Abstract
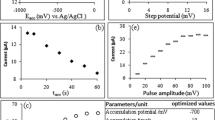
We studied the effects of homeopathically potentiated substances in ultralow doses on physicochemical properties of solutions. Inverse voltammetric signals during Hg(II) oxidation-reduction in the presence of potentiated water, lithium chloride, and mercury nitrate were studied by alternating-current inversion voltammetry. Potentiated substances modified oxidation-reduction processes in the electrochemical system. Potentiated solutions of Hg(II) increased the effective concentration (activity) of mercury ions. The effect was most significant in the presence of mercury nitrate in homeopathic dilutions C6+C12+C24. Our results indicate that potentiated substances specifically modulate kinetic and thermodynamic characteristics of solutions containing the same compounds.
Similar content being viewed by others

REFERENCES
O. I. Epstein, Byull. Sib. Otd. Ros. Akad. Med. Nauk, No. 1, 132-149 (1999).
Kh. Z. Brainina and E. Ya. Neiman, Solid-Phase Reactions in Electroanalytic Chemistry [in Russian], Moscow (1982).
S. I. Petrov and Zh. V. Ivanova, Zh. Analit. Khimii, 55,No. 11, 1224-1227 (2000).
Y. Keri, Search for General Principles in Development of the Earth and Cosmos [in Russian], Moscow (1991).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Petrov, S.I., Epstein, O.I. Effect of Potentiated Solutions on Mercury(II) Signal in Inversion Voltammetry. Bulletin of Experimental Biology and Medicine 135 (Suppl 7), 99–101 (2003). https://doi.org/10.1023/A:1024707519510
Issue Date:
DOI: https://doi.org/10.1023/A:1024707519510