Abstract
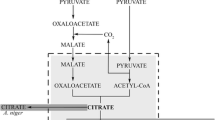
Intracellular reactive oxygen species (iROS) induction by HOCl was used as a novel strategy to improve enzyme productivities in Aspergillus niger growing in a bioreactor. With induced iROS, the specific intracellular activities of α-amylase, protease, catalase, and glucose oxidase were increased by about 170%, 250%, 320%, and 260%, respectively. The optimum specific iROS level for achieving maximum cell concentration and enzyme production was about 15 mmol g cell−1. The type of iROS inducing the enzyme production was identified to be a derivative of the superoxide radical.
Similar content being viewed by others
References
Beer R, Sizer F (1951) A spectrometric method for the breakdown of hydrogen peroxide. J. Biol. Chem. 226: 133–139.
Boll M, Messner B (1994) Cell suspension cultures of spruce (Picea abeis) inactivation of extracellular enzymes by fungal elicitorinduced transient release of H2O2 (oxidative burst). Plant Tiss. Org. Cult. 39: 69–78.
Borg DC (1976) Applications of electron spin resonance in biology. In: Pryor WA, ed. Free Radicals in Biology, Vol. 1. New York: Academic Press, pp. 69–147.
Eremin AN, Metelitsa DI, Moroz IV, Pavlovsakin Z, Mikhailova RV (2002) Kinetic characteristics of extracellular catalase from Penicillium piceum F-648 and variants of fungi. Prikladnaia biokem. Mikrobiol. 38: 374–380.
Halliwell B, Gutteridge JMC (1999) Free Radicals in Biology and Medicine, 3rd edn. Oxford: Oxford University Press.
Kelly RL, Reddy CA (1986) Identification of glucose oxidase activity as the primary source of hydrogen peroxide production in lingnolytic cultures of Phanerochaete chrysosporium. Arch. Microbiol. 144: 248–253.
Lee J, Dawes IW, Roe JH (1995) Adaptive response of Schizosaccharomyces pombe to hydrogen peroxide and menadione. Microbiology 141: 3127–3132.
ManjulaRao Y, Sureshkumar GK (2001) Improvement in bioreactor productivities using free radicals: HOCl-induced overproduction of xanthan gum from Xanthomonas campestris and its mechanism. Biotechnol. Bioeng. 72: 62–68.
Martindale JL, Holbrook J (2002) Cellular response to oxidative stress: signaling for suicide and survival. J Cell. Physiol. 192: 1–15.
Okuno S, Shimizu S, Ito T, Nomura M, Hamada E, Tsujimoto Y, Mastuda H (1998) Bcl-2 prevents the caspase-independent cell death. J. Biol. Chem. 273: 34272–34277.
Pryor WA (1976) The role of free radical reactions in biological systems. In: Pryor WA, ed. Free Radicals in Biology, Vol. 1. New York: Academic Press, pp. 1–49.
Rawool S, Sahoo S, Rao KK, Sureshkumar GK (2001) Involvement of enzyme productivities from mold cultivations using liquid phase oxygen supply strategy. Biotechnol. Prog. 17: 832–837.
Sarath G, Motte De La RS, Wagner FW (1989) Protease assay methods. In: Beynon RJ, Bond JS, eds. Proteolytic Enzymes: A Practical Approach. Oxford: IRL Press, pp. 25–55.
Schaur JR, Jerlich A, Stelmaszynska T (1998) Hypochlorous acid as reactive oxygen species. Curr. Top. Biophys. 22: 176–185.
Sriram G, ManjulaRao Y, Suresh AK, Sureshkumar GK (1998) Oxygen supply without gas-liquid film resistance to Xanthomonas campestris cultivation. Biotechnol. Bioeng. 59: 714–723.
Weickert MJ, Chambliss JH (1989) Genetic analysis of the promoter region of the Bacillus subtilis ?-amylase gene. J. Bacteriol. 171: 3656–3666.
Zweier JL (1988) Measurement of superoxide-derived free radicals in the reperfused heart. J. Biol. Chem. 263: 1353–1357.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sahoo, S., Krishnamurthy Rao, K. & Suraishkumar, G. Induced reactive oxygen species improve enzyme production from Aspergillus niger cultivation. Biotechnology Letters 25, 821–825 (2003). https://doi.org/10.1023/A:1023508609623
Issue Date:
DOI: https://doi.org/10.1023/A:1023508609623