Abstract
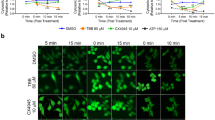
Transfection of PC12 pheochromocytoma cells with bcl-2 potentiates apoptosis induced by the antimitotic agent, neocarzinostatin (NCS). The mechanism of potentiation involves caspase 3-dependent cleavage of Bcl-2 to its pro-apoptotic counterpart, but the cellular events proximal to caspase 3 activation in this system are not known. Two min after initiation of NCS treatment, Bax begins to translocate from cytosol to the mitochondria; the mitochondrial localization of Bax persists for 30 min after NCS treatment. At the same time, cytochrome C is released from the mitochondria to cytosol. The mitochondrial membrane potential exhibits differential change in mock- and bcl-2-transfected PC12 cells. In mock-transfected PC12 cells, the mitochondrial membrane potential increases immediately, peaks at 15 min following initiation of NCS treatment, and drops thereafter. In contrast, in bcl-2-transfected PC12 cells, the membrane potential drops immediately following NCS treatment. Caspase 9 is activated and peaks at 10 min in both mock- and bcl-2 transfected PC12 cells, however, the peak activity of caspase 9 is higher and caspase 9 activation lasts longer (30 min) after the treatment in bcl-2 transfectants. Not until 30 min after initiation of a 1 h treatment with NCS is Bcl-2 protein cleaved in bcl-2-transfected cells. Thus, in bcl-2-transfected cells, the mitochondrial membrane potential drops and cytochrome C is released from the mitochondria despite the presence of large amounts of intact mitochondrial Bcl-2. This makes it unlikely that cleavage of Bcl-2 is the only factor involved in potentiation of NCS-induced apoptosis by Bcl-2.
Similar content being viewed by others
References
Dole M, Nunez G, Merchant AK, et al. Bcl-2 inhibits chemotherapy-induced apoptosis in neuroblastoma. Cancer Res 1994; 54: 3253–3259.
Miyashita T, Reed JC. Bcl-2 oncoprotein blocks chemotherapyinduced apoptosis in a human leukemia cell line. Blood 1993; 81: 151–157.
Teixeira C, Reed JC, Pratt MA. Estrogen promotes chemotherapeutic drug resistance by a mechanism involving Bcl-2 protooncogene expression in human breast cancer cells. Cancer Res 1995; 55: 3902–3907.
Kamesaki S, Kamesaki H, Jorgensen TJ, Tanizawa A, Pommier Y, Cossman J. Bcl-2 protein inhibits etoposide-induced apoptosis through its effects on events subsequent to topoisomerase II-induced DNA strand breaks and their repair. Cancer Res 1993; 53: 4251–4256.
Cortazzo M, Schor NF. Potentiation of enediyne-induced apoptosis and differentiation by Bcl-2. Cancer Res 1996; 56: 1199–1203.
Schor NF, Tyurina YY, Fabisiak JP, Tyurin VA, Lazo JS, Kagan VE. Selective oxidation and externalization of membrane phosphatidylserine: Bcl-2-induced potentiation of the final common pathway for apoptosis. Brain Res 1999; 831: 125–130.
Tyurina YY, Tyurin VA, Carta G, Quinn PJ, Schor NF, Kagan VE. Direct evidence for antioxidant effect of Bcl-2 in PC12 rat pheochromocytoma cells. Arch Biochem Biophys 1997; 344: 413–423.
Schor NF, Tyurina YY, Tyurin VA, Kagan VE. Differential membrane antioxidant effects of immediate and long-term estradiol treatment of MCF-7 breast cancer cells. Biochem Biophys Res Commun 1999; 260: 410–415.
Schor NF, Rudin CM, Hartman AR, Thompson CB, Tyurina YY, Kagan VE. Cell line dependence of Bcl-2-induced alteration of glutathione handling. Oncogene 2000; 19: 472–476.
Zoratti M, Szabo I. The mitochondrial permeability transition. Biochim Biophys Acta 1995; 1241: 139–176.
Bernardi P. The permeability transition pore. Control points of a cyclosporin A-sensitive mitochondrial channel involved in cell death. Biochim Biophys Acta 1996; 1275: 5–9.
Bernardi P, Petronilli V. The permeability transition pore as a mitochondrial calcium release channel: A critical appraisal. J Bioenerg Biomembr 1996; 28: 131–138.
Vercesi AE, Kowaltowski AJ, Grijalba MT, Meinicke AR, Castilho RF. The role of reactive oxygen species in mitochondrial permeability transition. Biosci Rep 1997; 17: 43–52.
Kowaltowski AJ, Vercesi AE, Fiskum G. Bcl-2 prevents mitochondrial permeability transition and cytochrome c release via maintenance of reduced pyridine nucleotides. Cell Death Differ 2000; 7: 903–910.
Kroemer G. The mitochondrion as an integrator/coordinator of cell death pathways. Cell Death Differ 1998; 5: 547.
Kane DJ, Sarafian TA, Anton R, et al. Bcl-2 inhibition of neural death: Decreased generation of reactive oxygen species. Science 1993; 262: 1274–1277.
Liang Y, Nylander KD, Yan C, Schor NF. Role of caspase 3-dependent Bcl-2 cleavage in potentiation of apoptosis by Bcl-2. Mol Pharmacol 2002; 61: 142–149.
Liang Y, Nylander KD, Schor NF. Requirement for caspase 3 in potentiation of apoptosis by Bcl-2. Pediatr Res 2001; 49: 211A.
Hartsell TL, Yalowich JC, Ritke MK, Martinez AJ, Schor NF. Induction of apoptosis in murine and human neuroblastoma cell lines by the enediyne natural product neocarzinostatin. J Pharmacol Exp Ther 1995; 275: 479–485.
Hartsell TL, Hinman LM, Hamann PR, Schor NF. Determinants of the response of neuroblastoma cells to DNA damage: The roles of pre-treatment cell morphology and chemical nature of the damage. J Pharmacol Exp Ther 1996; 277: 1158–1166.
Kroemer G, Reed JC. Mitochondrial control of cell death. Nat Med 2000; 6: 513–519.
Shimizu S, Tsujimoto Y. Proapoptotic BH3-only Bcl-2 family members induce cytochrome c release, but not mitochondrial membrane potential loss, and do not directly modulate voltage-dependent anion channel activity. Proc Natl Acad Sci USA 2000; 97: 577–582.
Madesh M, Hajnoczky G. VDAC-dependent permeabilization of the outer mitochondrial membrane by superoxide induces rapid and massive cytochrome c release. J Cell Biol 2001; 155: 1003–1015.
Debatin KM, Poncet D, Kroemer G. Chemotherapy: Targeting the mitochondrial cell death pathway. Oncogene 2002; 21: 8786–8803.
Banki K, Hutter E, Gonchoroff NJ, Perl A. Elevation of mitochondrial transmembrane potential and reactive oxygen intermediate levels are early events and occur independently from activation of caspases in Fas signaling. J Imrnunol 1999; 162: 1466–1479.
Li PF, Dietz R, von Harsdorf R. p53 regulates mitochondrial membrane potential through reactive oxygen species and induces cytochrome c-independent apoptosis blocked by Bcl-2. Embo J 1999; 18: 6027–6036.
Gergely P, Jr, Grossman C, Niland B, et al. Mitochondrial hyperpolarization and ATP depletion in patients with systemic lupus erythematosus. Arthritis Rheum 2002; 46: 175–190.
Ott M, Robertson JD, Gogvadze V, Zhivotovsky B, Orrenius S. Cytochrome c release from mitochondria proceeds by a two-step process. Proc Natl Acad Sci USA 2002; 99: 1259–1263.
Povirk LF, Goldberg IH. Stoichiometric uptake of molecular oxygen and consumption of sulfhydryl groups by neocarzinostatin chromophore bound to DNA. J Biol Chem 1983; 258: 11763–11767.
Harris MH, Thompson CB. The role of the Bcl-2 family in the regulation of outer mitochondrial membrane permeability. Cell Death Differ 2000; 7: 1182–1191.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liang, Y., Yan, C., Nylander, K.D. et al. Early events in Bcl-2-enhanced apoptosis. Apoptosis 8, 609–616 (2003). https://doi.org/10.1023/A:1026135509274
Issue Date:
DOI: https://doi.org/10.1023/A:1026135509274