Burk. 1976. Glutathione peroxidase activity in selenium-deficient rat liver. Biochem. Biophys. Res. Commun. 71:952-958.
Rogers, K. M. and R. C. Augusteyn. 1978. Glutathione reductase in normal and cataractous human lenses. Exp. Eye Res. 27:719-721.
Marklund, S. and G. Marklund. 1974. Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur. J. Biochem. 47:469-479.
Aebi, H. 1984. Catalase in vitro. Methods Enzymol. 105:121-126.
Anderson, M. E. 1985. Determination of glutathione and glutathione disulfide in biological samples. Methods Enzymol. 113:548-555.
Trush, M. A., P. A. Egner, and T. W. Kensler. 1994. Myeloperoxidase as a biomarker of skin irritation and inflammation. Food Chem. Toxicol. 32:143-147.
Shimaju, T., H. Ikeuchi, G. B. Hubbard, P. C. Langlinais, A. D. Mason, and B. A. Pruitt. 1990. Smoke inhalation injury and the effect of carbon monoxide in the sheep model. J. Trauma 30:170-175.
Hunt, J. L., R. N. Agee, and B. A. Pruitt. 1975. Fiberoptic bronchoscopy in acute inhalation injury. J. Trauma 15:641-646.
Demling, R. H. and C. Lalonde. 1990. Moderate smoke inhaltion produces decreased oxygen delivery, increased oxygen demands, and systemic but not lung parenchymal lipid peroxidation. Surgery 108:544-552.
Demling, R., C. Lalonde, L. Picard, and J. Blanchard. 1994. Changes in lung and systemic oxidant and antioxidant activity after smoke inhalation. Shock 1:101-107.
Dubick, M. A., S. C. Carden, B. S. Jordan, P. C. Langlinais, and D. W. Mozingo,2002. Indices of antioxidant status in rats subjected to wood smoke inhaltion and/or thermal injury. Toxicology 176:145-157.
Lalonde, C., L. Picard, C. Cambell, and R. Demling. 1994. Lung and systemic oxidant and antioxidant activity after graded smoke exposure in the rat. Circ. Shock 42:7-13.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bhattacharyya, S.N., Dubick, M.A., Yantis, L.D. et al. In Vivo Effect of Wood Smoke on the Expression of Two Mucin Genes in Rat Airways. Inflammation 28, 67–76 (2004). https://doi.org/10.1023/B:IFLA.0000033022.66289.04
Issue Date:
DOI: https://doi.org/10.1023/B:IFLA.0000033022.66289.04